IOMERVU- iomeprol injection injection, solution
IOMERVU by
Drug Labeling and Warnings
IOMERVU by is a Prescription medication manufactured, distributed, or labeled by BRACCO DIAGNOSTICS INC, BIPSO GmbH, SPIN S.p.A., BioChem Labor für biologishe und chemische Analytik GmbH. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use IOMERVU safely and effectively. See full prescribing information for IOMERVU.
IOMERVUTM (iomeprol) injection, for intra-arterial or intravenous use
Initial U.S. Approval: 2024WARNING: RISKS ASSOCIATED WITH INTRATHECAL ADMINISTRATION
Intrathecal administration, even if inadvertent, may cause death, convulsions, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, seizures, rhabdomyolysis, hyperthermia, and brain edema. IOMERVU is for intra-arterial or intravenous use only. (2.1, 5.1)
INDICATIONS AND USAGE
IOMERVU is a radiographic contrast agent indicated for:
Intra-arterial Procedures† (1.1)- Cerebral arteriography, includingintra-arterial digital subtraction angiography (IA-DSA), in adults and pediatric patients
- Visceral and peripheral arteriography and aortography, including IA-DSA, in adults and pediatric patients
- Coronary arteriography and cardiac ventriculography in adults
- Radiographic evaluation of cardiac chambers and related arteries in pediatric patients
Intravenous Procedures† (1.2)
- Computed tomography (CT) of the head and body in adults and pediatric patients
- CT angiography of intracranial, visceral, and lower extremity arteries in adults and pediatric patients
- Coronary CT angiography in adults and pediatric patients
- CT urography in adults and pediatric patients
†Specific concentrations are recommended for each type of imaging procedure. (2.2, 2.3, 2.4, 2.5)
DOSAGE AND ADMINISTRATION
- Individualize the volume and concentration according to the specific dosing tables accounting for factors such as age, body weight, vessel size, rate of blood flow within the vessel, and structures or areas to be examined. (2.2, 2.3, 2.4, 2.5)
- See full prescribing information for complete dosage and administration information. (2)
DOSAGE FORMS AND STRENGTHS
Injection: 250 mg Iodine/mL, 300 mg Iodine/mL, 350 mg Iodine/mL, and 400 mg Iodine/mL in single-dose vials or bottles (3)
CONTRAINDICATIONS
None (4)
WARNINGS AND PRECAUTIONS
- Hypersensitivity Reactions: Life-threatening or fatal reactions can occur. Always have emergency resuscitation equipment and trained personnel available. (5.2)
- Acute Kidney Injury: Acute injury including renal failure can occur. Minimize dose and maintain adequate hydration to minimize risk. (5.3)
- Cardiovascular Adverse Reactions: Hemodynamic disturbances including shock and cardiac arrest may occur during or after administration. (5.4)
- Thyroid Dysfunction in Pediatric Patients 0 Years to 3 Years of Age: Individualize thyroid function monitoring based on risk factors such as prematurity. (5.8)
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥0.5%) are feeling hot, headache, nausea, chest pain, back pain, and vomiting. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Bracco Diagnostics Inc. at 1-800-257-5181 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
Lactation: A lactating woman may pump and discard breast milk for 10 hours after IOMERVU administration. (8.2)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 11/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: RISKS ASSOCIATED WITH INTRATHECAL ADMINISTRATION
1 INDICATIONS AND USAGE
1.1 Intra-arterial Procedures†
1.2 Intravenous Procedures†
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosing and Administration Information
2.2 Recommended Dosage for Intra-arterial Procedures in Adults
2.3 Recommended Dosage for Intra-arterial Procedures in Pediatric Patients
2.4 Recommended Dosage for Intravenous Procedures in Adults
2.5 Recommended Dosage for Intravenous Procedures in Pediatric Patients
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Risks Associated with Inadvertent Intrathecal Administration
5.2 Hypersensitivity Reactions
5.3 Acute Kidney Injury
5.4 Cardiovascular Adverse Reactions
5.5 Thromboembolic Events
5.6 Extravasation and Injection Site Reactions
5.7 Thyroid Storm in Patients with Hyperthyroidism
5.8 Thyroid Dysfunction in Pediatric Patients 0 Years to 3 Years of Age
5.9 Hypertensive Crisis in Patients with Pheochromocytoma
5.10 Sickle Cell Crisis in Patients with Sickle Cell Disease
5.11 Severe Cutaneous Adverse Reactions
5.12 Interference with Laboratory Tests
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Post Marketing Experience
7 DRUG INTERACTIONS
7.1 Drug-Drug Interactions
7.2 Drug-Laboratory Test Interactions
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Intra-arterial Studies
14.2 Intravenous Studies
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: RISKS ASSOCIATED WITH INTRATHECAL ADMINISTRATION
Intrathecal administration, even if inadvertent, may cause death, convulsions, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, seizures, rhabdomyolysis, hyperthermia, and brain edema. IOMERVU is for intra-arterial or intravenous use only [see Dosage and Administration (2.1) and Warnings and Precautions (5.1)]. -
1 INDICATIONS AND USAGE
1.1 Intra-arterial Procedures†
- Cerebral arteriography, including intra-arterial digital subtraction angiography (IA-DSA), in adults and pediatric patients
- Visceral and peripheral arteriography and aortography, including IA-DSA, in adults and pediatric patients
- Coronary arteriography and cardiac ventriculography in adults
- Radiographic evaluation of cardiac chambers and related arteries in pediatric patients
1.2 Intravenous Procedures†
- Computed tomography (CT) of the head and body in adults and pediatric patients
- CT angiography of intracranial, visceral, and lower extremity arteries in adults and pediatric patients
- Coronary CT angiography in adults and pediatric patients
- CT urography in adults and pediatric patients
†Specific concentrations of IOMERVU are recommended for each type of imaging procedure [see Dosage and Administration (2.2, 2.3, 2.4, 2.5)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosing and Administration Information
- IOMERVU is for intra-arterial or intravenous use only and must not be administered intrathecally [see Warnings and Precautions (5.1)].
- Specific concentrations of IOMERVU are recommended for each type of imaging procedure [see Dosage and Administration (2.2, 2.3, 2.4, 2.5)].
- Individualize the volume, concentration, and injection rate of IOMERVU within the specified ranges [see Dosage and Administration (2.2, 2.3, 2.4, 2.5)]. Consider factors such as age, body weight, vessel size, rate of blood flow within the vessel, anticipated pathology, degree and extent of opacification required, structures or area to be examined, disease processes affecting the patient, and equipment and technique to be employed.
- Hydrate patients before and after IOMERVU administration [see Warnings and Precautions (5.3)].
- Use aseptic technique for all handling and administration of IOMERVU.
- IOMERVU may be administered at either body temperature (37°C, 98.6°F) or room temperature (20°C to 25°C, 68°F to 77°F).
- Visually inspect IOMERVU for particulate matter or discoloration before administration, whenever the solution and container permit. Do not administer IOMERVU if particulate matter or discoloration is observed.
- Do not mix IOMERVU with, or inject in intravenous lines containing, other drugs or total nutritional admixtures.
- Each single-dose container of IOMERVU injection is intended for one procedure only. Discard any unused portion.
2.2 Recommended Dosage for Intra-arterial Procedures in Adults
- Recommended doses of IOMERVU in adults for intra-arterial procedures are shown in Table 1.
- Inject at rates approximately equal to the flow rate in the vessel being injected.
Table 1. Recommended Concentrations and Volumes of IOMERVU to Administer per Single Injection into Selected Arteries for Intra-arterial Procedures in Adults Imaging Procedure Concentration (mg Iodine/mL) Volume
(mL)Maximum Total Dose (mL) Cerebral arteriography 300 - Carotid, subclavian, and vertebral arteries: 6 mL to 12 mL
- Aortic arch: 30 mL to 50 mL
200 mL Visceral and peripheral arteriography; aortography 300 - Aortography: 30 mL to 70 mL
- Renal arteries: 10 mL to 12 mL
- Other major branches of aorta: 20 mL to 60 mL
200 mL Intra-arterial digital subtraction angiography 300 - Carotid, subclavian, and vertebral arteries: 4 mL to 12 mL
- Aortic arch: 20 mL to 25 mL
- Aortography: 15 mL to 40 mL
- Renal arteries: 6 mL to 16 mL
- Other major branches of aorta: 10 mL to 40 mL
- Ilio-femoral runoff: 8 mL to 40 mL
200 mL Coronary arteriography and cardiac ventriculography 300 - Coronary arteries: 3 mL to 7 mL
- Cardiac ventriculography: 30 mL to 45 mL
286 mL 350 245 mL 400 215 mL 2.3 Recommended Dosage for Intra-arterial Procedures in Pediatric Patients
- Recommended doses of IOMERVU in pediatric patients for intra-arterial procedures are shown in Table 2.
- Inject at rates approximately equal to the flow rate in the vessel being injected.
Table 2. Recommended Concentrations and Volumes per Body Weight of IOMERVU to Administer per Single Injection for Intra-arterial Procedures in Pediatric Patients Imaging Procedure Concentration (mg Iodine/mL) Volume
(mL/kg body weight)Maximum Total Dose (mL/kg) Cerebral arteriography 300 0.5 mL/kg to
2 mL/kg- 5 mL/kg
- Do not exceed adult maximum dose (see Table 1)
Visceral and peripheral arteriography; aortography 300 0.5 mL/kg to
2 mL/kgIntra-arterial digital subtraction angiography 300 0.3 mL/kg to
1 mL/kgRadiographic evaluation of cardiac chambers and related arteries 300, 350, or 400 0.5 mL/kg to
2 mL/kg2.4 Recommended Dosage for Intravenous Procedures in Adults
Recommended doses of IOMERVU in adults for intravenous procedures are shown in Table 3.
Table 3. Recommended Concentrations, Volumes, and Injection Rates of IOMERVU for Intravenous Procedures in Adults Imaging Procedure Concentration
(mg Iodine/mL)Volume
(mL)Injection Rate3 (mL/s) CT of Head and Body 250 or 300 100 mL to 190 mL 2 mL/s to 4 mL/s 350 or 400 75 mL to 150 mL CT Angiography1 300, 350, or 400 80 mL to 130 mL 4 mL/s to 6 mL/s Coronary CT Angiography1 400 50 mL to 90 mL 4 mL/s to 6 mL/s CT Urography2 350 90 mL to 120 mL 2.5 mL/s 1 The IOMERVU volume may be immediately followed by a 40 mL to 50 mL 0.9% sodium chloride injection flush at the same flow rate as the contrast volume.
2 The IOMERVU volume may be administered either as a single bolus, or for dual-phase protocols as divided doses.
3 The injection rate of IOMERVU should be determined according to the clinical indication and the location, size, and type of the intravenous access.2.5 Recommended Dosage for Intravenous Procedures in Pediatric Patients
Recommended doses of IOMERVU in pediatric patients for intravenous procedures are shown in Table 4.
Table 4. Recommended Concentrations, Volumes per Body Weight, and Injection Rates of IOMERVU for Intravenous Procedures in Pediatric Patients Imaging Procedure Concentration (mg Iodine/mL) Volume
(mL/kg body weight)Injection Rate (mL/s)* CT of Head and Body 250 or 300 1.5 mL/kg to 2.5 mL/kg 1 mL/s to 2 mL/s 350 or 400 1 mL/kg to 2 mL/kg CT Angiography 300, 350, or 400 1 mL/kg to 2 mL/kg 2 mL/s to 3 mL/s Coronary CT Angiography 300 or 400 1 mL/kg to 2 mL/kg 2 mL/s to 3 mL/s CT Urography 300 1 mL/kg to 2 mL/kg 1 mL/s to 2 mL/s * The injection rate of IOMERVU should be determined according to the clinical indication and the location, size, and type of the intravenous access. In neonates and patients <15 kg in whom a 24-gauge angiocatheter is the only option, an injection rate of 1 mL/s is recommended.
-
3 DOSAGE FORMS AND STRENGTHS
Injection: clear, colorless to pale yellow solution available in the following iodine concentrations and configurations:
Concentration
(mg Iodine/mL)Package Size Package Type 250 100 mL Single-dose bottles 300 50 mL Single-dose vials 100 mL, 150 mL, and 200 mL Single-dose bottles 350 50 mL Single-dose vials 100 mL, 150 mL, and 200 mL Single-dose bottles 400 50 mL Single-dose vials 100 mL, 150 mL, and 200 mL Single-dose bottles - 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Risks Associated with Inadvertent Intrathecal Administration
IOMERVU is for intra-arterial or intravenous use only and must not be administered intrathecally [see Dosage and Administration (2.1)]. Intrathecal administration, even if inadvertent, can cause death, convulsions, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, seizures, rhabdomyolysis, hyperthermia, and brain edema.
5.2 Hypersensitivity Reactions
IOMERVU can cause life-threatening or fatal hypersensitivity reactions including anaphylaxis. Manifestations include respiratory arrest, laryngospasm, bronchospasm, angioedema, and shock [see Adverse Reactions (6.2)]. Most severe reactions develop shortly after the start of injection (e.g., within 1 to 3 minutes), but delayed reactions can occur. There is an increased risk in patients with a history of a previous reaction to contrast agents, and known allergic disorders (i.e., bronchial asthma, drug, or food allergies) or other hypersensitivities. Premedication with antihistamines or corticosteroids to minimize possible allergic reactions does not prevent serious life-threatening reactions, but may reduce their incidence and severity.
Obtain a history of allergy, hypersensitivity, and hypersensitivity reactions to iodinated contrast agents. Always have emergency resuscitation equipment and trained personnel available before use of IOMERVU. Monitor all patients for hypersensitivity reactions.
5.3 Acute Kidney Injury
Acute kidney injury, including renal failure, may occur after IOMERVU administration. Risk factors include pre-existing renal impairment, dehydration, diabetes mellitus, heart failure, advanced vascular disease, advanced age, concomitant use of nephrotoxic or diuretic medications, multiple myeloma or other paraproteinemia, and repetitive or large doses of IOMERVU.
Use the lowest necessary dose of IOMERVU in patients with renal impairment. Adequately hydrate patients prior to and following IOMERVU administration. Do not use laxatives, diuretics, or preparatory dehydration prior to IOMERVU administration.
5.4 Cardiovascular Adverse Reactions
IOMERVU increases the circulatory osmotic load and may induce acute or delayed hemodynamic disturbances in patients with heart failure, severely impaired renal function, combined renal and hepatic disease, or combined renal and cardiac disease, particularly when repetitive or large doses are administered.
Life-threatening or fatal cardiovascular reactions including hypotension, shock, and cardiac arrest have occurred with the use of IOMERVU. Most deaths occur within 10 minutes of injection, with cardiovascular disease as the main underlying factor. Cardiac decompensation, serious arrhythmias, and myocardial ischemia or infarction can occur during coronary arteriography and ventriculography.
Based upon literature reports, deaths from the administration of iodinated contrast agents range from 6.6 per 1 million (0.00066 percent) to 1 in 10,000 patients (0.01 percent). Use the lowest necessary dose of IOMERVU in patients with heart failure and always have emergency resuscitation equipment and trained personnel available. Monitor all patients for severe cardiovascular reactions.
5.5 Thromboembolic Events
Serious, in some cases fatal, thromboembolic events including myocardial infarction and stroke can occur during angiographic procedures with iodinated contrast agents including IOMERVU. During these procedures, increased thrombosis and activation of the complement system can occur. Risk factors for developing thromboembolic events include length of procedure, catheter and syringe material, underlying disease state, and concomitant medications.
To reduce risk of thromboembolic events, use meticulous angiographic techniques and minimize the length of the procedure. Avoid blood remaining in contact with syringes containing IOMERVU, which increases the risk of clotting. Avoid angiocardiography in patients with homocystinuria because of the risk of inducing thrombosis and embolism.
5.6 Extravasation and Injection Site Reactions
Extravasation can occur with IOMERVU administration, particularly in patients with severe arterial or venous disease. Inflammation, blistering, skin necrosis, and compartment syndrome have been reported following extravasation. In addition, injection site reactions such as pain and swelling at the injection site can also occur [see Adverse Reactions (6.1, 6.2)]. Ensure intravascular placement of catheters prior to injection. Monitor patients for extravasation and advise patients to seek medical care for progression of symptoms.
5.7 Thyroid Storm in Patients with Hyperthyroidism
Thyroid storm has occurred after the intravascular use of iodinated contrast agents in patients with hyperthyroidism or with an autonomously functioning thyroid nodule. Evaluate the risk in such patients before use of IOMERVU.
5.8 Thyroid Dysfunction in Pediatric Patients 0 Years to 3 Years of Age
Thyroid dysfunction characterized by hypothyroidism or transient thyroid suppression has been reported after both single exposure and multiple exposures to iodinated contrast agents in pediatric patients 0 years to 3 years of age.
Younger age, very low birth weight, prematurity, underlying medical conditions affecting thyroid function, admission to neonatal or pediatric intensive care units, and congenital cardiac conditions are associated with an increased risk of hypothyroidism after iodinated contrast agent exposure. Pediatric patients with congenital cardiac conditions may be at greatest risk given that they often require high doses of contrast during invasive cardiac procedures.
An underactive thyroid during early life may be harmful for cognitive and neurological development and may require thyroid hormone replacement therapy. After exposure to IOMERVU, individualize thyroid function monitoring based on underlying risk factors, especially in term and preterm neonates.
5.9 Hypertensive Crisis in Patients with Pheochromocytoma
Hypertensive crisis has occurred after the use of iodinated contrast agents in patients with pheochromocytoma. Closely monitor patients when administering IOMERVU if pheochromocytoma or catecholamine-secreting paragangliomas are suspected. Inject the minimum amount of IOMERVU necessary, assess blood pressure throughout the procedure, and have measures for treatment of a hypertensive crisis readily available.
5.10 Sickle Cell Crisis in Patients with Sickle Cell Disease
Iodinated contrast agents when administered intravascularly may promote sickling in individuals who are homozygous for sickle cell disease. Hydrate patients prior to and following IOMERVU administration and use IOMERVU only if the necessary imaging information cannot be obtained with alternative imaging modalities.
5.11 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCAR) may develop from 1 hour to several weeks after intravascular contrast agent administration. These reactions include Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP), and drug reaction with eosinophilia and systemic symptoms (DRESS). Reaction severity may increase and time to onset may decrease with repeat administration of a contrast agent; prophylactic medications may not prevent or mitigate severe cutaneous adverse reactions. Avoid administering IOMERVU to patients with a history of a severe cutaneous adverse reaction to IOMERVU.
5.12 Interference with Laboratory Tests
IOMERVU can interfere with protein-bound iodine tests [see Drug Interactions (7.2)].
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Risks Associated with Intrathecal Administration [see Warnings and Precautions (5.1)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
- Acute Kidney Injury [see Warnings and Precautions (5.3)]
- Cardiovascular Adverse Reactions [see Warnings and Precautions (5.4)]
- Thromboembolic Events [see Warnings and Precautions (5.5)]
- Extravasation and Injection Site Reactions [see Warnings and Precautions (5.6)]
- Thyroid Dysfunction in Pediatric Patients 0 Years to 3 Years of Age [see Warnings and Precautions (5.8)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.11)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse Reactions in Adult Patients
The safety of IOMERVU was evaluated in 4,621 adult patients who received 1,500 mg to 86,000 mg iodine doses of IOMERVU intra-arterially or intravenously in clinical trials. The average age was 60 years (range 18 years to 99 years), and 34% were female. The racial and ethnic distribution was 83% White, 10% Asian, 1% Black, 1% Hispanic, and 5% patients of other or unspecified groups.
Table 5 provides a summary of the adverse reactions reported in ≥0.5% of adult patients.
Table 5: Adverse Reactions Reported in ≥0.5% of 4,621 Adult Patients Receiving Intra-arterial or Intravenous Administration of IOMERVU in Clinical Trials Adverse Reaction Incidence (%) Feeling hot 2 Headache 1.2 Nausea 1 Chest pain 0.6 Back pain 0.5 Vomiting 0.5 The following adverse reactions were observed in <0.5% of the adult patients receiving IOMERVU:
Blood and lymphatic system disorders: activated partial thromboplastin time prolonged, prothrombin time prolonged
Cardiovascular disorders: ventricular fibrillation, hypertensive crisis, coronary arteriospasm, congestive cardiac failure, cardiac flutter, atrioventricular block, right bundle branch block, hypotension, arrhythmia, bradycardia, supraventricular extrasystoles, ventricular extrasystoles, increased blood pressure, flushing
Ear and labyrinth disorders: vertigo, ear discomfort
Eye disorder: vision blurred, periorbital edema, photopsia
Gastrointestinal: esophageal varices hemorrhage, abdominal pain, abdominal distension, alanine aminotransferase (ALT) increased, constipation, diarrhea, dry mouth, salivary hypersecretion, oral paresthesia
General disorders: pain, injection site reactions (pain, discomfort, or warmth), peripheral edema, chills, asthenia, malaise
Musculoskeletal and connective tissue disorders: arthralgia, pain in jaw
Nervous system disorders: cerebrovascular disorder, dysarthria, visual field defect, burning sensation, presyncope, dizziness, dysgeusia, paresthesia
Psychiatric disorders: delirium, anxiety, insomnia
Renal and urinary disorders: acute kidney injury, increased blood creatinine, urinary urgency
Respiratory, thoracic, and mediastinal disorders: respiratory arrest, pulmonary edema, bronchospasm, dyspnea, cough, rhinitis, throat irritation or tightness, sneezing
Skin and subcutaneous tissue disorders: urticaria, blister, purpura, ecchymosis, rash, pruritus, erythema
Adverse Reactions in Pediatric Patients
The safety of IOMERVU was evaluated in 184 pediatric patients who received 1,800 mg to 76,000 mg iodine doses of IOMERVU intra-arterially or intravenously in clinical trials. The average age was 6 years (range 11 days to 17 years), and 47% were female. The racial distribution was 98% White, 1% Black, and 1% patients of other or unspecified groups. The overall character, quality, and severity of adverse reactions reported in pediatric patients were similar to those reported in adult patients.
6.2 Post Marketing Experience
The following adverse reactions have been identified during post-approval use of IOMERVU outside the United States. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: thrombocytopenia, leukopenia, leukocytosis
Cardiovascular disorders: cardio-respiratory arrest, circulatory collapse or shock, myocardial infarction, atrial fibrillation, cyanosis, pallor
Endocrine disorders: hyperthyroidism, hypothyroidism
Eye disorders: transient blindness, conjunctivitis, increased lacrimation
Gastrointestinal disorders: salivary gland enlargement, increased aspartate aminotransferase (AST), dysphagia
General disorders and administration site conditions:injection site swelling (usually due to extravasation)
Immune system disorders: hypersensitivity reactions including fatal anaphylaxis
Nervous system disorders: coma, loss of consciousness, encephalopathy, transient ischemic attack, paralysis, convulsion, syncope, amnesia, somnolence
Psychiatric disorders: confusional state
Respiratory, thoracic, and mediastinal disorders: acute respiratory distress syndrome (ARDS), laryngeal edema, pharyngeal edema, dysphonia
Skin and subcutaneous tissue disorders: severe reactions (Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms (DRESS)) and mild reactions (rash, erythema, pruritus, urticaria, and skin discoloration)
-
7 DRUG INTERACTIONS
7.1 Drug-Drug Interactions
Stop metformin at the time of, or prior to, IOMERVU administration in patients with an eGFR between 30 and 60 mL/min/1.73 m2; in patients with a history of hepatic impairment, alcoholism, or heart failure; or in patients who will be administered intra-arterial iodinated contrast agents. Re-evaluate eGFR 48 hours after the imaging procedure, and reinstitute metformin only after renal function is stable.
Metformin can cause lactic acidosis in patients with renal impairment. Iodinated contrast agents appear to increase the risk of metformin-induced lactic acidosis, possibly as a result of worsening renal function.
Avoid thyroid therapy or testing using radioactive iodine for up to 6 weeks post IOMERVU. Administration of IOMERVU may interfere with thyroid uptake of radioactive iodine (I-131 and I-123) and decrease therapeutic and diagnostic efficacy.
7.2 Drug-Laboratory Test Interactions
Do not perform a protein-bound iodine test for at least 16 days following administration of IOMERVU.
Iodinated contrast agents, including IOMERVU, will temporarily increase protein-bound iodine in blood. However, thyroid function tests that do not depend on iodine estimations, e.g., triiodothyronine (T3) resin uptake and total or free thyroxine (T4) assays, are not affected.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Available data from literature and postmarketing reports on iomeprol use in pregnant women over decades have not identified a drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes. In animal reproduction studies, no adverse developmental outcomes were observed with intravenous administration of iomeprol to pregnant rats and rabbits at doses up to 0.45-times the maximum recommended human dose of 86,000 mg iodine. Animal studies show that iomeprol crosses the placenta (see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defects, loss or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Embryo-fetal developmental toxicity studies were performed with intravenous administration of iomeprol in rats at daily doses of 600 mg, 1,500 mg, or 4,000 mg iodine/kg (0.07-, 0.17- or 0.45-fold the human equivalent dose (HED, mg/m2) using the maximum human dose of 86,000 mg iodine per administration) from gestation days (GD) 6 to 15 and in rabbits at daily doses of 300 mg, 800 mg, or 2,000 mg iodine/kg (0.07-, 0.18- or 0.45-fold the HED using the maximum human dose of 86,000 mg iodine per administration) from GD 6 to 18. Iomeprol did not result in fetal harm at the highest doses evaluated, 0.45-times the maximum recommended human dose of 86,000 mg iodine.
A biodistribution study with a single intravenous administration of 1,000 mg iodine/kg (0.11-fold the HED using the maximum human dose of 86,000 mg iodine per administration) of radiolabeled iomeprol to pregnant rats showed that iomeprol crosses the placenta. No accumulation of radioactivity in fetal tissues was observed.
8.2 Lactation
There are no data on the presence of iomeprol in human milk, the effects on the breastfed infant, or the effects on milk production. Iomeprol is present in animal milk (see Data). When a drug is present in animal milk, it is likely that the drug will be present in human milk. Iodinated contrast agents are excreted unchanged in human milk in very low amounts, with poor absorption from the gastrointestinal tract of a breastfed infant. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for IOMERVU and any potential adverse effects on the breastfed infant from IOMERVU or from the underlying maternal condition.
Interruption of breastfeeding after exposure to iodinated contrast agents is not necessary because the potential exposure of the breastfed infant to iodine is small. However, a lactating woman may consider interrupting breastfeeding and pumping and discarding breast milk for 10 hours (approximately 5 elimination half-lives) after IOMERVU administration to minimize any potential drug exposure to a breastfed infant.
An animal study with a single intravenous administration of 500 mg iodine/kg (0.06-fold the HED using the maximum human dose of 86,000 mg iodine per administration) of radiolabeled iomeprol to lactating rats showed that iomeprol rapidly distributed into the milk.
8.4 Pediatric Use
The safety and effectiveness of IOMERVU have been established in pediatric patients for cerebral, visceral, and peripheral arteriography, aortography including intra-arterial digital subtraction angiography, and radiographic evaluation of cardiac chambers and related arteries.
Use of IOMERVU is supported by evidence from adequate and well-controlled studies of IOMERVU in adults, pharmacokinetic data in pediatric patients aged 3 years to 17 years, pharmacokinetic simulation in pediatric patients younger than 3 years of age, and safety data obtained in 44 pediatric patients including 29 who were < 2 years of age, 14 children (2 years to 11 years), and 1 adolescent (12 years to 17 years). In general, adverse reactions reported in pediatric patients were similar to those in adults [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)].
The safety and effectiveness of IOMERVU have been established in pediatric patients for CT of the head and body, CT angiography of intracranial, visceral, and lower extremity arteries, coronary CT angiography, and CT urography.
Use of IOMERVU is supported by evidence from adequate and well-controlled studies of IOMERVU in adults, pharmacokinetic data in pediatric patients aged 3 years to 17 years, pharmacokinetic simulation in pediatric patients younger than 3 years of age, and safety data obtained in 140 pediatric patients including 22 who were < 2 years of age, 92 children (2 years to 11 years), and 26 adolescents (12 years to 17 years). In general, adverse reactions reported in pediatric patients were similar to those in adults [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.2)].
Intra-arterial and Intravenous Use
Pediatric patients at higher risk of experiencing an adverse reaction during and after administration of any contrast agent may include those with asthma, sensitivity to medication and/or allergens, cyanotic and acyanotic heart disease, heart failure, or serum creatinine greater than 1.5 mg/dL, or those less than 12 months of age. Pediatric patients with immature renal function or dehydration may be at increased risk for adverse events due to slower elimination of iodinated contrast agents [See Clinical Pharmacology (12.3)].
Thyroid function tests indicative of thyroid dysfunction, characterized by hypothyroidism or transient thyroid suppression have been reported following iodinated contrast agent administration in pediatric patients, including term and preterm neonates. Some patients were treated for hypothyroidism. After exposure to iodinated contrast agents, individualize thyroid function monitoring in pediatric patients 0 years to 3 years of age based on underlying risk factors, especially in term and preterm neonates [See Warnings and Precautions (5.8) and Adverse Reactions (6.2)].
8.5 Geriatric Use
Of the total number of subjects in clinical studies of IOMERVU, 1,977 (43%) patients were 65 years and older, while 629 (14%) patients were 75 years and older. No overall differences in safety or effectiveness were observed between these patients and younger patients.
Iomeprol is excreted by the kidneys, and the risk of adverse reactions to IOMERVU is greater in patients with renal impairment. Because elderly patients are more likely to have renal impairment, care should be taken in dose selection, and it may be useful to monitor renal function [see Warnings and Precautions (5.3) and Use in Specific Populations (8.6)].
-
10 OVERDOSAGE
The adverse effects of overdosage are life-threatening and affect mainly the pulmonary and cardiovascular systems. Treatment of an overdosage is directed toward the support of all vital functions and prompt institution of symptomatic therapy. Iomeprol can be removed by dialysis [see Clinical Pharmacology (12.3)].
-
11 DESCRIPTION
IOMERVU (iomeprol) injection is a tri-iodinated, non-ionic radiographic contrast agent for intra-arterial or intravenous use. It is provided as a sterile, nonpyrogenic, clear, colorless to pale yellow solution.
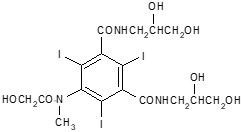
The chemical name for iomeprol is N,N’-bis(2,3-dihydroxypropyl)-5-[(hydroxyacetyl)-methylamino]-2,4,6-tri-iodo-1,3-benzenedicarboxamide. Iomeprol has a molecular formula of C17H22I3N3O8, a molecular weight of 777.09 (iodine content of 49%), and the following structural formula:
IOMERVU injection is available in four iodine concentrations:
- IOMERVU 250 mg Iodine/mL: Each mL contains 510 mg iomeprol (providing 250 mg bound iodine) and 1 mg tromethamine.
- IOMERVU 300 mg Iodine/mL: Each mL contains 612 mg iomeprol (providing 300 mg bound iodine) and 1 mg tromethamine.
- IOMERVU 350 mg Iodine/mL: Each mL contains 714 mg iomeprol (providing 350 mg bound iodine) and 1 mg tromethamine.
- IOMERVU 400 mg Iodine/mL: Each mL contains 816 mg iomeprol (providing 400 mg bound iodine) and 1 mg tromethamine.
The pH of IOMERVU has been adjusted to 6.5 to 7.2 with hydrochloric acid.
Physical characteristics are noted in Table 6. IOMERVU has osmolalities approximately 1.5 to 2.5 times that of plasma (285 mOsm/kg water) as shown in the table below and are hypertonic under conditions of use.
Table 6. Physical Characteristics of IOMERVU Concentration
(mg Iodine/mL)Density
(d204 ± 0.0002)Osmolality
(mOsmol/kg)Viscosity
(mPa·s)37°C 20°C 37°C 250 1.278 435 4.9 2.9 300 1.334 521 8.1 4.5 350 1.390 618 14.5 7.5 400 1.446 726 27.5 12.6 -
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Iomeprol is a radiographic iodinated contrast agent that opacifies the vessels and body structures where the contrast agent is present following intravenous or intra-arterial administration, permitting radiographic visualization of the internal structures through attenuation of X-ray photons.
In imaging of the body, iodinated contrast agents diffuse from the vessels into the extravascular space. In normal brain with an intact blood-brain barrier, the contrast agent does not diffuse into the extravascular space and contrast enhancement is generally due to the presence of contrast within the vascular space. In patients with a disrupted blood-brain barrier, the contrast agent accumulates in the extravascular space in the region of disruption.
12.2 Pharmacodynamics
The degree of radiographic enhancement by iomeprol is related to the iodine concentration in the tissue of interest following the administration of IOMERVU. However, the exposure-response relationships and time course of pharmacodynamic response of iomeprol have not been fully characterized.
12.3 Pharmacokinetics
The pharmacokinetic parameters of iomeprol are presented as mean (standard deviation, SD) unless otherwise specified.
The maximum concentration (Cmax) and area under the concentration-time curve (AUC) are dose-proportional across the dose range of 250 mg iodine/kg to 1,250 mg iodine/kg body weight.
Distribution
The volume of distribution of iomeprol is 0.28 (0.05) L/kg. Iomeprol does not bind to plasma proteins.Elimination
The elimination half-life of iomeprol is 1.8 (0.33) hours and the total body clearance is 0.10 (0.01) L/hr/kg.Metabolism
Iomeprol does not undergo significant metabolism.Excretion
Approximately 90% of the iomeprol dose is excreted unchanged in urine within 24 hours.No clinically significant differences in the pharmacokinetics of iomeprol were observed in pediatric patients aged 3 years to 17 years compared to adult patients who received IOMERVU. No clinically significant differences in Cmax and concentration of iomeprol within 5 minutes after IOMERVU administration between pediatric patients younger than 3 years of age and adults are expected based on pharmacokinetic simulations.
Patients with Renal Impairment
The renal clearance of iomeprol decreased by 28% in patients with mild (GFR 51 − 75 mL/min, estimated by inulin clearance (CLinulin)), 66% with moderate (GFR 26 − 50 mL/min, by CLinulin), and 84% with severe (GFR ≤ 25 mL/min, by CLinulin) renal impairment. Similarly, mean half-life increased 1.6-fold in mild, 2.9-fold in moderate, and 6.4-fold in severe renal impairment.Iomeprol is dialyzable. Iomeprol plasma concentrations decreased by 83% in patients with severe renal impairment who underwent hemodialysis 2 hours after a single administration of a 20,000 mg iodine dose of IOMERVU by intravenous route.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
No carcinogenicity studies of iomeprol have been conducted.Mutagenesis
Iomeprol did not demonstrate mutagenic or clastogenic potential in an in vitro bacterial reverse mutation assay (Ames test) or in an in vivo rat bone marrow micronucleus assay.Iomeprol did not impair the fertility of male or female rats when intravenously administered at doses up to 0.45-times the maximum recommended human dose.
-
14 CLINICAL STUDIES
14.1 Intra-arterial Studies
Cerebral arteriography was evaluated in one blinded read study incorporating two prospective, randomized, double-blind, multi-center clinical studies of 61 adult patients (35 male, 26 female) who were administered IOMERVU 300 mg Iodine/mL by intra-arterial route. The mean age was 52 years (range 17 years to 86 years), and 23% of patients were ≥65 years old. The mean total iodine dose administered was 29,000 mg. The racial and ethnic representations were 62% White, 15% Black, 20% Hispanic, and 3% Asian. Visualization was independently assessed as adequate or inadequate by three blinded readers. Visualization was rated as adequate in 100% of patients.
Visceral and peripheral arteriography were evaluated in one blinded read study incorporating two prospective, randomized, double-blind, multi-center clinical studies of 60 adult patients (36 male, 24 female) who were administered IOMERVU 300 mg Iodine/mL by intra-arterial route. The mean age was 67 years (range 29 years to 95 years) and 62% of patients were ≥65 years old. The mean total iodine dose administered was 48,000 mg. The racial and ethnic representations were 92% White, 7% Black, and 1% Hispanic. Visualization was independently assessed as adequate or inadequate by three blinded readers. Visualization was rated as adequate in 98% of patients.
Similar cerebral arteriography, visceral arteriography, and peripheral arteriography studies with digital subtraction angiography (DSA) were completed with comparable findings.
Coronary arteriography and cardiac ventriculography were evaluated in one blinded read study incorporating four prospective, randomized, double-blind, multi-center clinical studies of 59 adult patients (41 male, 18 female) who were administered IOMERVU 400 mg Iodine/mL, and 59 adult patients (43 male, 16 female) who were administered IOMERVU 300 mg Iodine/mL by intra-arterial route. The mean age was 60 years (range 22 years to 85 years), and 42% of patients were ≥65 years old. The mean total iodine dose administered was 45,000 mg. The racial and ethnic representations were 75% White, 15% Black, 6% Hispanic, 1% Asian, and 3% other racial or ethnic groups. Visualization was independently assessed as adequate or inadequate by three blinded readers. Visualization was rated as adequate in 93% - 100% of patients for both concentrations.
14.2 Intravenous Studies
CT of the head and body was evaluated in one blinded read study incorporating four prospective, randomized, double-blind, multi-center clinical studies of 59 adult patients (22 male, 37 female) who were administered IOMERVU 400 mg Iodine/mL and 59 adult patients (26 male, 33 female) who were administered IOMERVU 250 mg Iodine/mL by intravenous route. The mean age was 55 years (range 19 years to 80 years), and 30% of patients were ≥65 years old. The mean total iodine dose administered was 41,000 mg. The racial and ethnic representations were 78% White, 16% Black, 4% Hispanic, 1% Asian, and 1% other racial or ethnic groups. Visualization was independently assessed as adequate or inadequate by three blinded readers. Visualization was rated as adequate in 98% - 100% of patients for both concentrations.
CT angiography was evaluated in two prospective, single-center clinical studies enrolling a total of 262 adult patients (202 male, 60 female) with suspected or known peripheral arterial disease who were administered IOMERVU 400 mg Iodine/mL by intravenous route. The mean age was 62 years (range 36 years to 88 years). Both studies assessed the diagnostic performance for detection of significant stenosis at the arterial segment level using digital subtraction angiography as the reference standard. In the first study, 212 patients (7,392 segments, 42% positive by reference standard) were independently evaluated by three blinded readers. The reported segment-level sensitivity and specificity (95% confidence interval) for the detection of ≥70% stenosis were 99% (98%, 99%) and 97% (96%, 97%), respectively. In the second study, 50 patients (929 to 933 lesions, 34% positive by reference standard) were evaluated by two blinded readers. The reported lesion-level sensitivity and specificity (95% confidence interval) for the detection of >50% stenosis were 93% (91%, 96%) and 97% (95%, 98%), respectively, for reader 1 and 90% (87%, 93%) and 96% (94%, 97%), respectively, for reader 2.
Coronary CT angiography was evaluated in two prospective, single-center clinical studies enrolling a total of 301 adult patients (259 male, 42 female) with suspected coronary artery disease who were administered IOMERVU 400 mg Iodine/mL by intravenous route. The mean age was 64 years. Both studies assessed the diagnostic performance for detection of ≥50% stenosis at the coronary artery segment level using invasive coronary angiography as the reference standard. In the first study, 210 patients (2,532 segments, 22% positive by reference standard) were independently evaluated by two blinded readers with discrepancies resolved by consensus. The segment-level sensitivity and specificity (95% confidence interval) were 84% (81%, 87%) and 94% (92%, 95%), respectively. In the second study, 91 patients (1,456 segments, 18% positive by reference standard) were independently evaluated by two blinded readers with discrepancies resolved by a third reader. The segment-level sensitivity and specificity (95% confidence interval) were 97% (95%, 99%) and 91% (89%, 92%), respectively.
CT urography was evaluated in two retrospective, single-center clinical studies of 185 adult patients (130 male, 55 female) who were administered IOMERVU 350 mg Iodine/mL by intravenous route. The mean age was 55 years (range 20 years to 89 years). In the first study, two readers assessed parenchymal image quality on a 3-point scale (inadequate, diagnostic, or very good or excellent) as very good or excellent in 64% to 98% of the patients depending on scan protocol. In the second study, two blinded readers assessed visualization quality for the urinary system overall (calyces, renal pelvis, ureter, and bladder), on a scale of 0 (absence of visualization) to 5 (excellent visualization), with the mean score (SD) for reader 1 being 4.2 (1.4) and for reader 2 being 4.1 (1.5).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
IOMERVU (iomeprol) injection is a clear, colorless to pale yellow solution supplied in clear glass single-dose vials or bottles in the following configurations:
Concentration
(mg Iodine/mL)Packages Size Package Type Sale Unit NDC 250 100 mL Single-dose bottles Carton of 10 0270-7013-14 300 50 mL Single-dose vials Carton of 10 0270-7016-15 100 mL Single-dose bottles Carton of 10 0270-7016-17 150 mL Carton of 10 0270-7016-18 200 mL Carton of 10 0270-7016-19 350 50 mL Single-dose vials Carton of 10 0270-7017-20 100 mL Single-dose bottles Carton of 10 0270-7017-21 150 mL Carton of 10 0270-7017-24 200 mL Carton of 10 0270-7017-25 400 50 mL Single-dose vials Carton of 10 0270-7018-26 100 mL Single-dose bottles Carton of 10 0270-7018-27 150 mL Carton of 10 0270-7018-28 200 mL Carton of 10 0270-7018-29 Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Keep in original carton with the cover closed to protect from light. Do not freeze.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient concerning the risk of hypersensitivity reactions that can occur both during and after IOMERVU administration. Advise the patient to report any signs or symptoms of hypersensitivity reactions during the procedure and to seek immediate medical attention for any signs or symptoms experienced after discharge [see Warnings and Precautions (5.2)].
Advise patients to inform their physician if they develop a rash after receiving IOMERVU [see Warnings and Precautions (5.11)].
Advise the patient concerning appropriate hydration to decrease the risk of kidney injury [see Warnings and Precautions (5.3)].
If extravasation occurs during injection, advise patients to seek medical care for progression of symptoms [see Warnings and Precautions (5.6)].
Advise parents or caregivers about the risk of developing thyroid dysfunction after IOMERVU administration. Advise parents or caregivers about when to seek medical care for their child to monitor for thyroid dysfunction [see Warnings and Precautions (5.8)].
Advise a lactating woman that interruption of breastfeeding is not necessary, however, to avoid any exposure a lactating woman may consider pumping and discarding breast milk for 10 hours after IOMERVU administration [see Use in Specific Populations (8.2)].
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
IOMERVU
iomeprol injection injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0270-7017 Route of Administration INTRA-ARTERIAL, INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IOMEPROL (UNII: 17E17JBP8L) (IOMEPROL - UNII:17E17JBP8L) IOMEPROL 714 mg in 1 mL Inactive Ingredients Ingredient Name Strength TROMETHAMINE (UNII: 023C2WHX2V) 1 mg in 1 mL Product Characteristics Color YELLOW Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0270-7017-20 10 in 1 CASE 01/01/2027 1 50 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product 2 NDC: 0270-7017-21 10 in 1 CASE 01/01/2026 2 100 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product 3 NDC: 0270-7017-24 10 in 1 CASE 01/01/2027 3 150 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product 4 NDC: 0270-7017-25 10 in 1 CASE 01/01/2026 4 200 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA216016 01/01/2026 IOMERVU
iomeprol injection injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0270-7018 Route of Administration INTRA-ARTERIAL, INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IOMEPROL (UNII: 17E17JBP8L) (IOMEPROL - UNII:17E17JBP8L) IOMEPROL 816 mg in 1 mL Inactive Ingredients Ingredient Name Strength TROMETHAMINE (UNII: 023C2WHX2V) 1 mg in 1 mL Product Characteristics Color YELLOW Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0270-7018-26 10 in 1 CASE 01/01/2027 1 50 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product 2 NDC: 0270-7018-27 10 in 1 CASE 01/01/2026 2 100 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product 3 NDC: 0270-7018-28 10 in 1 CASE 01/01/2027 3 150 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product 4 NDC: 0270-7018-29 10 in 1 CASE 01/01/2026 4 200 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA216016 01/01/2026 Labeler - BRACCO DIAGNOSTICS INC (849234661) Registrant - BRACCO DIAGNOSTICS INC (849234661) Establishment Name Address ID/FEI Business Operations BIPSO GmbH 342104149 MANUFACTURE(0270-7017, 0270-7018) , ANALYSIS(0270-7018, 0270-7017) Establishment Name Address ID/FEI Business Operations SPIN S.p.A. 434967237 API MANUFACTURE(0270-7018, 0270-7017) Establishment Name Address ID/FEI Business Operations BioChem Labor für biologishe und chemische Analytik GmbH 318354230 ANALYSIS(0270-7018, 0270-7017)
Trademark Results [IOMERVU]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 IOMERVU 79404602 not registered Live/Pending |
Bracco Suisse SA, 2024-07-11 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.