DAPZURA RT- daptomycin injection, powder, lyophilized, for solution
DAPZURA RT by
Drug Labeling and Warnings
DAPZURA RT by is a Prescription medication manufactured, distributed, or labeled by Baxter Healthcare Corporation, BAXTER PHARMACEUTICAL SOLUTIONS, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use DAPZURA RT safely and effectively. See full prescribing information for DAPZURA RT.
DAPZURA RT (daptomycin for injection), for Intravenous use
Initial U.S. Approval: 2003INDICATIONS AND USAGE
DAPZURA RT is a lipopeptide antibacterial indicated for the treatment of:
- Complicated skin and skin structure infections (cSSSI) in adult and pediatric patients (1 to 17 years of age) (1.1) and,
- Staphylococcus aureus bloodstream infections (bacteremia), in adult patients including those with right-sided infective endocarditis, (1.2)
- Staphylococcus aureus bloodstream infections (bacteremia) in pediatric patients (1 to 17 years of age).(1.3)
Limitations of Use:
- DAPZURA RT is not indicated for the treatment of pneumonia. (1.4)
- DAPZURA RT is not indicated for the treatment of left-sided infective endocarditis due to S. aureus. (1.4)
- DAPZURA RT is not recommended in pediatric patients younger than one year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs. (1.4)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of DAPZURA RT and other antibacterial drugs, DAPZURA RT should be used to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. (1.5)
DOSAGE AND ADMINISTRATION
Adult Patients
- Administer to adult patients intravenously in 0.9% sodium chloride, either by injection over a 2-minute period or by infusion over a 30-minute period. (2.1, 2.7)
- Recommended dosage regimen for adult patients (2.2, 2.4, 2.6):
Creatinine Clearance
(CLCR)Dosage Regimen cSSSI
For 7 to 14 daysS. aureus Bacteremia
For 2 to 6 weeks- * Administered following hemodialysis on hemodialysis days.
≥30 mL/min
4 mg/kg once every 24 hours
6 mg/kg once every 24 hours
<30 mL/min, including hemodialysis and CAPD
4 mg/kg once every 48 hours*
6 mg/kg once every 48 hours*
Pediatric Patients
- Unlike in adults, do NOT administer by injection over a two (2) minute period to pediatric patients. (2.1, 2.7)
- Administer to pediatric patients intravenously in 0.9% sodium chloride, by infusion over a 30- or 60-minute period, based on age. (2.1, 2.7)
- Recommended dosage regimen for pediatric patients (1 to 17 years of age) with cSSSI, based on age (2.3):
Age group Dosage* Duration of therapy - * Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established.
12 to 17 years
5 mg/kg once every 24 hours infused over 30 minutes
Up to 14 days
7 to 11 years
7 mg/kg once every 24 hours infused over 30 minutes
2 to 6 years
9 mg/kg once every 24 hours infused over 60 minutes
1 to less than 2 years
10 mg/kg once every 24 hours infused over 60 minutes
- Recommended dosage regimen for pediatric patients (1 to 17 years of age) with S. aureus bacteremia, based on age (2.5):
Age group Dosage* Duration of therapy - * Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established.
12 to 17 years
7 mg/kg once every 24 hours infused over 30 minutes
Up to 42 days
7 to 11 years
9 mg/kg once every 24 hours infused over 30 minutes
1 to 6 years
12 mg/kg once every 24 hours infused over 60 minutes
- There are other formulations of daptomycin that have differences concerning storage and reconstitution. Carefully follow the reconstitution and storage procedures described in this labeling. (2.7)
- Do not use in conjunction with ReadyMED® elastomeric infusion pumps in adult and pediatric patients. (2.9)
DOSAGE FORMS AND STRENGTHS
For Injection: 500 mg lyophilized powder for reconstitution in a single-dose vial (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Anaphylaxis/hypersensitivity reactions (including life-threatening): Discontinue DAPZURA RTand treat signs/symptoms. (5.1)
- Myopathy and rhabdomyolysis: Monitor CPK levels and follow muscle pain or weakness; if elevated CPK or myopathy occurs, consider discontinuation of DAPZURA RT. (5.2)
- Eosinophilic pneumonia: Discontinue DAPZURA RTand consider treatment with systemic steroids. (5.3)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue DAPZURA RTand institute appropriate treatment. (5.4)
- Tubulointerstitial Nephritis (TIN): Discontinue DAPZURA RTand institute appropriate treatment. (5.5)
- Peripheral neuropathy: Monitor for neuropathy and consider discontinuation. (5.6)
- Potential nervous system and/or muscular system effects in pediatric patients younger than 12 months: Avoid use of DAPZURA RT in this age group. (5.7)
- Clostridioides difficile–associated diarrhea: Evaluate patients if diarrhea occurs. (5.8)
- Persisting or relapsing S. aureus bacteremia/endocarditis: Perform susceptibility testing and rule out sequestered foci of infection. (5.9)
- Decreased efficacy was observed in adult patients with moderate baseline renal impairment. (5.10)
- Hereditary Fructose Intolerance (HFI): DAPZURA RT contains sorbitol. Risk of metabolic crisis with life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure. Obtain history of HFI symptoms in pediatric patients before DAPZURA RT administration (5.11, 8.4)
ADVERSE REACTIONS
- Adult cSSSI Patients: The most common adverse reactions that occurred in ≥2% of adult cSSSI patients receiving daptomycin for injection 4 mg/kg were diarrhea, headache, dizziness, rash, abnormal liver function tests, elevated creatine phosphokinase (CPK), urinary tract infections, hypotension, and dyspnea. (6.1)
- Pediatric cSSSI Patients: The most common adverse reactions that occurred in ≥2% of pediatric patients receiving daptomycin for injection were diarrhea, vomiting, abdominal pain, pruritus, pyrexia, elevated CPK, and headache. (6.1)
- Adult S. aureus bacteremia/endocarditis Patients: The most common adverse reactions that occurred in ≥5% of S. aureus bacteremia/endocarditis patients receiving daptomycin for injection 6 mg/kg were sepsis, bacteremia, abdominal pain, chest pain, edema, pharyngolaryngeal pain, pruritus, increased sweating, insomnia, elevated CPK, and hypertension. (6.1)
- Pediatric S. aureus bacteremia Patients: The most common adverse reactions that occurred in ≥5% of pediatric patients receiving daptomycin for injection were vomiting and elevated CPK. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Baxter Healthcare Corporation at 1-866-888-2472 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 1/2022
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Complicated Skin and Skin Structure Infections (cSSSI)
1.2 Staphylococcus aureus Bloodstream Infections (Bacteremia) in Adult Patients, Including Those with Right-Sided Infective Endocarditis, Caused by Methicillin-Susceptible and Methicillin-Resistant Isolates
1.3 Staphylococcus aureus Bloodstream Infections (Bacteremia) in Pediatric Patients (1 to 17 Years of Age)
1.4 Limitations of Use
1.5 Usage
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Duration Instructions
2.2 Dosage in Adults for cSSSI
2.3 Dosage in Pediatric Patients (1 to 17 Years of Age) for cSSSI
2.4 Dosage in Adult Patients with Staphylococcus aureus Bloodstream Infections (Bacteremia), Including Those with Right-Sided Infective Endocarditis, Caused by Methicillin-Susceptible and Methicillin-Resistant Isolates
2.5 Dosage in Pediatric Patients (1 to 17 Years of Age) with Staphylococcus aureus Bloodstream Infections (Bacteremia)
2.6 Dosage in Patients with Renal Impairment
2.7 Preparation and Administration of DAPZURA RT
2.8 Compatible Intravenous Solutions
2.9 Incompatibilities
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Anaphylaxis/Hypersensitivity Reactions
5.2 Myopathy and Rhabdomyolysis
5.3 Eosinophilic Pneumonia
5.4 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)
5.5 Tubulointerstitial Nephritis (TIN)
5.6 Peripheral Neuropathy
5.7 Potential Nervous System and/or Muscular System Effects in Pediatric Patients Younger than 12 Months
5.8 Clostridioides difficile-Associated Diarrhea
5.9 Persisting or Relapsing S. aureus Bacteremia/Endocarditis
5.10 Decreased Efficacy in Patients with Moderate Baseline Renal Impairment
5.11 Risk in Patients with Hereditary Fructose Intolerance (HFI)
5.12 Increased International Normalized Ratio (INR)/Prolonged Prothrombin Time
5.13 Development of Drug-Resistant Bacteria
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Post-Marketing Experience
7 DRUG INTERACTIONS
7.1 HMG-CoA Reductase Inhibitors
7.2 Drug-Laboratory Test Interactions
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Patients with Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
14.1 Complicated Skin and Skin Structure Infections
14.2 S. aureus Bacteremia/Endocarditis
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Complicated Skin and Skin Structure Infections (cSSSI)
DAPZURA RT is indicated for the treatment of adult and pediatric patients (1 to 17 years of age) with complicated skin and skin structure infections (cSSSI) caused by susceptible isolates of the following Gram-positive bacteria: Staphylococcus aureus (including methicillin-resistant isolates), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus dysgalactiae subsp. equisimilis, and Enterococcus faecalis (vancomycin-susceptible isolates only).
1.2 Staphylococcus aureus Bloodstream Infections (Bacteremia) in Adult Patients, Including Those with Right-Sided Infective Endocarditis, Caused by Methicillin-Susceptible and Methicillin-Resistant Isolates
DAPZURA RT is indicated for the treatment of adult patients with Staphylococcus aureus bloodstream infections (bacteremia), including adult patients with right-sided infective endocarditis, caused by methicillin-susceptible and methicillin-resistant isolates.
1.3 Staphylococcus aureus Bloodstream Infections (Bacteremia) in Pediatric Patients (1 to 17 Years of Age)
DAPZURA RT is indicated for the treatment of pediatric patients (1 to 17 years of age) with Staphylococcus aureus bloodstream infections (bacteremia).
1.4 Limitations of Use
DAPZURA RT is not indicated for the treatment of pneumonia.
DAPZURA RT is not indicated for the treatment of left-sided infective endocarditis due to S. aureus. The clinical trial of daptomycin for injection in adult patients with S. aureus bloodstream infections included limited data from patients with left-sided infective endocarditis; outcomes in these patients were poor [see Clinical Studies (14.2)]. Daptomycin for injection has not been studied in patients with prosthetic valve endocarditis.
DAPZURA RT is not recommended in pediatric patients younger than 1 year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs [see Warnings and Precautions (5.7) and Nonclinical Toxicology (13.2)].
1.5 Usage
Appropriate specimens for microbiological examination should be obtained in order to isolate and identify the causative pathogens and to determine their susceptibility to daptomycin.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of DAPZURA RT and other antibacterial drugs, DAPZURA RT should be used only to prevent infections that are proven or strongly suspected to be caused by susceptible bacteria.
When culture and susceptibility information is available, it should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy. Empiric therapy may be initiated while awaiting test results.
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Duration Instructions
Adults
Administer the appropriate volume of the reconstituted DAPZURA RT (concentration of 50 mg/mL) to adult patients intravenously either by injection over a two (2) minute period or by intravenous infusion over a thirty (30) minute period [see Dosage and Administration (2.2, 2.4, 2.7)].
Pediatric Patients (1 to 17 Years of Age)
Unlike in adults, do NOT administer DAPZURA RT by injection over a two (2) minute period to pediatric patients.
- Pediatric Patients 7 to 17 years of Age: Administer DAPZURA RT intravenously by infusion over a 30-minute period [see Dosage and Administration (2.3, 2.5, 2.7)].
- Pediatric Patients 1 to 6 years of Age: Administer DAPZURA RT intravenously by infusion over a 60-minute period [see Dosage and Administration (2.3, 2.5, 2.7)].
2.2 Dosage in Adults for cSSSI
Administer DAPZURA RT 4 mg/kg to adult patients intravenously once every 24 hours for 7 to 14 days.
2.3 Dosage in Pediatric Patients (1 to 17 Years of Age) for cSSSI
The recommended dosage regimens based on age for pediatric patients with cSSSI are shown in Table 1. Administer DAPZURA RT intravenously once every 24 hours for up to 14 days.
Table 1. Recommended Dosage of DAPZURA RT in Pediatric Patients (1 to 17 Years of Age) with cSSSI, Based on Age Age Range Dosage Regimen* Duration of therapy - * Recommended dosage regimen is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established.
12 to 17 years
5 mg/kg once every 24 hours infused over 30 minutes
Up to 14 days
7 to 11 years
7 mg/kg once every 24 hours infused over 30 minutes
2 to 6 years
9 mg/kg once every 24 hours infused over 60 minutes
1 to less than 2 years
10 mg/kg once every 24 hours infused over 60 minutes
2.4 Dosage in Adult Patients with Staphylococcus aureus Bloodstream Infections (Bacteremia), Including Those with Right-Sided Infective Endocarditis, Caused by Methicillin-Susceptible and Methicillin-Resistant Isolates
Administer DAPZURA RT 6 mg/kg to adult patients intravenously once every 24 hours for 2 to 6 weeks. There are limited safety data for the use of daptomycin for injection for more than 28 days of therapy. In the Phase 3 trial, there were a total of 14 adult patients who were treated with daptomycin for injection for more than 28 days.
2.5 Dosage in Pediatric Patients (1 to 17 Years of Age) with Staphylococcus aureus Bloodstream Infections (Bacteremia)
The recommended dosage regimens based on age for pediatric patients with S. aureus bloodstream infections (bacteremia) are shown in Table 2. Administer DAPZURA RT intravenously in 0.9% sodium chloride injection once every 24 hours for up to 42 days.
Table 2. Recommended Dosage of DAPZURA RT in Pediatric Patients (1 to 17 Years of Age) with S. aureus Bacteremia, Based on Age Age group Dosage* Duration of therapy - * Recommended dosage is for pediatric patients (1 to 17 years of age) with normal renal function. Dosage adjustment for pediatric patients with renal impairment has not been established.
12 to 17 years
7 mg/kg once every 24 hours infused over 30 minutes
Up to 42 days
7 to 11 years
9 mg/kg once every 24 hours infused over 30 minutes
1 to 6 years
12 mg/kg once every 24 hours infused over 60 minutes
2.6 Dosage in Patients with Renal Impairment
Adult Patients:
No dosage adjustment is required in adult patients with creatinine clearance (CLCR) greater than or equal to 30 mL/min. The recommended dosage regimen for DAPZURA RT in adult patients with CLCR less than 30 mL/min, including adult patients on hemodialysis or continuous ambulatory peritoneal dialysis (CAPD), is 4 mg/kg (cSSSI) or 6 mg/kg (S. aureus bloodstream infections) once every 48 hours (Table 3). When possible, DAPZURA RT should be administered following the completion of hemodialysis on hemodialysis days [see Warnings and Precautions (5.2, 5.10), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
Table 3. Recommended Dosage of DAPZURA RT in Adult Patients Creatinine Clearance
(CLCR)Dosage Regimen in Adults cSSSI S. aureus Bloodstream Infections - * When possible, administer DAPZURA RT following the completion of hemodialysis on hemodialysis days.
Greater than or equal to 30 mL/min
4 mg/kg once every 24 hours
6 mg/kg once every 24 hours
Less than 30 mL/min, including hemodialysis and CAPD
4 mg/kg once every 48 hours*
6 mg/kg once every 48 hours*
Pediatric Patients:
The dosage regimen for DAPZURA RT in pediatric patients with renal impairment has not been established.
2.7 Preparation and Administration of DAPZURA RT
There are other formulations of daptomycin that have differences concerning reconstitution and storage. Carefully follow the reconstitution and storage procedures described in this labeling.
Reconstitution of DAPZURA RT Vial
DAPZURA RT must be reconstituted within the vial only with either Sterile Water for Injection or Bacteriostatic Water for Injection.
Do NOT use saline based diluents for the reconstitution in the vial because this will result in a hyperosmotic solution that may result in infusion site reactions if the reconstituted product is administered as an intravenous injection over a period of 2 minutes.
DAPZURA RT is supplied in single-dose vials, each containing 500 mg daptomycin as a sterile, lyophilized powder. The contents of a DAPZURA RT vial should be reconstituted, using aseptic technique, to 50 mg/mL as follows:
- 1. Remove the polypropylene flip-off cap from the DAPZURA RT vial to expose the central portion of the rubber stopper.
- 2. Wipe the top of the rubber stopper with an alcohol swab or other antiseptic solution and allow to dry. After cleaning, do not touch the rubber stopper or allow it to touch any other surface.
- 3. Transfer 10 mL of Sterile Water for Injection or Bacteriostatic Water for Injection through the center of the rubber stopper into the DAPZURA RT vial. Use a beveled sterile transfer needle that is 21 gauge or smaller in diameter, pointing the transfer needle toward the wall of the vial.
- 4. Rotate or swirl the vial contents for a few minutes, as needed, to obtain a completely reconstituted solution.
Administration Instructions
Parenteral drug products should be inspected visually for particulate matter prior to administration.
Slowly remove reconstituted liquid (50 mg daptomycin/mL) from the vial using a beveled sterile needle that is 21 gauge or smaller in diameter. Administer as an intravenous injection or infusion as described below:
Adults
Intravenous Injection over a period of 2 minutes
- For intravenous (IV) injection over a period of 2 minutes in adult patients only: Administer the appropriate volume of the reconstituted DAPZURA RT (concentration of 50 mg/mL).
Intravenous Infusion over a period of 30 minutes
- For IV infusion over a period of 30 minutes in adult patients: The appropriate volume of the reconstituted DAPZURA RT (concentration of 50 mg/mL) should be further diluted, using aseptic technique, into a 50 mL IV infusion bag containing 0.9% sodium chloride injection.
Pediatric Patients (1 to 17 Years of Age)
Intravenous Infusion over a period of 30 or 60 minutes
- Unlike in Adults, do NOT administer DAPZURA RT by injection over a two (2) minute period to pediatric patients [see Dosage and Administration (2.1)].
- For Intravenous infusion over a period of 60 minutes in pediatric patients 1 to 6 years of age: The appropriate volume of the reconstituted DAPZURA RT (concentration of 50 mg/mL) should be further diluted, using aseptic technique, into an intravenous infusion bag containing 25 mL of 0.9% sodium chloride injection. The infusion rate should be maintained at 0.42 mL/minute over the 60-minute period.
- For Intravenous infusion over a period of 30 minutes in pediatric patients 7 to 17 years of age: The appropriate volume of the reconstituted DAPZURA RT (concentration of 50 mg/mL) should be further diluted, using aseptic technique, into a 50 mL IV infusion bag containing 0.9% sodium chloride injection. The infusion rate should be maintained at 1.67 mL/minute over the 30-minute period.
No preservative or bacteriostatic agent is present in this product. Aseptic technique must be used in the preparation of final IV solution. Table 4 below provides in-use storage conditions for reconstituted DAPZURA RT in acceptable intravenous diluents in the syringe, vial and intravenous bag (for reconstitution and dilution). Do not exceed the listed shelf-life of reconstituted and diluted solutions of DAPZURA RT. Discard unused portions of DAPZURA RT.
Table 4. In-Use Storage Conditions for DAPZURA RT Once Reconstituted in Acceptable Intravenous Diluents Container Diluent In-Use Shelf-Life Room Temperature
(20°C–25°C, 68°F–77°F)Refrigerated
(2°C–8°C, 36°F–46°F)- * Polypropylene syringe with elastomeric plunger stopper.
Vial
Sterile Water for Injection
18 Hours
3 Days
Bacteriostatic Water for Injection
2 Days
5 Days
Syringe*
Sterile Water for Injection
18 Hours
3 Days
Bacteriostatic Water for Injection
2 Days
5 Days
Intravenous Bag
Vial reconstituted with Sterile Water for Injection and immediately diluted with 0.9% sodium chloride.
1 Day
3 Days
Vial reconstituted with Bacteriostatic Water for Injection and immediately diluted with 0.9% sodium chloride injection.
2 Days
5 Days
2.8 Compatible Intravenous Solutions
Reconstituted DAPZURA RT is compatible with Sterile Water for Injection, Bacteriostatic Water for Injection, and 0.9% sodium chloride injection. [See Dosage and Administration (2.7)]
2.9 Incompatibilities
Daptomycin for injection is not compatible with dextrose-containing diluents.
DAPZURA RT should not be used in conjunction with ReadyMED® elastomeric infusion
pumps. Stability studies of daptomycin for injection solutions stored in ReadyMED®
elastomeric infusion pumps identified an impurity (2-mercaptobenzothiazole) leaching
from this pump system into the daptomycin for injection solution.
Because only limited data are available on the compatibility of daptomycin for injection with other IV substances, additives and other medications should not be added to DAPZURA RT single-dose vials or infusion bags, or infused simultaneously with DAPZURA RT through the same IV line. If the same IV line is used for sequential infusion of different drugs, the line should be flushed with a compatible intravenous solution before and after infusion with DAPZURA RT.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
DAPZURA RT is contraindicated in:
Patients with known hypersensitivity to daptomycin [see Warnings and Precautions (5.1)].
- Patients with known or suspected Hereditary Fructose Intolerance (HFI) [see Warnings and Precautions (5.11)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Anaphylaxis/Hypersensitivity Reactions
Anaphylaxis/hypersensitivity reactions have been reported with the use of antibacterial agents, including daptomycin for injection, and may be life-threatening. If an allergic reaction to DAPZURA RT occurs, discontinue the drug and institute appropriate therapy [see Adverse Reactions (6.2)].
5.2 Myopathy and Rhabdomyolysis
Myopathy, defined as muscle aching or muscle weakness in conjunction with increases in creatine phosphokinase (CPK) values to greater than 10 times the upper limit of normal (ULN), has been reported with the use of daptomycin for injection. Rhabdomyolysis, with or without acute renal failure, has been reported [see Adverse Reactions (6.2)].
Patients receiving DAPZURA RT should be monitored for the development of muscle pain or weakness, particularly of the distal extremities. In patients who receive DAPZURA RT, CPK levels should be monitored weekly, and more frequently in patients who received recent prior or concomitant therapy with an HMG-CoA reductase inhibitor or in whom elevations in CPK occur during treatment with DAPZURA RT.
In adult patients with renal impairment, both renal function and CPK should be monitored more frequently than once weekly [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
In Phase 1 studies and Phase 2 clinical trials in adults, CPK elevations appeared to be more frequent when daptomycin for injection was dosed more than once daily. Therefore, DAPZURA RT should not be dosed more frequently than once a day.
DAPZURA RT should be discontinued in patients with unexplained signs and symptoms of myopathy in conjunction with CPK elevations to levels >1,000 U/L (~5× ULN), and in patients without reported symptoms who have marked elevations in CPK, with levels >2,000 U/L (≥10× ULN). In addition, consideration should be given to suspending agents associated with rhabdomyolysis, such as HMG-CoA reductase inhibitors, temporarily in patients receiving DAPZURA RT [see Drug Interactions (7.1)].
5.3 Eosinophilic Pneumonia
Eosinophilic pneumonia has been reported in patients receiving daptomycin for injection [see Adverse Reactions (6.2)]. In reported cases associated with daptomycin for injection, patients developed fever, dyspnea with hypoxic respiratory insufficiency, and diffuse pulmonary infiltrates or organizing pneumonia. In general, patients developed eosinophilic pneumonia 2 to 4 weeks after starting daptomycin for injection and improved when daptomycin for injection was discontinued and steroid therapy was initiated. Recurrence of eosinophilic pneumonia upon re-exposure has been reported. Patients who develop these signs and symptoms while receiving DAPZURA RT should undergo prompt medical evaluation, and DAPZURA RT should be discontinued immediately. Treatment with systemic steroids is recommended.
5.4 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)
DRESS has been reported in post-marketing experience with daptomycin for injection [see Adverse Reactions (6.2)]. Patients who develop skin rash, fever, peripheral eosinophilia, and systemic organ (for example, hepatic, renal, pulmonary) impairment while receiving DAPZURA RT should undergo medical evaluation. If DRESS is suspected, discontinue DAPZURA RT promptly and institute appropriate treatment.
5.5 Tubulointerstitial Nephritis (TIN)
TIN has been reported in post-marketing experience with daptomycin for injection [see Adverse Reactions (6.2)]. Patients who develop new or worsening renal impairment while receiving DAPZURA RT should undergo medical evaluation. If TIN is suspected, discontinue DAPZURA RT promptly and institute appropriate treatment.
5.6 Peripheral Neuropathy
Cases of peripheral neuropathy have been reported during the daptomycin for injection postmarketing experience [see Adverse Reactions (6.2)]. Therefore, physicians should be alert to signs and symptoms of peripheral neuropathy in patients receiving DAPZURA RT. Monitor for neuropathy and consider discontinuation.
5.7 Potential Nervous System and/or Muscular System Effects in Pediatric Patients Younger than 12 Months
Avoid use of DAPZURA RTin pediatric patients younger than 12 months due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs with intravenous daptomycin [see Nonclinical Toxicology (13.2)].
5.8 Clostridioides difficile-Associated Diarrhea
Clostridioides difficile–associated diarrhea (CDAD) has been reported with the use of nearly all systemic antibacterial agents, including daptomycin for injection, and may range in severity from mild diarrhea to fatal colitis [see Adverse Reactions (6.2)]. Treatment with antibacterial agents alters the normal flora of the colon, leading to overgrowth of C. difficile.
C. difficile produces toxins A and B, which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, since these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur more than 2 months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
5.9 Persisting or Relapsing S. aureus Bacteremia/Endocarditis
Patients with persisting or relapsing S. aureus bacteremia/endocarditis or poor clinical response should have repeat blood cultures. If a blood culture is positive for S. aureus, minimum inhibitory concentration (MIC) susceptibility testing of the isolate should be performed using a standardized procedure, and diagnostic evaluation of the patient should be performed to rule out sequestered foci of infection. Appropriate surgical intervention (e.g., debridement, removal of prosthetic devices, valve replacement surgery) and/or consideration of a change in antibacterial regimen may be required.
Failure of treatment due to persisting or relapsing S. aureus bacteremia/endocarditis may be due to reduced daptomycin susceptibility (as evidenced by increasing MIC of the S. aureus isolate) [see Clinical Studies (14.2)].
5.10 Decreased Efficacy in Patients with Moderate Baseline Renal Impairment
Limited data are available from the two Phase 3 complicated skin and skin structure infection (cSSSI) trials regarding clinical efficacy of daptomycin for injection treatment in adult patients with creatinine clearance (CLCR) <50 mL/min; only 31/534 (6%) patients treated with daptomycin for injection in the intent-to-treat (ITT) population had a baseline CLCR <50 mL/min. Table 5 shows the number of adult patients by renal function and treatment group who were clinical successes in the Phase 3 cSSSI trials.
Table 5. Clinical Success Rates by Renal Function and Treatment Group in Phase 3 cSSSI Trials in Adult Patients (Population: ITT) CLCR
Success Rate n/N (%)
Daptomycin for Injection 4 mg/kg
every 24h
Comparator
50- 70 mL/min
25/38 (66%)
30/48 (63%)
30-<50 mL/min
7/15 (47%)
20/35 (57%)
In a subgroup analysis of the ITT population in the Phase 3 S. aureus bacteremia/endocarditis trial, clinical success rates, as determined by a treatment-blinded Adjudication Committee [see Clinical Studies (14.2)], in the daptomycin for injection-treated adult patients were lower in patients with baseline CLCR <50 mL/min (see Table 6). A decrease of the magnitude shown in Table 6 was not observed in comparator-treated patients.
Table 6. Adjudication Committee Clinical Success Rates at Test of Cure by Baseline Creatinine Clearance and Treatment Subgroup in the S. aureus Bacteremia/Endocarditis Trial in Adult Patients (Population: ITT) Baseline CLCR
Success Rate n/N (%)
Daptomycin for Injection 6 mg/kg every 24h
Comparator
Bacteremia
- Right-Sided
- Infective Endocarditis
Bacteremia
- Right-Sided
- Infective Endocarditis
>80 mL/min
30/50 (60%)
7/14 (50%)
19/42 (45%)
5/11 (46%)
50–80 mL/min
12/26 (46%)
1/4 (25%)
13/31 (42%)
1/2 (50%)
30–<50 mL/min
2/14 (14%)
0/1 (0%)
7/17 (41%)
1/1 (100%)
Consider these data when selecting antibacterial therapy for use in adult patients with baseline moderate to severe renal impairment.
5.11 Risk in Patients with Hereditary Fructose Intolerance (HFI)
DAPZURA RT contains sorbitol, an inactive ingredient, and may precipitate a metabolic crisis that may include, but is not limited to life-threatening hypoglycemia, hypophosphatemia, lactic acidosis, and hepatic failure in patients with HFI. The minimum amount of sorbitol at which serious adverse reactions may occur in these patients is not known.
Obtain a careful history of HFI symptoms (nausea, vomiting, abdominal pain) with sorbitol/fructose/sucrose exposure prior to DAPZURA RT administration because a diagnosis of HFI may not yet be established in pediatric patients [see Contraindications (4) and Use in Specific Populations (8.4)].
5.12 Increased International Normalized Ratio (INR)/Prolonged Prothrombin Time
Clinically relevant plasma concentrations of daptomycin have been observed to cause a significant concentration-dependent false prolongation of prothrombin time (PT) and elevation of International Normalized Ratio (INR) when certain recombinant thromboplastin reagents are utilized for the assay [see Drug Interactions (7.2)].
-
6 ADVERSE REACTIONS
The following adverse reactions are described, or described in greater detail, in other sections:
- Anaphylaxis/Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.2)]
- Eosinophilic Pneumonia [see Warnings and Precautions (5.3)]
- Drug Reaction with Eosinophilia and Systemic Symptoms [see Warnings and Precautions (5.4)]
- Tubulointerstitial Nephritis [see Warnings and Precautions (5.5)]
- Peripheral Neuropathy [see Warnings and Precautions (5.6)]
- Increased International Normalized Ratio (INR)/Prolonged Prothrombin Time [see Warnings and Precautions (5.12) and Drug Interactions (7.2)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Clinical Trial Experience in Adult Patients
Clinical trials enrolled 1,864 adult patients treated with daptomycin for injection and 1,416 treated with comparator.
Complicated Skin and Skin Structure Infection Trials in Adults
In Phase 3 complicated skin and skin structure infection (cSSSI) trials in adult patients, daptomycin for injection was discontinued in 15/534 (2.8%) patients due to an adverse reaction, while comparator was discontinued in 17/558 (3.0%) patients.
The rates of the most common adverse reactions, organized by body system, observed in adult patients with cSSSI (receiving 4 mg/kg daptomycin for injection) are displayed in Table 7.
Table 7. Incidence of Adverse Reactions that Occurred in ≥2% of Adult Patients in the Daptomycin for Injection Treatment Group and ≥ the Comparator Treatment Group in Phase 3 cSSSI Trials Adverse Reaction Adult Patients (%) - * Comparator: vancomycin (1 g IV every 12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 4 to 12 g/day IV in divided doses).
Daptomycin for Injection 4
mg/kg (N=534)
Comparator* (N=558)
Gastrointestinal disorders
Diarrhea
5.2
4.3
Nervous system disorders
Headache
5.4
5.4
Dizziness
2.2
2.0
Skin/subcutaneous disorders
Rash
4.3
3.8
Diagnostic investigations
Abnormal liver function tests
3.0
1.6
Elevated CPK
2.8
1.8
Infections
Urinary tract infections
2.4
0.5
Vascular disorders
Hypotension
2.4
1.4
Respiratory disorders
Dyspnea
2.1
1.6
Drug-related adverse reactions (possibly or probably drug-related) that occurred in <1% of adult patients receiving daptomycin for injection in the cSSSI trials are as follows:
Body as a Whole: fatigue, weakness, rigors, flushing, hypersensitivity
Blood/Lymphatic System: leukocytosis, thrombocytopenia, thrombocytosis, eosinophilia, increased International Normalized Ratio (INR)
Cardiovascular System: supraventricular arrhythmia
Dermatologic System: eczema
Digestive System: abdominal distension, stomatitis, jaundice, increased serum lactate dehydrogenase
Metabolic/Nutritional System: hypomagnesemia, increased serum bicarbonate, electrolyte disturbance
Musculoskeletal System: myalgia, muscle cramps, muscle weakness, arthralgia
Nervous System: vertigo, mental status change, paresthesia
Special Senses: taste disturbance, eye irritation
- S. aureus Bacteremia/Endocarditis Trial in Adults
In the S. aureus bacteremia/endocarditis trial involving adult patients, daptomycin for injection was discontinued in 20/120 (16.7%) patients due to an adverse reaction, while comparator was discontinued in 21/116 (18.1%) patients.
Serious Gram-negative infections (including bloodstream infections) were reported in 10/120 (8.3%) daptomycin for injection-treated patients and 0/115 comparator-treated patients. Comparator-treated patients received dual therapy that included initial gentamicin for 4 days. Infections were reported during treatment and during early and late follow-up. Gram-negative infections included cholangitis, alcoholic pancreatitis, sternal osteomyelitis/mediastinitis, bowel infarction, recurrent Crohn’s disease, recurrent line sepsis, and recurrent urosepsis caused by a number of different Gram-negative bacteria.
The rates of the most common adverse reactions, organized by System Organ Class (SOC), observed in adult patients with S. aureus bacteremia/endocarditis (receiving 6 mg/kg daptomycin for injection) are displayed in Table 8.
Table 8. Incidence of Adverse Reactions that Occurred in ≥5% of Adult Patients in the Daptomycin for Injection Treatment Group and ≥ the Comparator Treatment Group in the S. aureus Bacteremia/Endocarditis Trial Adverse Reaction* Adult Patients
n (%)- * NOS, not otherwise specified.
- † Comparator: vancomycin (1 g IV every 12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 2 g IV every 4h), each with initial low-dose gentamicin.
Daptomycin for Injection 6 mg/kg (N=120)
Comparator† (N=116)
Infections and infestations
Sepsis NOS
6 (5%)
3 (3%)
Bacteremia
6 (5%)
0 (0%)
Gastrointestinal disorders
Abdominal pain NOS
7 (6%)
4 (3%)
General disorders and administration site conditions
Chest pain
8 (7%)
7 (6%)
Edema NOS
8 (7%)
5 (4%)
Respiratory, thoracic and mediastinal disorders
Pharyngolaryngeal pain
10 (8%)
2 (2%)
Skin and subcutaneous tissue disorders
Pruritus
7 (6%)
6 (5%)
Sweating increased
6 (5%)
0 (0%)
Psychiatric disorders
Insomnia
11 (9%)
8 (7%)
Investigations
Blood creatine phosphokinase increased
8 (7%)
1 (1%)
Vascular disorders
Hypertension NOS
7 (6%)
3 (3%)
The following reactions, not included above, were reported as possibly or probably drug-related in the daptomycin for injection-treated group:
Blood and Lymphatic System Disorders: eosinophilia, lymphadenopathy, thrombocythemia, thrombocytopenia
Cardiac Disorders: atrial fibrillation, atrial flutter, cardiac arrest
Ear and Labyrinth Disorders: tinnitus
Eye Disorders: vision blurred
Gastrointestinal Disorders: dry mouth, epigastric discomfort, gingival pain, hypoesthesia oral
Infections and Infestations: candidal infection NOS, vaginal candidiasis, fungemia, oral candidiasis, urinary tract infection fungal
Investigations: blood phosphorous increased, blood alkaline phosphatase increased, INR increased, liver function test abnormal, alanine aminotransferase increased, aspartate aminotransferase increased, prothrombin time prolonged
Metabolism and Nutrition Disorders: appetite decreased NOS
Musculoskeletal and Connective Tissue Disorders: myalgia
Nervous System Disorders: dyskinesia, paresthesia
Psychiatric Disorders: hallucination NOS
Renal and Urinary Disorders: proteinuria, renal impairment NOS
Skin and Subcutaneous Tissue Disorders: pruritus generalized, rash vesicular
Other Trials in Adults
In Phase 3 trials of community-acquired pneumonia (CAP) in adult patients, the death rate and rates of serious cardiorespiratory adverse events were higher in daptomycin for injection-treated patients than in comparator-treated patients. These differences were due to lack of therapeutic effectiveness of daptomycin for injection in the treatment of CAP in patients experiencing these adverse events [see Indications and Usage (1.4)].
Laboratory Changes in Adults
Complicated Skin and Skin Structure Infection Trials in Adults
In Phase 3 cSSSI trials of adult patients receiving daptomycin for injection at a dose of 4 mg/kg, elevations in CPK were reported as clinical adverse events in 15/534 (2.8%) daptomycin for injection-treated patients, compared with 10/558 (1.8%) comparator-treated patients. Of the 534 patients treated with daptomycin for injection, 1 (0.2%) had symptoms of muscle pain or weakness associated with CPK elevations to greater than 4 times the upper limit of normal (ULN). The symptoms resolved within 3 days and CPK returned to normal within 7 to 10 days after treatment was discontinued [see Warnings and Precautions (5.2)]. Table 9 summarizes the CPK shifts from Baseline through End of Therapy in the cSSSI adult trials.
Table 9. Incidence of CPK Elevations from Baseline during Therapy in Either the Daptomycin for Injection Treatment Group or the Comparator Treatment Group in Phase 3 cSSSI Adult Trials - * Comparator: vancomycin (1 g IV every 12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 4 to 12 g/day IV in divided doses).
- † ULN (Upper Limit of Normal) is defined as 200 U/L.
All Adult Patients
Adult Patients with Normal CPK at Baseline
Change in CPK
Daptomycin for Injection
4 mg/kg(N=430)
Comparator*
(N=459)
Daptomycin for
Injection
4 mg/kg(N=374)
Comparator*
(N=392)%
n
%
n
%
n
%
n
No Increase
90.7
390
91.1
418
91.2
341
91.1
357
Maximum Value>1× ULN†
9.3
40
8.9
41
8.8
33
8.9
35
>2× ULN
4.9
21
4.8
22
3.7
14
3.1
12
>4× ULN
1.4
6
1.5
7
1.1
4
1.0
4
>5× ULN
1.4
6
0.4
2
1.1
4
0.0
0
>10× ULN
0.5
2
0.2
1
0.2
1
0.0
0
Note: Elevations in CPK observed in adult patients treated with daptomycin for injection or comparator were not clinically or statistically significantly different.
S. aureus Bacteremia/Endocarditis Trial in Adults
In the S. aureus bacteremia/endocarditis trial in adult patients, at a dose of 6 mg/kg, 11/120 (9.2%) daptomycin for injection-treated patients, including two patients with baseline CPK levels >500 U/L, had CPK elevations to levels >500 U/L, compared with 1/116 (0.9%) comparator-treated patients. Of the 11 daptomycin for injection-treated patients, 4 had prior or concomitant treatment with an HMG-CoA reductase inhibitor. Three of these 11 daptomycin for injection-treated patients discontinued therapy due to CPK elevation, while the one comparator-treated patient did not discontinue therapy [see Warnings and Precautions (5.2)].
Clinical Trial Experience in Pediatric Patients
Complicated Skin and Skin Structure Infection Trial in Pediatric Patients
The safety of daptomycin for injection was evaluated in one clinical trial (in cSSSI), which included 256 pediatric patients (1 to 17 years of age) treated with intravenous daptomycin for injection and 133 patients treated with comparator agents. Patients were given age-dependent doses once daily for a treatment period of up to 14 days (median treatment period was 3 days). The doses given by age group were as follows: 10mg/kg for 1 to < 2 years, 9 mg/kg for 2 to 6 years, 7mg/kg for 7 to 11 years and 5 mg/kg for 12 to 17 years of age [see Clinical Studies (14)]. Patients treated with daptomycin for injection were (51%) male, (49%) female and (46%) Caucasian and (32%) Asian.
Adverse Reactions Leading to Discontinuation
In the cSSSI study, daptomycin for injection was discontinued in 7/256 (2.7%) patients due to an adverse reaction, while comparator was discontinued in 7/133 (5.3%) patients.
Most Common Adverse Reactions
The rates of the most common adverse reactions, organized by body system, observed in these pediatric patients with cSSSI are displayed in Table 10.
Table 10. Adverse Reactions that Occurred in ≥2% of Pediatric Patients in the Daptomycin for Injection Treatment-Arm and Greater Than or Equal to the Comparator Treatment-Arm in the cSSSI Pediatric Trial Adverse Reaction Daptomycin for Injection (N = 256) Comparator*
(N = 133)- * Comparators included intravenous therapy with either vancomycin, clindamycin, or an anti-staphylococcal semi-synthetic penicillin (nafcillin, oxacillin or cloxacillin)
n (%)
n (%)
Gastrointestinal disorders
Diarrhea
18 (7.0)
7 (5.3)
Vomiting
7 (2.7)
1 (0.8)
Abdominal Pain
5 (2.0)
0
Skin and subcutaneous tissue disorders
Pruritus
8 (3.1)
2 (1.5)
General disorders and administration site conditions
Pyrexia
10 (3.9)
4 (3.0)
Investigations
Blood CPK increased
14 (5.5)
7 (5.3)
Nervous system disorders
Headache
7 (2.7)
3 (2.3)
The safety profile in the clinical trial of cSSSI pediatric patients was similar to that observed in the cSSSI adult patients.
S. aureus Bacteremia Trial in Pediatric Patients
The safety of daptomycin for injection was evaluated in one clinical trial (in S. aureus bacteremia), which treated 55 pediatric patients with intravenous daptomycin for injection and 26 patients with comparator agents. Patients were given age-dependent doses once daily for a treatment period of up to 42 days (mean duration of IV treatment was 12 days). The doses by age group were as follows: 12 mg/kg for 1 to <6 years, 9 mg/kg for 7 to 11 years and 7 mg/kg for 12 to 17 years of age [see Clinical Studies (14)]. Patients treated with daptomycin for injection were (69%) male and (31%) female. No patients 1 to <2 years of age were enrolled.
Adverse Reactions Leading to Discontinuation
In the bacteremia study, daptomycin for injection was discontinued in 3/55 (5.5%) patients due to an adverse reaction, while comparator was discontinued in 2/26 (7.7%) patients.
Most Common Adverse Reactions
The rates of the most common adverse reactions, organized by body system, observed in these pediatric patients with bacteremia are displayed in Table 11.
Table 11. Incidence of Adverse Reactions that Occurred in ≥5% of Pediatric Patients in the Daptomycin for Injection Treatment-Arm and Greater Than or Equal to the Comparator Treatment-Arm in the Pediatric Bacteremia Trial Adverse Reaction Daptomycin for Injection (N = 55) Comparator (N = 26)* - * Comparators included intravenous therapy with either vancomycin, cefazolin, or an anti-staphylococcal semi-synthetic penicillin (nafcillin, oxacillin or cloxacillin)
n (%)
n (%)
Gastrointestinal disorders
Vomiting
6 (10.9)
2 (7.7)
Investigations
Blood CPK increased
4 (7.3)
0
6.2 Post-Marketing Experience
The following adverse reactions have been identified during post-approval use of daptomycin for injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: anemia, thrombocytopenia
General and administration site conditions: pyrexia
Immune System Disorders: anaphylaxis; hypersensitivity reactions, including angioedema, pruritus, hives, shortness of breath, difficulty swallowing, truncal erythema, and pulmonary eosinophilia [see Contraindications (4) and Warnings and Precautions (5.1)]
Infections and Infestations: Clostridioides difficile–associated diarrhea [see Warnings and Precautions (5.8)]
Laboratory Investigations: platelet count decreased
Musculoskeletal Disorders: myoglobin increased; rhabdomyolysis (some reports involved patients treated concurrently with daptomycin for injection and HMG-CoA reductase inhibitors) [see Warnings and Precautions (5.2), Drug Interactions (7.1), and Clinical Pharmacology (12.3)]
Respiratory, Thoracic, and Mediastinal Disorders: cough, eosinophilic pneumonia, organizing pneumonia [see Warnings and Precautions (5.3)]
Nervous System Disorders: peripheral neuropathy [see Warnings and Precautions (5.6)]
Skin and Subcutaneous Tissue Disorders: serious skin reactions, including drug reaction with eosinophilia and systemic symptoms (DRESS), vesiculobullous rash (with or without mucous membrane involvement, including Stevens-Johnson syndrome [SJS] and toxic epidermal necrolysis [TEN]), acute generalized exanthematous pustulosis [see Warnings and Precautions (5.4)]
Gastrointestinal Disorders: nausea, vomiting
Renal and urinary disorders: acute kidney injury, renal insufficiency, renal failure, and tubulointerstitial nephritis (TIN) [see Warnings and Precautions (5.5)]
Special Senses: visual disturbances
-
7 DRUG INTERACTIONS
7.1 HMG-CoA Reductase Inhibitors
In healthy adult subjects, concomitant administration of daptomycin for injection and simvastatin had no effect on plasma trough concentrations of simvastatin, and there were no reports of skeletal myopathy [see Clinical Pharmacology (12.3)].
However, inhibitors of HMG-CoA reductase may cause myopathy, which is manifested as muscle pain or weakness associated with elevated levels of creatine phosphokinase (CPK). In the adult Phase 3 S. aureus bacteremia/endocarditis trial, some patients who received prior or concomitant treatment with an HMG-CoA reductase inhibitor developed elevated CPK [see Adverse Reactions (6.1)]. Experience with the coadministration of HMG-CoA reductase inhibitors and daptomycin for injection in patients is limited; therefore, consideration should be given to suspending use of HMG-CoA reductase inhibitors temporarily in patients receiving DAPZURA RT.
7.2 Drug-Laboratory Test Interactions
Clinically relevant plasma concentrations of daptomycin have been observed to cause a significant concentration-dependent false prolongation of prothrombin time (PT) and elevation of International Normalized Ratio (INR) when certain recombinant thromboplastin reagents are utilized for the assay. The possibility of an erroneously elevated PT/INR result due to interaction with a recombinant thromboplastin reagent may be minimized by drawing specimens for PT or INR testing near the time of trough plasma concentrations of daptomycin. However, sufficient daptomycin concentrations may be present at trough to cause interaction.
If confronted with an abnormally high PT/INR result in a patient being treated with DAPZURA RT, it is recommended that clinicians:
- 12. Repeat the assessment of PT/INR, requesting that the specimen be drawn just prior to the next DAPZURA RT dose (i.e., at trough concentration). If the PT/INR value obtained at trough remains substantially elevated above what would otherwise be expected, consider evaluating PT/INR utilizing an alternative method.
- 13. Evaluate for other causes of abnormally elevated PT/INR results.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Limited published data on use of daptomycin for injection in pregnant women are insufficient to inform a drug-associated risk for major birth defects and miscarriage. In animal reproduction studies performed in rats and rabbits daptomycin was administered intravenously during organogenesis at doses 2 and 4–times, respectively, the recommended 6 mg/kg human dose (on a body surface area basis). No evidence of adverse developmental outcomes was observed.
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Animal Data
In pregnant rats, daptomycin was administered intravenously at doses of 5, 20, or 75 mg/kg/day during the gestation days 6 to 18. Maternal body weight gain was decreased at 75 mg/kg/day. No embryo/fetal effects were noted at the highest dose of 75 mg/kg/day, a dose approximately 2-fold higher than in humans at the recommended maximum dose of 6 mg/kg (based on body surface area).
In pregnant rabbits, daptomycin was administered intravenously at doses of 5, 20, or 75 mg/kg/day during the gestation days 6 to 15. Maternal body weight gain and food consumption were decreased at 75 mg/kg/day. No embryo/fetal effects were noted at the highest dose of 75 mg/kg/day, a dose approximately 4-fold higher than in humans at the maximum recommended dose of 6 mg/kg (based on body surface area).
In a combined fertility and pre/postnatal development study, daptomycin was administered intravenously to female rats at doses of 2, 25, 75 mg/kg/day from 14-days pre-mating through lactation/postpartum day 20). No effects on pre/postnatal development were observed up to the highest dose of 75 mg/kg/day, a dose approximately 2-fold higher than the maximum recommended human dose of 6 mg/kg (based on body surface area)1.
8.2 Lactation
Risk Summary
Limited published data report that daptomycin is present in human milk at infant doses of 0.1% of the maternal dose [see Data]2,3,4. There is no information on the effects of daptomycin on the breastfed infant or the effects of daptomycin on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for daptomycin for injection and any potential adverse effects on the breastfed infant from daptomycin for injection or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of daptomycin for injection in the treatment of cSSSI and S. aureus bloodstream infections (bacteremia) have been established in the age groups 1 to 17 years of age. Use of daptomycin for injection in these age groups is supported by evidence from adequate and well-controlled studies in adults, with additional data from pharmacokinetic studies in pediatric patients, and from safety, efficacy and PK studies in pediatric patients with cSSSI and S. aureus bloodstream infections [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1, 14.2)].
Safety and effectiveness in pediatric patients below the age of one year have not been established. Avoid use of DAPZURA RT in pediatric patients younger than one year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs [see Warnings and Precautions (5.7) and Nonclinical Toxicology (13.2)].
DAPZURA RT is not indicated in pediatric patients with renal impairment because dosage has not been established in these patients.
DAPZURA RT has not been studied in pediatric patients with other bacterial infections.
DAPZURA RT is contraindicated in adult and pediatric patients with HFI. Because a diagnosis of HFI may not yet be established in pediatric patients, obtain a careful history of HFI symptoms with sorbitol/fructose/sucrose exposure prior to administration of DAPZURA RT [see Warnings and Precautions (5.11)].
8.5 Geriatric Use
Of the 534 adult patients treated with daptomycin for injection in Phase 3 controlled clinical trials of complicated skin and skin structure infections (cSSSI), 27% were 65 years of age or older and 12% were 75 years of age or older. Of the 120 adult patients treated with daptomycin for injection in the Phase 3 controlled clinical trial of S. aureus bacteremia/endocarditis, 25% were 65 years of age or older and 16% were 75 years of age or older. In Phase 3 adult clinical trials of cSSSI and S. aureus bacteremia/endocarditis, clinical success rates were lower in patients ≥65 years of age than in patients <65 years of age. In addition, treatment-emergent adverse events were more common in patients ≥65 years of age than in patients <65 years of age.
The exposure of daptomycin was higher in healthy elderly subjects than in healthy young adult subjects. However, no adjustment of DAPZURA RT dosage is warranted for elderly patients with creatinine clearance (CLCR) ≥30 mL/min [see Dosage and Administration (2.6) and Clinical Pharmacology (12.3)].
8.6 Patients with Renal Impairment
Daptomycin is eliminated primarily by the kidneys; therefore, a modification of DAPZURA RT dosage interval is recommended for adult patients with CLCR <30 mL/min, including patients receiving hemodialysis or continuous ambulatory peritoneal dialysis (CAPD).
In adult patients with renal impairment, both renal function and creatine phosphokinase (CPK) should be monitored more frequently than once weekly [see Dosage and Administration (2.6), Warnings and Precautions (5.2, 5.10), and Clinical Pharmacology (12.3)].
The dosage regimen for DAPZURA RT in pediatric patients with renal impairment has not been established.
-
10 OVERDOSAGE
In the event of overdosage, supportive care is advised with maintenance of glomerular filtration. Daptomycin is cleared slowly from the body by hemodialysis (approximately 15% of the administered dose is removed over 4 hours) and by peritoneal dialysis (approximately 11% of the administered dose is removed over 48 hours). The use of high-flux dialysis membranes during 4 hours of hemodialysis may increase the percentage of dose removed compared with that removed by low-flux membranes.
-
11 DESCRIPTION
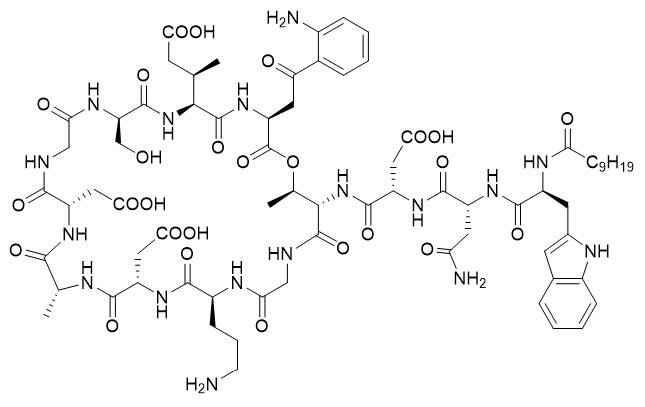
DAPZURA RT (daptomycin for injection) contains daptomycin, a cyclic lipopeptide antibacterial agent derived from the fermentation of Streptomyces roseosporus. The chemical name is N-decanoyl-L-tryptophyl-D-asparaginyl-L-aspartyl-L-threonylglycyl-L-ornithyl-L-aspartyl-D-alanyl-L-aspartylglycyl-D-seryl-threo-3-methyl-L-glutamyl-3-anthraniloyl-L-alanine ℇ1-lactone. The chemical structure is:
The empirical formula is C72H101N17O26; the molecular weight is 1620.67. DAPZURA RTis supplied in a single-dose vial as a sterile, preservative-free, pale yellow to light brown, lyophilized powder containing 500 mg of daptomycin for intravenous (IV) use following reconstitution [see Dosage and Administration (2.7)]. Each vial also contains 238 mg sorbitol and 238 mg of mannitol and sodium hydroxide and/or hydrochloric acid is used to adjust the pH. The pH of the solution upon reconstitution is 6.8. Freshly reconstituted solutions of DAPZURA RT range in color from pale yellow to light brown.
-
12 CLINICAL PHARMACOLOGY
12.2 Pharmacodynamics
Based on animal models of infection, the antimicrobial activity of daptomycin appears to correlate with the AUC/MIC (area under the concentration-time curve/minimum inhibitory concentration) ratio for certain pathogens, including S. aureus. The principal pharmacokinetic/pharmacodynamic parameter best associated with clinical and microbiological cure has not been elucidated in clinical trials with daptomycin for injection.
12.3 Pharmacokinetics
Daptomycin for Injection Administered over a 30-Minute Period in Adults
The mean and standard deviation (SD) pharmacokinetic parameters of daptomycin at steady-state following intravenous (IV) administration of daptomycin for injection over a 30-minute period at 4 to 12 mg/kg every 24h to healthy young adults are summarized in .
Table 12. Mean (SD) Daptomycin Pharmacokinetic Parameters in Healthy Adult Volunteers at Steady-State Dose*†
(mg/kg)Pharmacokinetic Parameters‡ - * Daptomycin for injection was administered by IV infusion over a 30-minute period.
- † Doses of daptomycin for injection in excess of 6 mg/kg have not been approved.
- ‡ AUC0-24, area under the concentration-time curve from 0 to 24 hours; t1/2, elimination half-life; Vss, volume of distribution at steady-state; CLT, total plasma clearance; Cmax, maximum plasma concentration.
AUC0-24 (mcgh/mL)
t1/2 (h)
Vss (L/kg)
CLT (mL/h/kg)
Cmax (mcg/mL)
4 (N=6)
494 (75)
8.1 (1.0)
0.096 (0.009)
8.3 (1.3)
57.8 (3.0)
6 (N=6)
632 (78)
7.9 (1.0)
0.101 (0.007)
9.1 (1.5)
93.9 (6.0)
8 (N=6)
858 (213)
8.3 (2.2)
0.101 (0.013)
9.0 (3.0)
123.3 (16.0)
10 (N=9)
1039 (178)
7.9 (0.6)
0.098 (0.017)
8.8 (2.2)
141.1 (24.0)
12 (N=9)
1277 (253)
7.7 (1.1)
0.097 (0.018)
9.0 (2.8)
183.7 (25.0)
Daptomycin pharmacokinetics were generally linear and time-independent at daptomycin for injection doses of 4 to 12 mg/kg every 24h administered by IV infusion over a 30-minute period for up to 14 days. Steady-state trough concentrations were achieved by the third daily dose. The mean (SD) steady-state trough concentrations attained following the administration of 4, 6, 8, 10, and 12 mg/kg every 24h were 5.9 (1.6), 6.7 (1.6), 10.3 (5.5), 12.9 (2.9), and 13.7 (5.2) mcg/mL, respectively.
Daptomycin for Injection Administered over a 2-Minute Period in Adults
Following IV administration of daptomycin for injection over a 2-minute period to healthy adult volunteers at doses of 4 mg/kg (N=8) and 6 mg/kg (N=12), the mean (SD) steady-state systemic exposure (AUC) values were 475 (71) and 701 (82) mcgh/mL, respectively. Values for maximum plasma concentration (Cmax) at the end of the 2-minute period could not be determined adequately in this study. However, using pharmacokinetic parameters from 14 healthy adult volunteers who received a single dose of daptomycin for injection 6 mg/kg IV administered over a 30-minute period in a separate study, steady-state Cmax values were simulated for daptomycin for injection 4 and 6 mg/kg IV administered over a 2-minute period. The simulated mean (SD) steady-state Cmax values were 77.7 (8.1) and 116.6 (12.2) mcg/mL, respectively.
Distribution
Daptomycin is reversibly bound to human plasma proteins, primarily to serum albumin, in a concentration-independent manner. The overall mean binding ranges from 90 to 93%.
In clinical studies, mean serum protein binding in adult subjects with creatinine clearance (CLCR) ≥30 mL/min was comparable to that observed in healthy adult subjects with normal renal function. However, there was a trend toward decreasing serum protein binding among subjects with CLCR <30 mL/min (88%), including those receiving hemodialysis (86%) and continuous ambulatory peritoneal dialysis (CAPD) (84%). The protein binding of daptomycin in adult subjects with moderate hepatic impairment (Child-Pugh Class B) was similar to that in healthy adult subjects.
The volume of distribution at steady-state (Vss) of daptomycin in healthy adult subjects was approximately 0.1 L/kg and was independent of dose.
Metabolism
In in vitro studies, daptomycin was not metabolized by human liver microsomes.
In 5 healthy adults after infusion of radiolabeled 14C-daptomycin, the plasma total radioactivity was similar to the concentration determined by microbiological assay. Inactive metabolites were detected in urine, as determined by the difference between total radioactive concentrations and microbiologically active concentrations. In a separate study, no metabolites were observed in plasma on Day 1 following the administration of daptomycin for injection at 6 mg/kg to adult subjects. Minor amounts of three oxidative metabolites and one unidentified compound were detected in urine. The site of metabolism has not been identified.
Excretion
Daptomycin is excreted primarily by the kidneys. In a mass balance study of 5 healthy adult subjects using radiolabeled daptomycin, approximately 78% of the administered dose was recovered from urine based on total radioactivity (approximately 52% of the dose based on microbiologically active concentrations), and 5.7% of the administered dose was recovered from feces (collected for up to 9 days) based on total radioactivity.
Specific Populations
Patients with Renal Impairment
Population-derived pharmacokinetic parameters were determined for infected adult patients (complicated skin and skin structure infections [cSSSI] and S. aureus bacteremia) and noninfected adult subjects with various degrees of renal function (Table 13). Total plasma clearance (CLT), elimination half-life (t1/2), and volume of distribution at steady-state (Vss) in patients with cSSSI were similar to those in patients with S. aureus bacteremia. Following administration of daptomycin for injection 4 mg/kg every 24h by IV infusion over a 30-minute period, the mean CLT was 9%, 22%, and 46% lower among subjects and patients with mild (CLCR 50– 80 mL/min), moderate (CLCR 30–<50 mL/min), and severe (CLCR <30 mL/min) renal impairment, respectively, than in those with normal renal function (CLCR >80 mL/min). The mean steady-state systemic exposure (AUC), t1/2, and Vss increased with decreasing renal function, although the mean AUC for patients with CLCR 30–80 mL/min was not markedly different from the mean AUC for patients with normal renal function. The mean AUC for patients with CLCR <30 mL/min and for patients on dialysis (CAPD and hemodialysis dosed post-dialysis) was approximately 2 and 3 times higher, respectively, than for patients with normal renal function. The mean Cmax ranged from 60 to 70 mcg/mL in patients with CLCR ≥30 mL/min, while the mean Cmax for patients with CLCR <30 mL/min ranged from 41 to 58 mcg/mL. After administration of daptomycin for injection 6 mg/kg every 24h by IV infusion over a 30-minute period, the mean Cmax ranged from 80 to 114 mcg/mL in patients with mild to moderate renal impairment and was similar to that of patients with normal renal function.
Table 13. Mean (SD) Daptomycin Population Pharmacokinetic Parameters Following Infusion of Daptomycin for Injection 4 mg/kg or 6 mg/kg to Infected Adult Patients and Noninfected Adult Subjects with Various Degrees of Renal Function Renal Function Pharmacokinetic Parameters* - * CLCR, creatinine clearance estimated using the Cockcroft-Gault equation with actual body weight; CAPD, continuous ambulatory peritoneal dialysis; AUC0-∞, area under the concentration-time curve extrapolated to infinity; AUCss, area under the concentration-time curve calculated over the 24-hour dosing interval at steady-state; Cmin,ss, trough concentration at steady-state; NA, not applicable.
- † Parameters obtained following a single dose from patients with complicated skin and skin structure infections and healthy subjects.
- ‡ Parameters obtained at steady-state from patients with S. aureus bacteremia.
t1/2†
(h)
4 mg/kg
Vss†
(L/kg)
4 mg/kg
CLT† (mL/h/kg)
4 mg/kgAUC0-∞†(mcgh/mL)
4 mg/kgAUCss‡(mcgh/mL)
6 mg/kgCmin,ss‡(mcg/mL)
6 mg/kgNormal
(CLCR >80 mL/min)
9.39 (4.74)
N=165
0.13 (0.05)
N=165
10.9 (4.0)
N=165
417 (155)
N=165
545 (296)
N=62
6.9 (3.5)
N=61
Mild Renal
Impairment
(CLCR 50– 80 mL/min)
10.75 (8.36)
N=64
0.12 (0.05)
N=64
9.9 (4.0)
N=64
466 (177)
N=64
637 (215)
N=29
12.4 (5.6)
N=29
Moderate Renal
Impairment
(CLCR 30– <50 mL/min)
14.70 (10.50)
N=24
0.15 (0.06)
N=24
8.5 (3.4)
N=24
560 (258)
N=24
868 (349)
N=15
19.0 (9.0)
N=14
Severe Renal Impairment
(CLCR <30 mL/min)
27.83 (14.85)
N=8
0.20 (0.15)
N=8
5.9 (3.9)
N=8
925 (467)
N=8
1050 (892)
N=2
24.4
(21.4)
N=2
Hemodialysis
30.51 (6.51)
N=16
0.16 (0.04)
N=16
3.9 (2.1)
N=16
1193 (399)
N=16
NA
NA
CAPD
27.56 (4.53)
N=5
0.11 (0.02)
N=5
2.9 (0.4)
N=5
1409 (238)
N=5
NA
NA
Note: Daptomycin for injection was administered over a 30-minute period.
Because renal excretion is the primary route of elimination, adjustment of DAPZURA RTdosage interval is necessary in adult patients with severe renal impairment (CLCR <30 mL/min) [see Dosage and Administration (2.6)].
Patients with Hepatic Impairment
The pharmacokinetics of daptomycin were evaluated in 10 adult subjects with moderate hepatic impairment (Child-Pugh Class B) and compared with those in healthy adult volunteers (N=9) matched for gender, age, and weight. The pharmacokinetics of daptomycin were not altered in subjects with moderate hepatic impairment. No dosage adjustment is warranted when DAPZURA RT is administered to patients with mild to moderate hepatic impairment. The pharmacokinetics of daptomycin in patients with severe hepatic impairment (Child-Pugh Class C) have not been evaluated.
Gender
No clinically significant gender-related differences in daptomycin pharmacokinetics have been observed. No dosage adjustment is warranted based on gender when DAPZURA RTis administered.
Geriatric Patients
The pharmacokinetics of daptomycin were evaluated in 12 healthy elderly subjects (≥75 years of age) and 11 healthy young adult controls (18 to 30 years of age). Following administration of a single 4 mg/kg dose of daptomycin for injection by IV infusion over a 30-minute period, the mean total clearance of daptomycin was approximately 35% lower and the mean AUC0-∞ was approximately 58% higher in elderly subjects than in healthy young adult subjects. There were no differences in Cmax [see Use in Specific Populations (8.5)].
Obese Patients
The pharmacokinetics of daptomycin were evaluated in 6 moderately obese (Body Mass Index [BMI] 25 to 39.9 kg/m2) and 6 extremely obese (BMI ≥40 kg/m2) adult subjects and controls matched for age, gender, and renal function. Following administration of daptomycin for injection by IV infusion over a 30-minute period as a single 4 mg/kg dose based on total body weight, the total plasma clearance of daptomycin normalized to total body weight was approximately 15% lower in moderately obese subjects and 23% lower in extremely obese subjects than in nonobese controls. The AUC0-∞ of daptomycin was approximately 30% higher in moderately obese subjects and 31% higher in extremely obese subjects than in nonobese controls. The differences were most likely due to differences in the renal clearance of daptomycin. No adjustment of DAPZURA RT dosage is warranted in obese patients.
Pediatric Patients
The pharmacokinetics of daptomycin in pediatric subjects was evaluated in 3 single-dose pharmacokinetic studies. In general, body weight-normalized total body clearance in pediatric patients was higher than in adults and increased with a decrease of age, whereas elimination half-life tends to decrease with a decrease of age. Body weight-normalized total body clearance and elimination half-life of daptomycin in children 2 to 6 years of age were similar at different doses.
A study was conducted to assess safety, efficacy, and pharmacokinetics of daptomycin in pediatric patients (1 to 17 years old, inclusive) with cSSSI caused by Gram-positive pathogens. Patients were enrolled into 4 age groups [see Clinical Studies (14.1)], and intravenous daptomycin for injection doses of 5 to 10 mg/kg once daily were administered. Following administration of multiple doses, daptomycin exposure (AUCss and Cmax,ss) was similar across different age groups after dose adjustment based on body weight and age (Table 14).
Table 14. Mean (SD) Daptomycin Population Pharmacokinetic Parameters in cSSSI Pediatric Patients Age Pharmacokinetic Parameters - * Mean is calculated from N=2
Dose (mg/kg)
- Infusion
Duration
(min)
AUCss (mcgh/mL)
t1/2
(h)
Vss (mL)
CLT (mL/h/kg)
Cmax,ss
(mcg/mL)
12 to 17 years
(N=6)
5
30
434 (67.9)
7.1 (0.9)
8200
(3250)
11.8 (2.15)
76.4 (6.75)
7 to 11 years
(N=2)
7
30
543*
6.8*
4470*
13.2*
92.4*
2 to 6 years
(N=7)
9
60
452 (93.1)
4.6 (0.8)
2750 (832)
20.8 (4.29)
90.3 (14.0)
1 to less than 2 years
(N=27)
10
60
462 (138)
4.8 (0.6)
1670
(446)
23.1 (5.43)
81.6 (20.7)
AUCss, area under the concentration-time curve at steady state; CLT, clearance normalized to body weight;
Vss, volume of distribution at steady state; t½, terminal half-life
A study was conducted to assess safety, efficacy, and pharmacokinetics of daptomycin in pediatric patients with S. aureus bacteremia. Patients were enrolled into 3 age groups [see Clinical Studies (14.2)], and intravenous doses of 7 to 12 mg/kg once daily were administered. Following administration of multiple doses, daptomycin exposure (AUCss and Cmax,ss) was similar across different age groups after dose adjustment based on body weight and age (Table 15).
Table 15. Mean (SD) of Daptomycin Pharmacokinetics in Bacteremia Pediatric Patients Age Pharmacokinetic Parameters Dose
(mg/kg)
Infusion
Duration
(min)
AUCss
(mcgh/mL)
t1/2 (h)
Vss
(mL)
CLT
(mL/h/kg)
Cmax,ss
(mcg/mL)
12 to 17 years
(N=13)
7
30
656 (334)
7.5 (2.3)
6420
(1980)
12.4 (3.9)
104 (35.5)
7 to 11 years
(N=19)
9
30
579 (116)
6.0 (0.8)
4510
(1470)
15.9 (2.8)
104 (14.5)
2 to 6 years
(N=19)
12
60
620 (109)
5.1 (0.6)
2200 (570)
19.9 (3.4)
106 (12.8)
AUCss, area under the concentration-time curve at steady state; CLT, clearance normalized to body weight;
Vss, volume of distribution at steady state; t½, terminal half-life
No patients 1 to <2 years of age were enrolled in the study. Simulation using a population
pharmacokinetic model demonstrated that the AUCss of daptomycin in pediatric patients 1 to <2 years of age receiving 12 mg/kg once daily would be comparable to that in adult patients receiving 6 mg/kg once daily.Drug Interactions Studies
In Vitro Studies
In vitro studies with human hepatocytes indicate that daptomycin does not inhibit or induce the activities of the following human cytochrome P450 isoforms: 1A2, 2A6, 2C9, 2C19, 2D6, 2E1, and 3A4. It is unlikely that daptomycin will inhibit or induce the metabolism of drugs metabolized by the P450 system.
Aztreonam
In a study in which 15 healthy adult subjects received a single dose of daptomycin for injection 6 mg/kg IV and a combination dose of daptomycin for injection 6 mg/kg IV and aztreonam 1 g IV, administered over a 30-minute period, the Cmax and AUC0-∞ of daptomycin were not significantly altered by aztreonam.
Tobramycin
In a study in which 6 healthy adult males received a single dose of daptomycin for injection 2 mg/kg IV, tobramycin 1 mg/kg IV, and both in combination, administered over a 30-minute period, the mean Cmax and AUC0-∞ of daptomycin were 12.7% and 8.7% higher, respectively, when daptomycin for injection was coadministered with tobramycin. The mean Cmax and AUC0-∞ of tobramycin were 10.7% and 6.6% lower, respectively, when tobramycin was coadministered with daptomycin for injection. These differences were not statistically significant. The interaction between daptomycin and tobramycin with a clinical dose of DAPZURA RT is unknown.
Warfarin
In 16 healthy adult subjects, administration of daptomycin for injection 6 mg/kg every 24h by IV infusion over a 30-minute period for 5 days, with coadministration of a single oral dose of warfarin (25 mg) on the 5th day, had no significant effect on the pharmacokinetics of either drug and did not significantly alter the INR (International Normalized Ratio).
Simvastatin
In 20 healthy adult subjects on a stable daily dose of simvastatin 40 mg, administration of daptomycin for injection 4 mg/kg every 24h by IV infusion over a 30-minute period for 14 days (N=10) had no effect on plasma trough concentrations of simvastatin and was not associated with a higher incidence of adverse events, including skeletal myopathy, than in subjects receiving placebo once daily (N=10) [see Warnings and Precautions (5.2) and Drug Interactions (7.1)].
Probenecid
Concomitant administration of probenecid (500 mg 4 times daily) and a single dose of daptomycin for injection 4 mg/kg by IV infusion over a 30-minute period in adults did not significantly alter the Cmax or AUC0-∞ of daptomycin.
12.4 Microbiology
Daptomycin belongs to the cyclic lipopeptide class of antibacterials. Daptomycin has clinical utility in the treatment of infections caused by aerobic, Gram-positive bacteria. The in vitro spectrum of activity of daptomycin encompasses most clinically relevant Gram-positive pathogenic bacteria.
Daptomycin exhibits rapid, concentration-dependent bactericidal activity against Gram-positive bacteria in vitro. This has been demonstrated both by time-kill curves and by MBC/MIC (minimum bactericidal concentration/minimum inhibitory concentration) ratios using broth dilution methodology. Daptomycin maintained bactericidal activity in vitro against stationary phase S. aureus in simulated endocardial vegetations. The clinical significance of this is not known.
Mechanism of Action
Daptomycin binds to bacterial cell membranes and causes a rapid depolarization of membrane potential. This loss of membrane potential causes inhibition of DNA, RNA, and protein synthesis, which results in bacterial cell death.
Resistance
The mechanism(s) of daptomycin resistance is not fully understood. Currently, there are no known transferable elements that confer resistance to daptomycin.
Interactions with Other Antibacterials
In vitro studies have investigated daptomycin interactions with other antibacterials. Antagonism, as determined by kill curve studies, has not been observed. In vitro synergistic interactions of daptomycin with aminoglycosides, β-lactam antibacterials, and rifampin have been shown against some isolates of staphylococci (including some methicillin-resistant isolates) and enterococci (including some vancomycin-resistant isolates).
Complicated Skin and Skin Structure Infection (cSSSI) Trials in Adults
The emergence of daptomycin non-susceptible isolates occurred in 2 infected patients across the set of Phase 2 and pivotal Phase 3 clinical trials of cSSSI in adult patients. In one case, a non-susceptible S. aureus was isolated from a patient in a Phase 2 trial who received daptomycin for injection at less than the protocol-specified dose for the initial 5 days of therapy. In the second case, a non-susceptible Enterococcus faecalis was isolated from a patient with an infected chronic decubitus ulcer who was enrolled in a salvage trial.
S. aureus Bacteremia/Endocarditis and Other Post-Approval Trials in Adults
In subsequent clinical trials in adult patients, non-susceptible isolates were recovered. S. aureus was isolated from a patient in a compassionate-use trial and from 7 patients in the S. aureus bacteremia/endocarditis trial [see Clinical Studies (14.2)]. An E. faecium was isolated from a patient in a vancomycin-resistant enterococci trial.
Antimicrobial Activity
Daptomycin has been shown to be active against most isolates of the following microorganisms both in vitro and in clinical infections [see Indications and Usage (1)].
- Gram-Positive Bacteria
- Enterococcus faecalis (vancomycin-susceptible isolates only)
- Staphylococcus aureus (including methicillin-resistant isolates)
- Streptococcus agalactiae
- Streptococcus dysgalactiae subsp. equisimilis
- Streptococcus pyogenes
The following in vitro data are available, but their clinical significance is unknown. At least 90 percent of the following bacteria exhibit an in vitro minimum inhibitory concentration (MIC) less than or equal to the susceptible breakpoint for daptomycin against isolates of similar genus or organism group. However, the efficacy of daptomycin in treating clinical infections caused by these bacteria has not been established in adequate and well-controlled clinical trials.
- Gram-Positive Bacteria
- Corynebacterium jeikeium
- Enterococcus faecalis (vancomycin-resistant isolates)
- Enterococcus faecium (including vancomycin-resistant isolates)
- Staphylococcus epidermidis (including methicillin-resistant isolates)
- Staphylococcus haemolyticus
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for daptomycin, please see: https://www.fda.gov/STIC.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity studies in animals have not been conducted to evaluate the carcinogenic potential of daptomycin for injection. However, neither mutagenic nor clastogenic potential was found in a battery of genotoxicity tests, including the Ames assay, a mammalian cell gene mutation assay, a test for chromosomal aberrations in Chinese hamster ovary cells, an in vivo micronucleus assay, an in vitro DNA repair assay, and an in vivo sister chromatid exchange assay in Chinese hamsters.
Daptomycin did not affect the fertility or reproductive performance of male and female rats when administered intravenously at doses of 25, 75, or 150 mg/kg/day, which is approximately up to 9 times the estimated human exposure level based upon AUCs (or approximately up to 4 times the recommended human dose of 6 mg/kg based on body surface area comparison).
13.2 Animal Toxicology and/or Pharmacology
Adult Animals
In animals, daptomycin administration has been associated with effects on skeletal muscle. However, there were no changes in cardiac or smooth muscle. Skeletal muscle effects were characterized by microscopic degenerative/regenerative changes and variable elevations in creatine phosphokinase (CPK). No fibrosis or rhabdomyolysis was evident in repeat-dose studies up to the highest doses tested in rats (150 mg/kg/day) and dogs (100 mg/kg/day). The degree of skeletal myopathy showed no increase when treatment was extended from 1 month to up to 6 months. Severity was dose-dependent. All muscle effects, including microscopic changes, were fully reversible within 30 days following the cessation of dosing.
In adult animals, effects on peripheral nerve (characterized by axonal degeneration and frequently accompanied by significant losses of patellar reflex, gag reflex, and pain perception) were observed at daptomycin doses higher than those associated with skeletal myopathy. Deficits in the dogs’ patellar reflexes were seen within 2 weeks after the start of treatment at 40 mg/kg/day (9 times the human Cmax at the 6 mg/kg/day dose), with some clinical improvement noted within 2 weeks after the cessation of dosing. However, at 75 mg/kg/day for 1 month, 7 of 8 dogs failed to regain full patellar reflex responses within a 3-month recovery period. In a separate study in dogs receiving doses of 75 and 100 mg/kg/day for 2 weeks, minimal residual histological changes were noted at 6 months after the cessation of dosing. However, recovery of peripheral nerve function was evident.
Tissue distribution studies in rats showed that daptomycin is retained in the kidney but appears to penetrate the blood-brain barrier only minimally following single and multiple doses.
Juvenile Animals
Target organs of daptomycin-related effects in 7-week-old juvenile dogs were skeletal muscle and nerve, the same target organs as in adult dogs. In juvenile dogs, nerve effects were noted at lower daptomycin blood concentrations than in adult dogs following 28 days of dosing. In contrast to adult dogs, juvenile dogs also showed evidence of effects in nerves of the spinal cord as well as peripheral nerves after 28 days of dosing. No nerve effects were noted in juvenile dogs following 14 days of dosing at doses up to 75 mg/kg/day.
Administration of daptomycin to 7-week-old juvenile dogs for 28 days at doses of 50 mg/kg/day produced minimal degenerative effects on the peripheral nerve and spinal cord in several animals, with no corresponding clinical signs. A dose of 150 mg/kg/day for 28 days produced minimal degeneration in the peripheral nerve and spinal cord as well as minimal to mild degeneration of the skeletal muscle in a majority of animals, accompanied by slight to severe muscle weakness evident in most dogs. Following a 28-day recovery phase, microscopic examination revealed recovery of the skeletal muscle and the ulnar nerve effects, but nerve degeneration in the sciatic nerve and spinal cord was still observed in all 150 mg/kg/day dogs.
Following once-daily administration of daptomycin to juvenile dogs for 28 days, microscopic effects in nerve tissue were noted at a Cmax value of 417 mcg/mL, which is approximately 3-fold less than the Cmax value associated with nerve effects in adult dogs treated once daily with daptomycin for 28 days (1308 mcg/mL).
Neonatal Animals
Neonatal dogs (4 to 31 days old) were more sensitive to daptomycin-related adverse nervous system and/or muscular system effects than either juvenile or adult dogs. In neonatal dogs, adverse nervous system and/or muscular system effects were associated with a Cmax value approximately 3-fold less than the Cmax in juvenile dogs, and 9-fold less than the Cmax in adult dogs following 28 days of dosing. At a dose of 25 mg/kg/day with associated Cmax and AUCinf values of 147 mcg/mL and 717 mcgh/mL, respectively (1.6 and 1.0-fold the adult human Cmax and AUC, respectively, at the 6 mg/kg/day dose), mild clinical signs of twitching and one incidence of muscle rigidity were observed with no corresponding effect on body weight. These effects were found to be reversible within 28 days after treatment had stopped.
At higher dose levels of 50 and 75 mg/kg/day with associated Cmax and AUCinf values of ≥321 mcg/mL and ≥1470 mcgh/mL, respectively, marked clinical signs of twitching, muscle rigidity in the limbs, and impaired use of limbs were observed. Resulting decreases in body weights and overall body condition at doses ≥50 mg/kg/day necessitated early discontinuation by postnatal day (PND) 19.
Histopathological assessment did not reveal any daptomycin-related changes in the peripheral and central nervous system tissue, as well as in the skeletal muscle or other tissues assessed, at any dose level.
No adverse effects were observed in the dogs that received daptomycin at 10 mg/kg/day, the NOAEL, with associated Cmax and AUCinf values of 62 mcg/mL and 247 mcgh/mL, respectively (or 0.6 and 0.4-fold the adult human Cmax and AUC, respectively at the 6 mg/kg dose).
-
14 CLINICAL STUDIES
14.1 Complicated Skin and Skin Structure Infections
Adults with cSSSI
Adult patients with clinically documented complicated skin and skin structure infections (cSSSI) (Table 16) were enrolled in two randomized, multinational, multicenter, investigator-blinded trials comparing daptomycin for injection (4 mg/kg IV every 24h) with either vancomycin (1 g IV every 12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 4 to 12 g IV per day). Patients could switch to oral therapy after a minimum of 4 days of IV treatment if clinical improvement was demonstrated. Patients known to have bacteremia at baseline were excluded. Patients with creatinine clearance (CLCR) between 30 and 70 mL/min were to receive a lower dose of daptomycin for injection as specified in the protocol; however, the majority of patients in this subpopulation did not have the dose of daptomycin for injection adjusted.
Table 16. Investigator’s Primary Diagnosis in the cSSSI Trials in Adult Patients (Population: ITT) Primary Diagnosis Adult Patients (Daptomycin for Injection / Comparator*) - * Comparator: vancomycin (1 g IV every 12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 4 to 12 g/day IV in divided doses).
- † The majority of cases were subsequently categorized as complicated cellulitis, major abscesses, or traumatic wound infections.
Study 9801
N=264 / N=266
Study 9901
N=270 / N=292
Pooled
N=534 / N=558
Wound Infection
99 (38%) / 116 (44%)
102 (38%) / 108 (37%)
201 (38%) / 224 (40%)
Major Abscess
55 (21%) / 43 (16%)
59 (22%) / 65 (22%)
114 (21%) / 108 (19%)
Ulcer Infection
71 (27%) / 75 (28%)
53 (20%) / 68 (23%)
124 (23%) / 143 (26%)
Other Infection†
39 (15%) / 32 (12%)
56 (21%) / 51 (18%)
95 (18%) / 83 (15%)
One trial was conducted primarily in the United States and South Africa (study 9801), and the second was conducted at non-US sites only (study 9901). The two trials were similar in design but differed in patient characteristics, including history of diabetes and peripheral vascular disease. There were a total of 534 adult patients treated with daptomycin for injection and 558 treated with comparator in the two trials. The majority (89.7%) of patients received IV medication exclusively.
The efficacy endpoints in both trials were the clinical success rates in the intent-to-treat (ITT) population and in the clinically evaluable (CE) population. In study 9801, clinical success rates in the ITT population were 62.5% (165/264) in patients treated with daptomycin for injection and 60.9% (162/266) in patients treated with comparator drugs. Clinical success rates in the CE population were 76.0% (158/208) in patients treated with daptomycin for injection and 76.7% (158/206) in patients treated with comparator drugs. In study 9901, clinical success rates in the ITT population were 80.4% (217/270) in patients treated with daptomycin for injection and 80.5% (235/292) in patients treated with comparator drugs. Clinical success rates in the CE population were 89.9% (214/238) in patients treated with daptomycin for injection and 90.4% (226/250) in patients treated with comparator drugs.
The success rates by pathogen for microbiologically evaluable patients are presented in Table 17.
Table 17. Clinical Success Rates by Infecting Pathogen in the cSSSI Trials in Adult Patients (Population: Microbiologically Evaluable) - * Comparator: vancomycin (1 g IV every 12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 4 to 12 g/day IV in divided doses).
- † As determined by the central laboratory.
Pathogen
Success Rate n/N (%)
Daptomycin for Injection
Comparator*
Methicillin-susceptible Staphylococcus aureus (MSSA)†
170/198 (86%)
180/207 (87%)
Methicillin-resistant Staphylococcus aureus (MRSA)†
21/28 (75%)
25/36 (69%)
Streptococcus pyogenes
79/84 (94%)
80/88 (91%)
Streptococcus agalactiae
23/27 (85%)
22/29 (76%)
Streptococcus dysgalactiae subsp. equisimilis
8/8 (100%)
9/11 (82%)
Enterococcus faecalis (vancomycin-susceptible only)
27/37 (73%)
40/53 (76%)
Pediatric Patients (1 to 17 Years of Age) with cSSSI
The cSSSI pediatric trial was a single prospective multi-center, randomized, comparative trial. A total of 396 pediatric patients aged 1 to 17 years with cSSSI caused by Gram positive pathogens were enrolled into the study. Patients known to have bacteremia, osteomyelitis, endocarditis, and pneumonia at baseline were excluded. Patients were enrolled in a stepwise approach into four age groups and given age-dependent doses of daptomycin for injection once daily for up to 14 days. The different age groups and doses evaluated were as follows: Adolescents (12 to 17 years) treated with 5 mg/kg of daptomycin for injection (n=113), Children (7 to 11 years) treated with 7 mg/kg of daptomycin for injection (n=113), Children (2 to 6 years) treated with 9 mg/kg of daptomycin for injection (n=125) and Infants (1 to <2 years) treated with 10 mg/kg (n= 45).
Patients were randomized 2:1 to receive daptomycin for injection or a standard of care (SOC) comparator, which included intravenous therapy with either vancomycin, clindamycin, or an anti-staphylococcal semi-synthetic penicillin (nafcillin, oxacillin, or cloxacillin). Patients could switch to oral therapy after clinical improvement was demonstrated (no minimum IV dosing was required).
The primary objective of this study was to evaluate the safety of daptomycin for injection. The clinical outcome was determined by resolution or improvement of symptoms at the End-of-Treatment (EOT), 3 days after the last dose, and Test-of-Cure (TOC), 7-14 days after the last dose. Investigator observed outcomes were verified in a blinded fashion. Of the 396 subjects randomized in the study, 389 subjects were treated with daptomycin for injection or comparator and included in the ITT population. Of these, 257 subjects were randomized to the daptomycin for injection group and 132 subjects were randomized to the comparator group. Approximately 95% of subjects switched to oral therapy. The mean day of switch was day 4, and ranged from day 1 to day 14. The clinical success rates determined at 7–14 days after last dose of therapy (IV and oral) (TOC visit) were 88% (227/257) for daptomycin for injection and 86% (114/132) for comparator.
14.2 S. aureus Bacteremia/Endocarditis
Adults with S. aureus Bacteremia/Endocarditis
The efficacy of daptomycin for injection in the treatment of adult patients with S. aureus bacteremia was demonstrated in a randomized, controlled, multinational, multicenter, open-label trial. In this trial, adult patients with at least one positive blood culture for S. aureus obtained within 2 calendar days prior to the first dose of study drug and irrespective of source were enrolled and randomized to either daptomycin for injection (6 mg/kg IV every 24h) or standard of care [an anti-staphylococcal semi-synthetic penicillin 2 g IV every 4h (nafcillin, oxacillin, cloxacillin, or flucloxacillin) or vancomycin 1 g IV every 12h, each with initial gentamicin 1 mg/kg IV every 8 hours for first 4 days]. Of the patients in the comparator group, 93% received initial gentamicin for a median of 4 days, compared with 1 patient (<1%) in the daptomycin for injection group. Patients with prosthetic heart valves, intravascular foreign material that was not planned for removal within 4 days after the first dose of study medication, severe neutropenia, known osteomyelitis, polymicrobial bloodstream infections, creatinine clearance <30 mL/min, and pneumonia were excluded.
Upon entry, patients were classified for likelihood of endocarditis using the modified Duke criteria (Possible, Definite, or Not Endocarditis). Echocardiography, including a transesophageal echocardiogram (TEE), was performed within 5 days following study enrollment. The choice of comparator agent was based on the oxacillin susceptibility of the S. aureus isolate. The duration of study treatment was based on the investigator’s clinical diagnosis. Final diagnoses and outcome assessments at Test of Cure (6 weeks after the last treatment dose) were made by a treatment-blinded Adjudication Committee, using protocol-specified clinical definitions and a composite primary efficacy endpoint (clinical and microbiological success) at the Test of Cure visit.
A total of 246 patients ≥18 years of age (124 daptomycin for injection, 122 comparator) with S. aureus bacteremia were randomized from 48 centers in the US and Europe. In the ITT population, 120 patients received daptomycin for injection and 115 received comparator (62 received an anti-staphylococcal semi-synthetic penicillin and 53 received vancomycin). Thirty-five patients treated with an anti-staphylococcal semi-synthetic penicillin received vancomycin initially for 1 to 3 days, pending final susceptibility results for the S. aureus isolates. The median age among the 235 patients in the ITT population was 53 years (range: 21 to 91 years); 30/120 (25%) in the daptomycin for injection group and 37/115 (32%) in the comparator group were ≥65 years of age. Of the 235 ITT patients, there were 141 (60%) males and 156 (66%) Caucasians across the two treatment groups. In addition, 176 (75%) of the ITT population had systemic inflammatory response syndrome (SIRS) at baseline and 85 (36%) had surgical procedures within 30 days prior to onset of the S. aureus bacteremia. Eighty-nine patients (38%) had bacteremia caused by methicillin-resistant S. aureus (MRSA). Entry diagnosis was based on the modified Duke criteria and comprised 37 (16%) Definite, 144 (61%) Possible, and 54 (23%) Not Endocarditis. Of the 37 patients with an entry diagnosis of Definite Endocarditis, all (100%) had a final diagnosis of infective endocarditis, and of the 144 patients with an entry diagnosis of Possible Endocarditis, 15 (10%) had a final diagnosis of infective endocarditis as assessed by the Adjudication Committee. Of the 54 patients with an entry diagnosis of Not Endocarditis, 1 (2%) had a final diagnosis of infective endocarditis as assessed by the Adjudication Committee.
In the ITT population, there were 182 patients with bacteremia and 53 patients with infective endocarditis as assessed by the Adjudication Committee, including 35 with right-sided endocarditis and 18 with left-sided endocarditis. The 182 patients with bacteremia comprised 121 with complicated S. aureus bacteremia and 61 with uncomplicated S. aureus bacteremia.
Complicated bacteremia was defined as S. aureus isolated from blood cultures obtained on at least 2 different calendar days, and/or metastatic foci of infection (deep tissue involvement), and classification of the patient as not having endocarditis according to the modified Duke criteria. Uncomplicated bacteremia was defined as S. aureus isolated from blood culture(s) obtained on a single calendar day, no metastatic foci of infection, no infection of prosthetic material, and classification of the patient as not having endocarditis according to the modified Duke criteria. The definition of right-sided infective endocarditis (RIE) used in the clinical trial was Definite or Possible Endocarditis according to the modified Duke criteria and no echocardiographic evidence of predisposing pathology or active involvement of either the mitral or aortic valve. Complicated RIE comprised patients who were not intravenous drug users, had a positive blood culture for MRSA, serum creatinine ≥2.5 mg/dL, or evidence of extrapulmonary sites of infection. Patients who were intravenous drug users, had a positive blood culture for methicillin-susceptible S. aureus (MSSA), had serum creatinine <2.5 mg/dL, and were without evidence of extrapulmonary sites of infection were considered to have uncomplicated RIE.
The coprimary efficacy endpoints in the trial were the Adjudication Committee success rates at the Test of Cure visit (6 weeks after the last treatment dose) in the ITT and Per Protocol (PP) populations. The overall Adjudication Committee success rates in the ITT population were 44.2% (53/120) in patients treated with daptomycin for injection and 41.7% (48/115) in patients treated with comparator (difference = 2.4% [95% CI −10.2, 15.1]).
The success rates in the PP population were 54.4% (43/79) in patients treated with daptomycin for injection and 53.3% (32/60) in patients treated with comparator (difference = 1.1% [95% CI −15.6, 17.8]).
Adjudication Committee success rates are shown in Table 18.
Table 18. Adjudication Committee Success Rates at Test of Cure in the S. aureus Bacteremia/Endocarditis Trial in Adult Patients (Population: ITT) Population Success Rate n/N (%) Difference:
Daptomycin for
Injection –Comparator
(Confidence Interval)- * Comparator: vancomycin (1 g IV every 12h) or an anti-staphylococcal semi-synthetic penicillin (i.e., nafcillin, oxacillin, cloxacillin, or flucloxacillin; 2 g IV every 4h), each with initial low-dose gentamicin.
- † 95% Confidence Interval
- ‡ 97.5% Confidence Interval (adjusted for multiplicity)
- § According to the modified Duke criteria5
- ¶ 99% Confidence Interval (adjusted for multiplicity)
Daptomycin for
Injection 6 mg/kg
Comparator*
Overall
53/120 (44%)
48/115 (42%)
2.4% (−10.2, 15.1)†
Baseline Pathogen
- Methicillin-susceptible S. aureus
33/74 (45%)
34/70 (49%)
−4.0% (−22.6, 14.6)‡
- Methicillin-resistant S. aureus
20/45 (44%)
14/44 (32%)
12.6% (−10.2, 35.5)‡
Entry Diagnosis§
- Definite or Possible Infective Endocarditis
41/90 (46%)
37/91 (41%)
4.9% (−11.6, 21.4)‡
- Not Infective Endocarditis
12/30 (40%)
11/24 (46%)
−5.8% (−36.2, 24.5)‡
Final Diagnosis
- Uncomplicated Bacteremia
18/32 (56%)
16/29 (55%)
1.1% (−31.7, 33.9)¶
- Complicated Bacteremia
26/60 (43%)
23/61 (38%)
5.6% (−17.3, 28.6)¶
- Right-Sided Infective Endocarditis
8/19 (42%)
7/16 (44%)
−1.6% (−44.9, 41.6)¶
- Uncomplicated Right-Sided Infective Endocarditis
3/6 (50%)
1/4 (25%)
25.0% (−51.6, 100.0)¶
- Complicated Right-Sided Infective Endocarditis
5/13 (39%)
6/12 (50%)
−11.5% (−62.4, 39.4)¶
- Left-Sided Infective Endocarditis
1/9 (11%)
2/9 (22%)
−11.1% (−55.9, 33.6)¶
Eighteen (18/120) patients in the daptomycin for injection arm and 19/116 patients in the comparator arm died during the trial. These comprise 3/28 daptomycin for injection-treated patients and 8/26 comparator-treated patients with endocarditis, as well as 15/92 daptomycin for injection treated patients and 11/90 comparator-treated patients with bacteremia. Among patients with persisting or relapsing S. aureus infections, 8/19 daptomycin for injection-treated patients and 7/11 comparator-treated patients died.
Overall, there was no difference in time to clearance of S. aureus bacteremia between daptomycin for injection and comparator. The median time to clearance in patients with MSSA was 4 days and in patients with MRSA was 8 days.
Failure of treatment due to persisting or relapsing S. aureus infections was assessed by the Adjudication Committee in 19/120 (16%) daptomycin for injection-treated patients (12 with MRSA and 7 with MSSA) and 11/115 (10%) comparator-treated patients (9 with MRSA treated with vancomycin and 2 with MSSA treated with an anti-staphylococcal semi-synthetic penicillin). Among all failures, isolates from 6 daptomycin for injection-treated patients and 1 vancomycin-treated patient developed increasing MICs (reduced susceptibility) by central laboratory testing during or following therapy. Most patients who failed due to persisting or relapsing S. aureus infection had deep-seated infection and did not receive necessary surgical intervention [see Warnings and Precautions (5.9)].
Pediatric Patients (1 to 17 Years of Age) with S. aureus Bacteremia
The pediatric S. aureus bacteremia study was designed as a prospective multi-center, randomized, comparative trial to treat pediatric patients aged 1 to 17 years with bacteremia. Patients known to have endocarditis or pneumonia at baseline were excluded. Patients were enrolled in a stepwise approach into three age groups and given age-dependent doses of daptomycin for injection once daily for up to 42 days. The different age groups and doses evaluated were as follows: Adolescents (12 to 17 years, n=14 patients) treated with daptomycin for injection dosed at 7 mg/kg once daily, Children (7 to 11 years, n=19 patients) treated with daptomycin for injection dosed at 9 mg/kg once daily and Children (2 to 6 years, n=22 patients) treated with daptomycin for injection dosed at 12 mg/kg once daily. No patients 1 to <2 years of age were enrolled.
Patients were randomized 2:1 to receive daptomycin for injection or a standard of care comparator, which included intravenous therapy with vancomycin, semi-synthetic penicillin, first generation cephalosporin or clindamycin. Patients could switch to oral therapy after clinical improvement was demonstrated (no minimum IV dosing was required).
The primary objective of this study was to assess the safety of daptomycin for injection. The clinical outcome was determined by resolution or improvement of symptoms at test-of-cure (TOC) visit, 7 to 14 days after the last dose, which was assessed by the site level Blinded Evaluator.
Of the 82 subjects randomized in the study, 81 subjects were treated with daptomycin for injection or comparator and included in the safety population, and 73 had a proven S. aureus bacteremia at Baseline. Of these, 51 subjects were randomized to the daptomycin for injection group and 22 subjects were randomized to the comparator group. The mean duration of IV therapy was 12 days, with a range of 1 to 44 days. Forty-eight subjects switched to oral therapy, and the mean duration of oral therapy was 21 days. The clinical success rates determined at 7 to 14 days after last dose of therapy (IV and oral) (TOC visit) were 88% (45/51) for daptomycin for injection and 77% (17/22) for comparator.
- 15 REFERENCES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
- DAPZURA RT (daptomycin for injection) is supplied as a sterile pale yellow to light brown lyophilized powder in a single-dose 10 mL vial containing 500 mg of daptomycin: Package of 1 (NDC: 60977-145-01). The vial stopper is not made with natural rubber latex.
- Store original packages at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Storage conditions for the reconstituted and diluted solutions are described in another section of the prescribing information [see Dosage and Administration (2.7)].
-
17 PATIENT COUNSELING INFORMATION
Allergic Reactions
Advise patients that allergic reactions, including serious skin, kidney, lung, or other organ reactions, could occur and that these serious reactions require immediate treatment. Patients should report any previous allergic reactions to daptomycin. [see Warnings and Precautions (5.1, 5.4, 5.5).]
Muscle Pain or Weakness (Myopathy and Rhabdomyolysis, Peripheral Neuropathy)
Advise patients to report muscle pain or weakness, especially in the forearms and lower legs, as well as tingling or numbness. [see Warnings and Precautions (5.2, 5.6).]
Cough, Breathlessness or Fever (Eosinophilic Pneumonia)
Advise patients to report any symptoms of cough, breathlessness, or fever. [see Warnings and Precautions (5.3).]
C. difficile-Associated Diarrhea (CDAD)
Advise patients that diarrhea is a common problem caused by antibacterials, including daptomycin for injection, that usually ends when the antibacterial is discontinued. Sometimes after starting treatment with antibacterials, including DAPZURA RT, patients can develop watery and bloody stools (with or without stomach cramps and fever), even as late as 2 or more months after having received the last dose of the antibacterial. If this occurs, patients should contact their physician as soon as possible. [see Warnings and Precautions (5.8).]
Patients with Hereditary Fructose Intolerance (HFI)
Inform patients and caregivers that DAPZURA RT contains sorbitol and can be life-threatening when administered to patients with hereditary fructose intolerance (HFI) [see Warnings and Precautions (5.11)]. Inquire for symptoms of sorbitol, fructose and/or sucrose intolerance.
Antibacterial Resistance
Patients should be counseled that antibacterial drugs, including DAPZURA RT, should be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When DAPZURA RT is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be administered exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by DAPZURA RT or other antibacterial drugs in the future.
Baxter Logo
Baxter Healthcare Corporation
Deerfield, IL 60015 USA
Made in the USA
Baxter and Dapzura RT are trademarks of Baxter International Inc.
0719004045
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
Barcode (FPO)
Use 10 mL Sterile or Bacte-
riostatic Water for Injection
for reconstitution only.NDC 60977-145-01
Rx OnlyStore at 20°C to 25°C (68°F
to 77°F). See package insert
for Usual Dosage and storage
of reconstituted and further
diluted product.
07-09-00-0427Baxter Healthcare Corporation
DAPZURA RT
(daptomycin for injection)
500 mg per vialFor Intravenous Use
Single-dose vial –
Discard Unused PortionBaxter Logo
Lot: (FPO)
Exp.: (FPO)For Intravenous Use
Rx Only
Single-dose vial – Discard Unused PortionBaxter Logo
2D Barcode Location
DAPZURA RT
(daptomycin for injection)
500 mg per vialNDC 60977-145-01
DAPZURA RT
(daptomycin for injection)
500 mg per vialFor Intravenous Use
Reconstitute viral only with
Sterile Water for Injection or
Bacteriostatic Water for Injection.Single-dose vial -
Discard Unused Portion1D Barcode
(01) 20360977145017Recommended Dosage: See Prescribing
Information.This package contains one single-dose
vial of sterile DAPZURA RT (daptomycin for
injection) and one package insert.
Store at 20°C to 25°C (68°F to 77°F);
excursions permitted to 15°C to 30°C
(59°F to 86°F) [see USP controlled Room
Temperature.] See package insert for
storage of reconstituted and further
diluted product.Not made with Natural rubber latex
Baxter Logo
Baxter Healthcare Corporation
Deerfield, IL 60015Made in the USA
2VR500
07-01-00-08943-6118-520
NDC 60977-145-01
DAPZURA RT
(daptomycin for injection)
500 mg per vialFor Intravenous Use
Reconstitute viral only with
Sterile Water for Injection or
Bacteriostatic Water for Injection.Single-dose vial -
Discard Unused PortionRx Only
Baxter Logo
FPO 2D Barcode location
See enclosed package insert for
reconstitution instructions and complete
information on dosage and administration.DAPZURA RT (daptomycin for injection)
contains 500 mg/vial of daptomycin.Reconstitute with 10 mL Sterile Water
for Injection or Bacteriostatic Water for
Injection to obtain a final concentration
of 50 mg/mL.Each vial also contains 238 mg sorbitol
and 238 mg of mannitol. Sodium
hydroxide and/or hydrochloric acid are
used to adjust pH.Contains no preservatives.
Note: Parenteral drug products should be
inspected visually for particulate matter
prior to administration. -
INGREDIENTS AND APPEARANCE
DAPZURA RT
daptomycin injection, powder, lyophilized, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60977-145 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DAPTOMYCIN (UNII: NWQ5N31VKK) (DAPTOMYCIN - UNII:NWQ5N31VKK) DAPTOMYCIN 500 mg in 10 mL Inactive Ingredients Ingredient Name Strength SORBITOL (UNII: 506T60A25R) 238 mg in 10 mL MANNITOL (UNII: 3OWL53L36A) 238 mg in 10 mL SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) Product Characteristics Color YELLOW (pale yellow to light brown) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60977-145-01 1 in 1 CARTON 01/25/2022 01/25/2022 1 10 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA213645 01/25/2022 Labeler - Baxter Healthcare Corporation (005083209) Establishment Name Address ID/FEI Business Operations Baxter Healthcare Corporation 194684502 ANALYSIS(60977-145) , LABEL(60977-145) , MANUFACTURE(60977-145) , PACK(60977-145) , STERILIZE(60977-145)
Trademark Results [DAPZURA RT]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 DAPZURA RT 90142495 not registered Live/Pending |
Baxter International Inc. 2020-08-27 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.