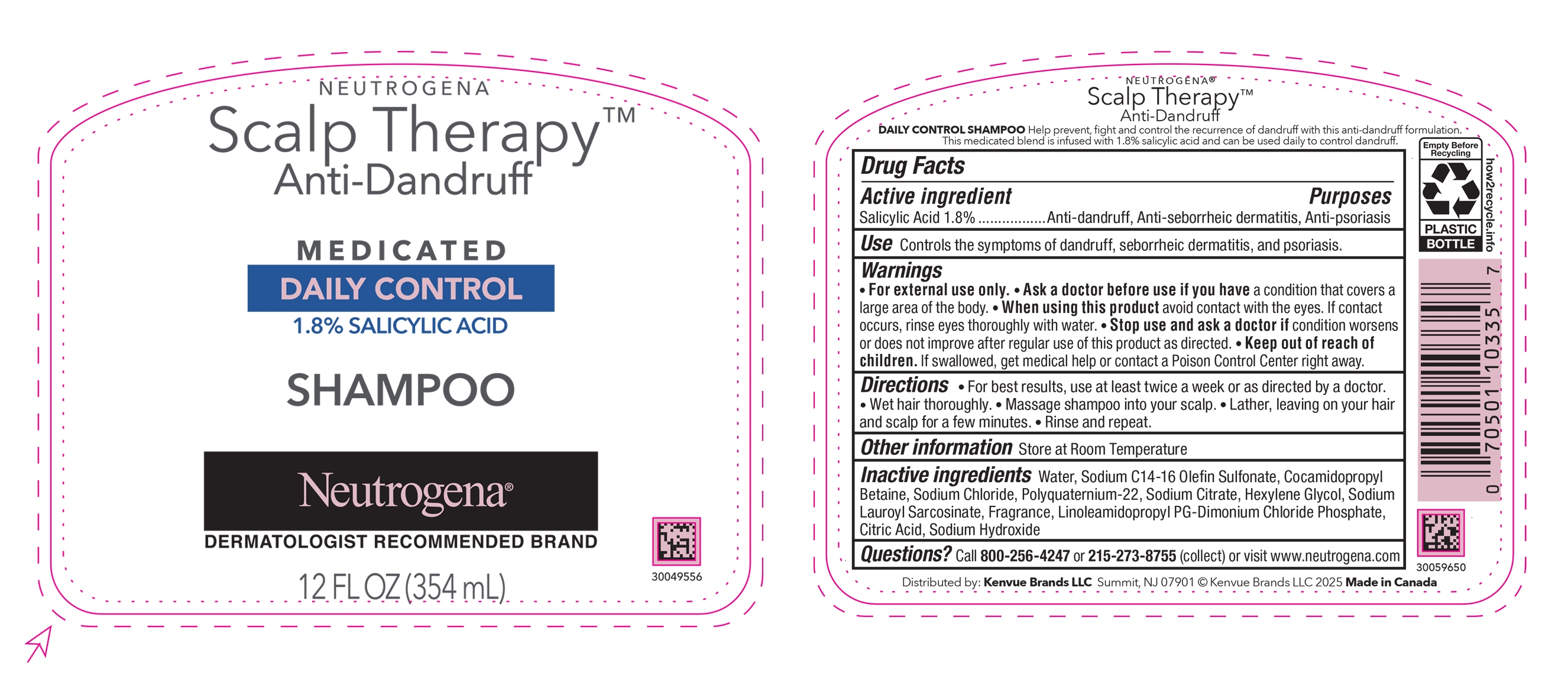
NEUTROGENA ® Scalp Therapy TM Anti-Dandruff Daily Control Shampoo
Neutrogena Scalp Therapy Anti-Dandruff Daily Control by
Drug Labeling and Warnings
Neutrogena Scalp Therapy Anti-Dandruff Daily Control by is a Otc medication manufactured, distributed, or labeled by Johnson & Johnson Consumer Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
NEUTROGENA SCALP THERAPY ANTI-DANDRUFF DAILY CONTROL- salicylic acid shampoo
Kenvue Brands LLC
----------
NEUTROGENA ® Scalp Therapy TM Anti-Dandruff Daily Control Shampoo
Warnings
- For external use only.
- When using this product avoid contact with the eyes. If contact occurs, rinse eyes thoroughly with water.
Directions
- For best results, use at least twice a week or as directed by a doctor.
- Wet hair thoroughly.
- Massage shampoo into your scalp.
- Lather, leaving on your hair and scalp for a few minutes.
- Rinse and repeat.
| NEUTROGENA SCALP THERAPY ANTI-DANDRUFF DAILY CONTROL
salicylic acid shampoo |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - Kenvue Brands LLC (118772437) |
Revised: 6/2025
Document Id: 36ba20cd-dc15-97aa-e063-6294a90a90cd
Set id: ca4944cf-7fdb-4720-e053-2995a90a35e9
Version: 5
Effective Time: 20250604
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.