GENOSYL- nitric oxide gas
GENOSYL by
Drug Labeling and Warnings
GENOSYL by is a Prescription medication manufactured, distributed, or labeled by VERO BIOTECH LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use GENOSYL® safely and effectively. See full prescribing information for GENOSYL.GENOSYL (nitric oxide), for inhalation use
Initial U.S. Approval: 1999INDICATIONS AND USAGE
GENOSYL® is a vasodilator indicated to improve oxygenation and reduce the need for extracorporeal membrane oxygenation in term and near-term (>34 weeks gestation) neonates with hypoxic respiratory failure associated with clinical or echocardiographic evidence of pulmonary hypertension in conjunction with ventilatory support and other appropriate agents (1).
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
GENOSYL (nitric oxide) is a gas, available at concentrations up to 800 ppm. (3)
CONTRAINDICATIONS
Neonates dependent on right-to-left shunting of blood (4).
WARNINGS AND PRECAUTIONS
Rebound Pulmonary Hypertension: Abrupt discontinuation of GENOSYL may lead to worsening oxygenation and increasing pulmonary artery pressure (5.1).
Methemoglobinemia: Methemoglobin increases with the dose of nitric oxide; following discontinuation or reduction of nitric oxide, methemoglobin levels return to baseline over a period of hours (5.2).
Elevated NO2 Levels: Monitor NO2 levels (5.3).
Heart Failure: In patients with pre-existing left ventricular dysfunction, GENOSYL may increase pulmonary capillary wedge pressure leading to pulmonary edema (5.4).
ADVERSE REACTIONS
The most common adverse reaction is hypotension (6).
To report SUSPECTED ADVERSE REACTIONS, contact Vero Biotech at 1-877-337-4118 and http://www.vero-biotech.com/ or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Nitric oxide donor compounds may increase the risk of developing methemoglobinemia (7).
Revised: 12/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dosage
2.2 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Rebound Pulmonary Hypertension Syndrome following Abrupt Discontinuation
5.2 Hypoxemia from Methemoglobinemia
5.3 Airway Injury from Nitrogen Dioxide
5.4 Worsening Heart Failure
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Nitric Oxide Donor Agents
8 USE IN SPECIFIC POPULATIONS
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Treatment of Hypoxic Respiratory Failure (HRF)
14.2 Ineffective in Adult Respiratory Distress Syndrome (ARDS)
14.3 Ineffective in Prevention of Bronchopulmonary Dysplasia (BPD)
16 HOW SUPPLIED/STORAGE AND HANDLING
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
GENOSYL® is indicated to improve oxygenation and reduce the need for extracorporeal membrane oxygenation in term and near-term (>34 weeks gestation) neonates with hypoxic respiratory failure associated with clinical or echocardiographic evidence of pulmonary hypertension in conjunction with ventilatory support and other appropriate agents.
-
2 DOSAGE AND ADMINISTRATION
2.1 Dosage
Term and near-term neonates with hypoxic respiratory failure
The recommended dose of GENOSYL is 20 ppm. Maintain treatment up to 14 days or until the underlying oxygen desaturation has resolved and the neonate is ready to be weaned from GENOSYL therapy.
Doses greater than 20 ppm are not recommended [see Warnings and Precautions (5.2)].
2.2 Administration
Nitric Oxide Delivery System
GENOSYL must be administered using a calibrated GENOSYL Delivery System. Only validated ventilator systems should be used in conjunction with GENOSYL [see Description (11)].
Consult the GENOSYL Delivery System Operator’s Manual or call 1-877-337-4118 or visit www.vero-biotech.com for needed information on training and technical support for users of GENOSYL with the GENOSYL Delivery System .
Keep available a backup power supply to address power failures. The GENOSYL Delivery System consists of a primary system and a fully functional second system that can be used as backup in the event of primary system failure.
Monitoring
Measure methemoglobin within 4-8 hours after initiation of treatment with GENOSYL and periodically throughout treatment [see Warnings and Precautions (5.2)].
Monitor for PaO2 and inspired NO2 during GENOSYL administration [see Warnings and Precautions (5.3)].
The concentration of nitric oxide, nitrogen dioxide and air is constantly monitored. The GENOSYL Delivery System will shutdown if nitrogen dioxide reaches 3 ppm.
Weaning and Discontinuation
Avoid abrupt discontinuation of GENOSYL [see Warnings and Precautions (5.1)]. To wean GENOSYL, down titrate in several steps, pausing several hours at each step to monitor for hypoxemia.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Rebound Pulmonary Hypertension Syndrome following Abrupt Discontinuation
Wean from GENOSYL [see Dosage and Administration (2.2)]. Abrupt discontinuation of GENOSYL may lead to worsening oxygenation and increasing pulmonary artery pressure, i.e., Rebound Pulmonary Hypertension Syndrome. Signs and symptoms of Rebound Pulmonary Hypertension Syndrome include hypoxemia, systemic hypotension, bradycardia, and decreased cardiac output. If Rebound Pulmonary Hypertension occurs, reinstate GENOSYL therapy immediately.
5.2 Hypoxemia from Methemoglobinemia
Nitric oxide combines with hemoglobin to form methemoglobin, which does not transport oxygen. Methemoglobin levels increase with the dose of GENOSYL; it can take 8 hours or more before steady-state methemoglobin levels are attained. Monitor methemoglobin and adjust the dose of GENOSYL to optimize oxygenation.
If methemoglobin levels do not resolve with decrease in dose or discontinuation of GENOSYL, additional therapy may be warranted to treat methemoglobinemia [see Overdosage (10)].
5.3 Airway Injury from Nitrogen Dioxide
Nitrogen dioxide (NO2) forms in gas mixtures containing NO and O2. Nitrogen dioxide may cause airway inflammation and damage to lung tissues.
If there is an unexpected change in NO2 concentration, or if the NO2 concentration reaches 0.5 ppm when measured in the breathing circuit, then the delivery system should be assessed in accordance with the GENOSYL Delivery System Operator's Manual troubleshooting section, and the NO2 analyzer should be recalibrated. The dose of GENOSYL and/or FiO2 should be adjusted as appropriate.
5.4 Worsening Heart Failure
Patients with left ventricular dysfunction treated with GENOSYL may experience pulmonary edema, increased pulmonary capillary wedge pressure, worsening of left ventricular dysfunction, systemic hypotension, bradycardia and cardiac arrest. Discontinue GENOSYL while providing symptomatic care.
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed elsewhere in the label;
Hypoxemia [see Warnings and Precautions (5.2)]
Worsening Heart Failure [see Warnings and Precautions (5.4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The adverse reaction information from the clinical studies does, however, provide a basis for identifying the adverse events that appear to be related to drug use and for approximating rates.
Controlled studies have included 325 patients on nitric oxide doses of 5 to 80 ppm and 251 patients on placebo. Total mortality in the pooled trials was 11% on placebo and 9% on nitric oxide gas for inhalation, a result adequate to exclude nitric oxide mortality being more than 40% worse than placebo.
In both the NINOS and CINRGI studies, the duration of hospitalization was similar in nitric oxide gas for inhalation and placebo-treated groups.
From all controlled studies, at least 6 months of follow-up is available for 278 patients who received nitric oxide gas and 212 patients who received placebo. Among these patients, there was no evidence of an adverse effect of treatment on the need for re-hospitalization, special medical services, pulmonary disease, and neurological sequelae.
In the NINOS study, treatment groups were similar with respect to the incidence and severity of intracranial hemorrhage, Grade IV hemorrhage, periventricular leukomalacia, cerebral infarction, seizures requiring anticonvulsant therapy, pulmonary hemorrhage, or gastrointestinal hemorrhage.
In CINRGI, the only adverse reaction (>2% higher incidence on nitric oxide gas for inhalation than on placebo) was hypotension (14% vs. 11%).
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.4 Pediatric Use
The safety and efficacy of nitric oxide for inhalation has been demonstrated in term and near-term neonates with hypoxic respiratory failure associated with evidence of pulmonary hypertension [see Clinical Studies (14.1)]. Additional studies conducted in premature neonates for the prevention of bronchopulmonary dysplasia have not demonstrated substantial evidence of efficacy [see Clinical Studies (14.3)]. No information about its effectiveness in other age populations is available.
-
10 OVERDOSAGE
Overdosage with nitric oxide gas is manifest by elevations in methemoglobin and pulmonary toxicities associated with inspired NO2. Elevated NO2 may cause acute lung injury. Elevations in methemoglobin reduce the oxygen delivery capacity of the circulation. In clinical studies, NO2 levels >3 ppm or methemoglobin levels >7% were treated by reducing the dose of, or discontinuing, nitric oxide gas.
Methemoglobinemia that does not resolve after reduction or discontinuation of therapy can be treated with intravenous vitamin C, intravenous methylene blue, or blood transfusion, based upon the clinical situation.
-
11 DESCRIPTION
GENOSYL (nitric oxide) is administered by inhalation. Nitric oxide is a pulmonary vasodilator. Nitric oxide is generated from liquid dinitrogen tetroxide (N2O4) by the cassette in the GENOSYL Delivery System. Upon initiation of GENOSYL Delivery System, the liquid N2O4 is heated and the equilibrium shifts to nitrogen dioxide (NO2) gas. The NO2 is then converted into nitric oxide (NO) using the antioxidant cartridges, and nitric oxide is delivered to the patient by means of a ventilator or a nasal cannula. The amount of nitric oxide administered to the patient is set by controlling the temperature of the N2O4 liquid module, which controls the pressure inside the liquid module, which in turn controls the mass of NO2 that is sent to the primary cartridges, and hence the mass of nitric oxide. The mass flow of nitric oxide, together with the air from the pump, control the nitric oxide concentration. A nitric oxide sensor monitors the nitric oxide in the patient line. GENOSYL Delivery System is designed to deliver a controlled level of nitric oxide blended with breathing air or oxygen-enriched breathing air.
The GENOSYL Delivery System controls the flow of nitric oxide mixed with air delivered to the patient.
The structural formula of nitric oxide (NO) is shown below:
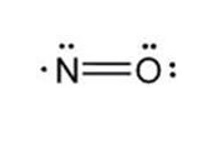
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Nitric oxide relaxes vascular smooth muscle by binding to the heme moiety of cytosolic guanylate cyclase, activating guanylate cyclase and increasing intracellular levels of cyclic guanosine 3',5'-monophosphate, which then leads to vasodilation. When inhaled, nitric oxide selectively dilates the pulmonary vasculature, and because of efficient scavenging by hemoglobin, has minimal effect on the systemic vasculature.
GENOSYL appears to increase the partial pressure of arterial oxygen (PaO2) by dilating pulmonary vessels in better ventilated areas of the lung, redistributing pulmonary blood flow away from lung regions with low ventilation/perfusion (V/Q) ratios toward regions with normal ratios.
12.2 Pharmacodynamics
Effects on Pulmonary Vascular Tone in PPHN
Persistent pulmonary hypertension of the newborn (PPHN) occurs as a primary developmental defect or as a condition secondary to other diseases such as meconium aspiration syndrome (MAS), pneumonia, sepsis, hyaline membrane disease, congenital diaphragmatic hernia (CDH), and pulmonary hypoplasia. In these states, pulmonary vascular resistance (PVR) is high, which results in hypoxemia secondary to right-to-left shunting of blood through the patent ductus arteriosus and foramen ovale. In neonates with PPHN, nitric oxide gas for inhalation improves oxygenation (as indicated by significant increases in PaO2).
12.3 Pharmacokinetics
The pharmacokinetics of nitric oxide has been studied in adults.
Absorption and Distribution
Nitric oxide is absorbed systemically after inhalation. Most of it traverses the pulmonary capillary bed where it combines with hemoglobin that is 60% to 100% oxygen-saturated. At this level of oxygen saturation, nitric oxide combines predominantly with oxyhemoglobin to produce methemoglobin and nitrate. At low oxygen saturation, nitric oxide can combine with deoxyhemoglobin to transiently form nitrosylhemoglobin, which is converted to nitrogen oxides and methemoglobin upon exposure to oxygen. Within the pulmonary system, nitric oxide can combine with oxygen and water to produce nitrogen dioxide and nitrite, respectively, which interact with oxyhemoglobin to produce methemoglobin and nitrate. Thus, the end products of nitric oxide that enter the systemic circulation are predominantly methemoglobin and nitrate.
Metabolism
Methemoglobin disposition has been investigated as a function of time and nitric oxide exposure concentration in neonates with respiratory failure. The methemoglobin (MetHb) concentration-time profiles during the first 12 hours of exposure to 0, 5, 20, and 80 ppm nitric oxide are shown in Figure 1.
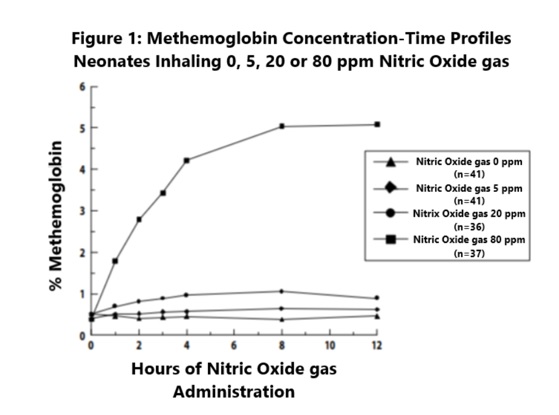
Methemoglobin concentrations increased during the first 8 hours of nitric oxide exposure. The mean methemoglobin level remained below 1% in the placebo group and in the 5 ppm and 20 ppm nitric oxide gas groups, but reached approximately 5% in the 80 ppm nitric oxide gas group. Methemoglobin levels >7% were attained only in patients receiving 80 ppm, where they comprised 35% of the group. The average time to reach peak methemoglobin was 10 ± 9 (SD) hours (median, 8 hours) in these 13 patients, but one patient did not exceed 7% until 40 hours.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No evidence of a carcinogenic effect was apparent, at inhalation exposures up to the recommended dose (20 ppm), in rats for 20 hr/day for up to two years. Higher exposures have not been investigated.
Nitric oxide gas has demonstrated genotoxicity in Salmonella (Ames Test), human lymphocytes, and after in vivo exposure in rats. There are no animal or human studies to evaluate nitric oxide for effects on fertility.
-
14 CLINICAL STUDIES
14.1 Treatment of Hypoxic Respiratory Failure (HRF)
The efficacy of nitric oxide gas was investigated in term and near-term newborns with hypoxic respiratory failure (HRF) resulting from a variety of etiologies. Inhalation of nitric oxide gas reduces the oxygenation index (OI= mean airway pressure in cm H2O × fraction of inspired oxygen concentration [FiO2]× 100 divided by systemic arterial concentration in mm Hg [PaO2]) and increases PaO2 [see Clinical Pharmacology (12.1)].
NINOS Study
The Neonatal Inhaled Nitric Oxide Study (NINOS) was a double-blind, randomized, placebo-controlled, multicenter trial in 235 neonates with hypoxic respiratory failure. The objective of the study was to determine whether inhaled nitric oxide would reduce the occurrence of death and/or initiation of extracorporeal membrane oxygenation (ECMO) in a prospectively defined cohort of term or near-term neonates with hypoxic respiratory failure unresponsive to conventional therapy. Hypoxic respiratory failure was caused by meconium aspiration syndrome (MAS; 49%), pneumonia/sepsis (21%), idiopathic primary pulmonary hypertension of the newborn (PPHN; 17%), or respiratory distress syndrome (RDS; 11%). Infants 14 days of age (mean, 1.7 days) with a mean PaO2 of 46 mm Hg and a mean oxygenation index (OI) of 43 cm H2O / mm Hg were initially randomized to receive 100% O2 with (n=114) or without (n=121) 20 ppm nitric oxide for up to 14 days. Response to study drug was defined as a change from baseline in PaO2 30 minutes after starting treatment (full response = >20 mm Hg, partial = 10-20 mm Hg, no response = <10 mm Hg). Neonates with a less than full response were evaluated for a response to 80 ppm nitric oxide or control gas. The primary results of this study are presented in Table 1.
Table 1: Summary of Clinical Results from Hypoxic Respiratory Failure Study Control (n=121) Nitric Oxide gas (n=114) P value - * Death or need for ECMO was the primary end point of this study
- † Extracorporeal membrane oxygenation
Death or ECMO*† 77 (64%) 52 (46%) 0.006 Death 20 (17%) 16 (14%) 0.60 ECMO 66 (55%) 44 (39%) 0.014 Although the incidence of death by 120 days of age was similar in both groups (NO, 14%; control 17%), significantly fewer infants in the nitric oxide group required ECMO compared with controls (39% vs. 55%, p = 0.014). The combined incidence of death and/or initiation of ECMO showed a significant advantage for the nitric oxide treated group (46% vs. 64%, p = 0.006). The nitric oxide group also had significantly greater increases in PaO2 and greater decreases in the OI and the alveolar-arterial oxygen gradient than the control group (p<0.001 for all parameters). Significantly more patients had at least a partial response to the initial administration of study drug in the nitric oxide group (66%) than the control group (26%, p<0.001). Of the 125 infants who did not respond to 20 ppm nitric oxide control, similar percentages of NO-treated (18%) and control (20%) patients had at least a partial response to 80 ppm nitric oxide gas for inhalation or control drug, suggesting a lack of additional benefit for the higher dose of nitric oxide. No infant had study drug discontinued for toxicity. Inhaled nitric oxide gas had no detectable effect on mortality. The adverse events collected in the NINOS trial occurred at similar incidence rates in both treatment groups [see Adverse Reactions (6.1)]. Follow-up exams were performed at 18-24 months for the infants enrolled in this trial. In the infants with available follow-up, the two treatment groups were similar with respect to their mental, motor, audiologic, or neurologic evaluations.
CINRGI Study
This study was a double-blind, randomized, placebo-controlled, multi-center trial of 186 term and near-term neonates with pulmonary hypertension and hypoxic respiratory failure. The primary objective of the study was to determine whether nitric oxide gas would reduce the receipt of ECMO in these patients. Hypoxic respiratory failure was caused by MAS (35%), idiopathic PPHN (30%), pneumonia/sepsis (24%), or RDS (8%). Patients with a mean PaO2 of 54 mm Hg and a mean OI of 44 cm H2O / mm Hg were randomly assigned to receive either 20 ppm nitric oxide gas (n=97) or nitrogen gas (placebo; n=89) in addition to their ventilatory support. Patients who exhibited a PaO2 >60 mm Hg and a pH < 7.55 were weaned to 5 ppm nitric oxide gas or placebo. The primary results from the CINRGI study are presented in Table 2.
Table 2: Summary of Clinical Results from Persistent Pulmonary Hypertension of the Newborn Study Placebo Nitric oxide gas P value - * ECMO was the primary end point of this study
- † Extracorporeal membrane oxygenation
ECMO*† 51/89 (57%) 30/97 (31%) <0.001 Death 5/89 (6%) 3/97 (3%) 0.48 Significantly fewer neonates in the nitric oxide gas group required ECMO compared to the control group (31% vs. 57%, p<0.001). While the number of deaths were similar in both groups (Nitric oxide gas, 3%; placebo, 6%), the combined incidence of death and/or receipt of ECMO was decreased in the nitric oxide gas group (33% vs. 58%, p<0.001).
In addition, the nitric oxide gas group had significantly improved oxygenation as measured by PaO2, OI, and alveolar-arterial gradient (p<0.001 for all parameters). Of the 97 patients treated with nitric oxide gas, 2 (2%) were withdrawn from study drug due to methemoglobin levels >4%. The frequency and number of adverse events reported were similar in the two study groups [see Adverse Reactions (6.1)].
In clinical trials, reduction in the need for ECMO has not been demonstrated with the use of inhaled nitric oxide in neonates with congenital diaphragmatic hernia (CDH).
14.2 Ineffective in Adult Respiratory Distress Syndrome (ARDS)
In a randomized, double-blind, parallel, multicenter study, 385 patients with adult respiratory distress syndrome (ARDS) associated with pneumonia (46%), surgery (33%), multiple trauma (26%), aspiration (23%), pulmonary contusion (18%), and other causes, with PaO2/FiO2 <250 mm Hg despite optimal oxygenation and ventilation, received placebo (n=193) or nitric oxide gas (n=192), 5 ppm, for 4 hours to 28 days or until weaned because of improvements in oxygenation. Despite acute improvements in oxygenation, there was no effect of nitric oxide gas on the primary endpoint of days alive and off ventilator support. These results were consistent with outcome data from a smaller dose ranging study of nitric oxide (1.25 to 80 ppm). GENOSYL (nitric oxide) for inhalation is not indicated for use in ARDS.
14.3 Ineffective in Prevention of Bronchopulmonary Dysplasia (BPD)
The safety and efficacy of nitric oxide gas for the prevention of chronic lung disease [bronchopulmonary dysplasia (BPD)] in neonates ≤ 34 weeks gestational age requiring respiratory support has been studied in four large previously conducted multicenter, double-blind, placebo-controlled clinical trials in a total of 2,600 preterm infants. Of these, 1,290 received placebo, and 1,310 received inhaled nitric oxide at doses ranging from 5-20 ppm, for treatment periods of 7-24 days duration. The primary endpoint for these studies was alive and without BPD at 36 weeks postmenstrual age (PMA). The need for supplemental oxygen at 36 weeks PMA served as a surrogate endpoint for the presence of BPD. Overall, efficacy for the prevention of bronchopulmonary dysplasia in preterm infants was not established. There were no meaningful differences between treatment groups with regard to overall deaths, methemoglobin levels, or adverse events commonly observed in premature infants, including intraventricular hemorrhage, patent ductus arteriosus, pulmonary hemorrhage, and retinopathy of prematurity.
The use of GENOSYL (nitric oxide) for prevention of BPD in preterm neonates ≤34 weeks gestational age is not recommended.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
GENOSYL Delivery System cassettes produce at least 216 liters of 800 ppm nitric oxide gas (at standard temperature and pressure, STP) (NDC: 72385-001-01).
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL
Rx Only
GENOSYL® (nitric oxide) for inhalation
(For use with GENOSYL® Delivery System only)
800 PPM
NDC: 72385-001-01
LOT Z-XXXX-YYYY
SN GXXXX
EXP YYYY-MM-DD
Store at 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C –30°C (59°F –86°F). [see USP Controlled Room Temperature]
Recommended Dosage: See prescribing information.
Manufactured by.
VERO BIOTECH, LLC
387 Technology Circle NW, Suite 125
Atlanta, GA 30313 USA

GENOSYL® (nitric oxide) for inhalation
NDC: 72385-001-01
LOT Z-XXXX-YYYY
SN GXXXX
EXP YYYY-MM-DD
VERO BIOTECH, LLC
387 Technology Circle NW, Suits 125
Atlanta, GA 30313 USA

-
INGREDIENTS AND APPEARANCE
GENOSYL
nitric oxide gasProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 72385-001 Route of Administration RESPIRATORY (INHALATION) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NITRIC OXIDE (UNII: 31C4KY9ESH) (NITRIC OXIDE - UNII:31C4KY9ESH) NITRIC OXIDE 0.98 mg in 1 L Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 72385-001-01 216 L in 1 CARTRIDGE; Type 0: Not a Combination Product 12/20/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA202860 12/20/2019 Labeler - VERO BIOTECH LLC (872672477) Registrant - VERO BIOTECH LLC (872672477) Establishment Name Address ID/FEI Business Operations VERO BIOTECH LLC 872672477 manufacture(72385-001)
Trademark Results [GENOSYL]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 GENOSYL 87772550 not registered Live/Pending |
VERO BIOTECH, LLC 2018-01-26 |
 GENOSYL 85981080 4496333 Live/Registered |
GeNO LLC 2011-12-02 |
 GENOSYL 85485604 4700107 Live/Registered |
VERO BIOTECH, LLC 2011-12-02 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.