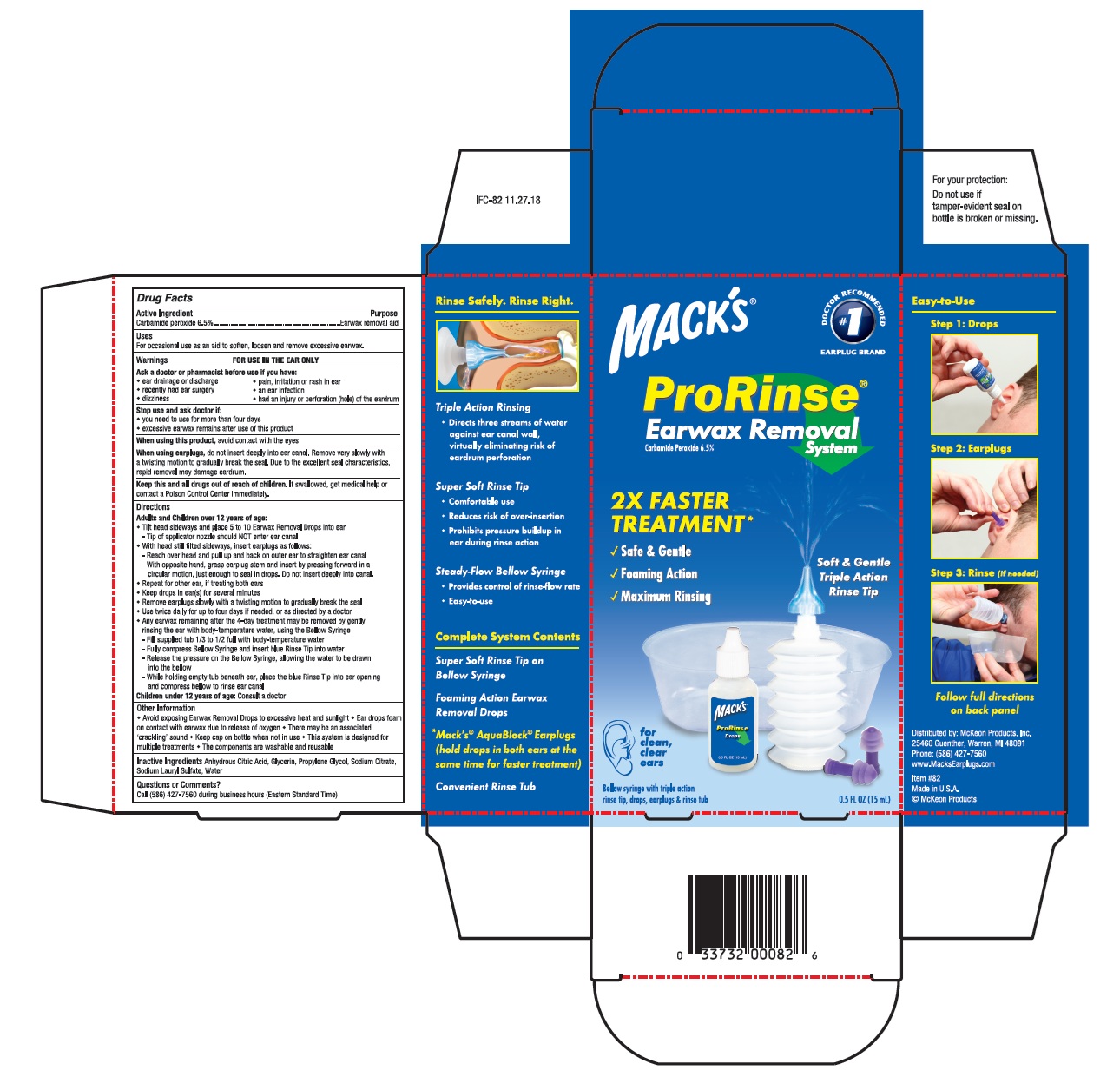
PRORINSE EARWAX REMOVAL SYSTEM- carbamide peroxide solution/ drops
ProRinse Earwax Removal System by
Drug Labeling and Warnings
ProRinse Earwax Removal System by is a Otc medication manufactured, distributed, or labeled by McKeon Products Inc, Bell Pharmaceuticals, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Drug Facts
- Active ingredient
- Uses
-
Warnings
For use in the ear only
Ask a doctor or pharmacist before use if you have:
- ear drainage or discharge
- recently had ear surgery
- dizziness
- pain, irritation, or rash in the ear
- an ear infection
- had an injury or perforation (hole) of the eardrum
Stop use and ask doctor if:
- you need to use for more than four days
- excessive earwax remains after use of this product
When using this product, avoid contact with the eyes
When using earplugs, do not insert deeply into ear canal. Remove very slowly with a twisting motion to gradually break the seal. Due to the excellent seal characteristics, rapid removal may damage eardrum
Keep this and all drugs out of reach of children. If swallowed, get medical help or contact a Poison Control Center immediately.
- KEEP OUT OF REACH OF CHILDREN
-
Directions
Adults and Children over 12 years of age:
- Tilt head sideways and place 5 to 10 Earwax Removal Drops into ear
Tip of applicator nozzle should NOT enter ear canal
- With head still sideways, insert earplugs as follows:
Reach over head and pull up and back on outer ear to straighten ear canal
With opposite hand, grasp earplug stem and insert by pressing forward in a circular motion, just enough to seal in drops. Do not insert deeply into canal.
- Repeat for other ear, if treating both ears
- Keep drops in ear(s) for several minutes
- Remove earplugs slowly with a twisting motion to gradually break the seal
- Use twice daily for up to four days if needed, or as directed by a doctor
- Any earwax remaining after the 4-day treatment may be removed by gently rinsing the ear with body-temperature water, using the Bellow Syringe
Fill supplied tub 1/3 to 1/2 full with body-temperature water
Fully compress Bellow Syringe and insert blue Rinse Tip into water
Release the pressure on the Bellow Syringe, allowing the water to be drawn into the bellow
While holding tub beneath ear, place the blue Rinse Tip into ear opening and compress bellow to rinse ear canal
Children under 12 years of age: Consult a doctor
- Other information
- Inactive ingredients
- Questions or comments?
- Product label
-
INGREDIENTS AND APPEARANCE
PRORINSE EARWAX REMOVAL SYSTEM
carbamide peroxide solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 66354-007 Route of Administration AURICULAR (OTIC) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARBAMIDE PEROXIDE (UNII: 31PZ2VAU81) (HYDROGEN PEROXIDE - UNII:BBX060AN9V) CARBAMIDE PEROXIDE 65 mg in 1 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM CITRATE (UNII: 1Q73Q2JULR) SODIUM LAURYL SULFATE (UNII: 368GB5141J) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 66354-007-82 1 in 1 BOX 02/05/2026 1 15 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M014 02/05/2026 Labeler - McKeon Products Inc (006522122) Establishment Name Address ID/FEI Business Operations Bell Pharmaceuticals, Inc. 140653770 manufacture(66354-007)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.