MITIGARE- colchicine capsule
MITIGARE by
Drug Labeling and Warnings
MITIGARE by is a Prescription medication manufactured, distributed, or labeled by Hikma Specialty USA Inc., Hikma Pharmaceuticals USA Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
Rx Only
These highlights do not include all the information needed to use MITIGARE® safely and effectively. See full prescribing information for MITIGARE®.
MITIGARE® (colchicine) capsules, for oral use
Initial U.S. Approval: 1961
INDICATIONS AND USAGE
- MITIGARE® is indicated for prophylaxis of gout flares in adults (1).
Limitations of use:
The safety and effectiveness of MITIGARE® for acute treatment of gout flares during prophylaxis has not been studied.MITIGARE® is not an analgesic medication and should not be used to treat pain from other causes.
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
- 0.6 mg Capsules (3).
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Fatal overdoses have been reported with colchicine in adults and children. Keep MITIGARE® out of the reach of children (5.1, 10).
- Blood dyscrasias: myelosuppression, leukopenia, granulocytopenia, thrombocytopenia, and aplastic anemia have been reported (5.2).
- Monitor for toxicity and if present consider temporary interruption or discontinuation of colchicine (5.2, 5.3, 5.4, 6, 10).
- Drug interaction with dual P-gp and CYP3A4 inhibitors: Co-administration of colchicine with dual P-gp and CYP3A4 inhibitors has resulted in life-threatening interactions and death (5.3, 7).
- Neuromuscular toxicity: Myotoxicity including rhabdomyolysis may occur, especially in combination with other drugs known to cause this effect. Consider temporary interruption or discontinuation of MITIGARE® (5.4, 7).
ADVERSE REACTIONS
The most commonly reported adverse reactions with colchicine are gastrointestinal symptoms, including diarrhea, nausea, vomiting, and abdominal pain (6).
To report SUSPECTED ADVERSE REACTIONS, contact Hikma Americas, Inc. at 1-800-962-8364 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Co-administration of P-gp or CYP3A4 inhibitors or inhibitors of both P-gp and CYP3A4 (e.g., clarithromycin or cyclosporine) have been reported to lead to colchicine toxicity. The potential for drug-drug interactions must be considered prior to and during therapy.
- Concomitant use of MITIGARE® and inhibitors of CYP3A4 or P-gp should be avoided if possible. If co-administration of MITIGARE® and an inhibitor of CYP3A4 or P-gp is necessary, the dose of MITIGARE® should be reduced and the patient should be monitored carefully for colchicine toxicity (7, 12.3).
USE IN SPECIFIC POPULATIONS
- In the presence of renal or hepatic impairment, patients should be monitored closely and dose adjustment should be considered as necessary (8.6, 8.7).
- Pregnancy: Use only if the potential benefit justifies the potential risk to the fetus (8.1).
- Nursing Mothers: Caution should be exercised when administered to a nursing woman (8.3).
- Geriatric Use: The recommended dose of colchicine should be based on renal/hepatic function (8.5).
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 7/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1. Gout Prophylaxis
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Fatal Overdose
5.2 Blood Dyscrasias
5.3 Interactions with CYP3A4 and P-gp Inhibitors
5.4 Neuromuscular Toxicity
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
7.1 CYP3A4
7.2 P-glycoprotein
7.3 HMG-CoA Reductase Inhibitors and Fibrates
7.4 Drug-Drug Interaction Studies
8 USE IN SPECIFIC POPULATIONS
8.1 Use in Pregnancy
8.2 Labor and Delivery
8.3 Use in Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
9 DRUG ABUSE AND DEPENDENCE
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
MITIGARE® (colchicine) capsules are indicated for prophylaxis of gout flares in adults.
Limitations of use: The safety and effectiveness of MITIGARE® for acute treatment of gout flares during prophylaxis has not been studied.
MITIGARE® is not an analgesic medication and should not be used to treat pain from other causes.
-
2 DOSAGE AND ADMINISTRATION
2.1. Gout Prophylaxis
For prophylaxis of gout flares, the recommended dosage of MITIGARE® is 0.6 mg once or twice daily. The maximum dose is 1.2 mg per day.
MITIGARE® is administered orally, without regard to meals.
The recommended dosage of MITIGARE® depends on the use of coadministered drugs [see Drug Interactions (7)].
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Patients with renal or hepatic impairment should not be given MITIGARE® with drugs that inhibit both P-glycoprotein and CYP3A4 inhibitors [see Drug Interactions (7)]. Combining these dual inhibitors with colchicine in patients with renal or hepatic impairment has resulted in life-threatening or fatal colchicine toxicity.
Patients with both renal and hepatic impairment should not be given MITIGARE®.
-
5 WARNINGS AND PRECAUTIONS
5.1 Fatal Overdose
Fatal overdoses, both accidental and intentional, have been reported in adults and children who have ingested colchicine [see Overdosage (10)]. MITIGARE® should be kept out of the reach of children.
5.2 Blood Dyscrasias
Myelosuppression, leukopenia, granulocytopenia, thrombocytopenia, pancytopenia, and aplastic anemia have been reported with colchicine used in therapeutic doses.
5.3 Interactions with CYP3A4 and P-gp Inhibitors
Because colchicine is a substrate for both the CYP3A4 metabolizing enzyme and the P-glycoprotein efflux transporter, inhibition of either of these pathways may lead to colchicine-related toxicity. Inhibition of both CYP3A4 and P-gp by dual inhibitors such as clarithromycin has been reported to produce life-threatening or fatal colchicine toxicity due to significant increases in systemic colchicine levels. Therefore, concomitant use of MITIGARE® and inhibitors of CYP3A4 or P-glycoprotein should be avoided [see Drug Interactions (7)]. If avoidance is not possible, reduced daily dose should be considered and the patient should be monitored closely for colchicine toxicity. Use of MITIGARE® in conjunction with drugs that inhibit both P-gp and CYP3A4 is contraindicated in patients with renal or hepatic impairment [see Contraindications (4)].
5.4 Neuromuscular Toxicity
Neuromuscular toxicity and rhabdomyolysis have been reported from chronic treatment with colchicine in therapeutic doses, especially in combination with other drugs known to cause this effect. Patients with impaired renal function and elderly patients (even those with normal renal and hepatic function) are at increased risk. Once colchicine treatment is ceased, the symptoms generally resolve within 1 week to several months.
-
6 ADVERSE REACTIONS
Gastrointestinal disorders are the most common adverse reactions with colchicine. They are often the first signs of toxicity and may indicate that the colchicine dose needs to be reduced or therapy stopped. These include diarrhea, nausea, vomiting, and abdominal pain.
Colchicine has been reported to cause neuromuscular toxicity, which may present as muscle pain or weakness [see Warnings and Precautions (5.4)].
Toxic manifestations associated with colchicine include myelosuppression, disseminated intravascular coagulation, and injury to cells in the renal, hepatic, circulatory, and central nervous system. These most often occur with excessive accumulation or overdosage [see Overdosage (10)].
The following reactions have been reported with colchicine. These have been generally reversible by interrupting treatment or lowering the dose of colchicine:
Digestive: abdominal cramping, abdominal pain, diarrhea, lactose intolerance, nausea, vomiting
Neurological: sensory motor neuropathy
Dermatological: alopecia, maculopapular rash, purpura, rash
Hematological: leukopenia, granulocytopenia, thrombocytopenia, pancytopenia, aplastic anemia
Hepatobiliary: elevated AST, elevated ALT
Musculoskeletal: myopathy, elevated CPK, myotonia, muscle weakness, muscle pain, rhabdomyolysis
Reproductive: azoospermia, oligospermia
To report SUSPECTED ADVERSE REACTIONS, contact Hikma Americas, Inc. at 1-800-962-8364 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
7 DRUG INTERACTIONS
Colchicine is a substrate of the efflux transporter P-glycoprotein (P-gp), and the CYP3A4 metabolizing enzyme. Fatal drug interactions have been reported when colchicine is administered with clarithromycin, a dual inhibitor of CYP3A4 and P-glycoprotein. Toxicities have also been reported when colchicine is administered with inhibitors of CYP3A4 that may not be potent inhibitors of P-gp (e.g., grapefruit juice, erythromycin, verapamil), or inhibitors of P-gp that may not be potent inhibitors of CYP3A4 (e.g., cyclosporine).
Patients with renal or hepatic impairment should not be given MITIGARE® with drugs that inhibit both P-glycoprotein and CYP3A4 [see Contraindications (4)]. Combining these dual inhibitors with MITIGARE® in patients with renal and hepatic impairment has resulted in life-threatening or fatal colchicine toxicity.
Physicians should ensure that patients are suitable candidates for treatment with MITIGARE® and remain alert for signs and symptoms of toxic reactions associated with increased colchicine exposure due to drug interactions. Signs and symptoms of colchicine toxicity should be evaluated promptly and, if toxicity is suspected, MITIGARE® should be discontinued immediately.
7.1 CYP3A4
The concomitant use of MITIGARE® and CYP3A4 inhibitors (e.g., clarithromycin, ketoconazole, grapefruit juice, erythromycin, verapamil, etc.) should be avoided due to the potential for serious and life-threatening toxicity [see Warnings and Precautions (5.3) and Clinical Pharmacology (12)].
If co-administration of MITIGARE® and a CYP3A4 inhibitor is necessary, the dose of MITIGARE® should be adjusted by either reducing the daily dose or reducing the dose frequency, and the patient should be monitored carefully for colchicine toxicity [see Clinical Pharmacology (12)].
7.2 P-glycoprotein
The concomitant use of MITIGARE® and inhibitors of P-glycoprotein (e.g. clarithromycin, ketoconazole, cyclosporine, etc.) should be avoided due to the potential for serious and life-threatening toxicity [see Warnings and Precautions (5.3) and Clinical Pharmacology (12)].
If co-administration of MITIGARE® and a P-gp inhibitor is necessary, the dose of MITIGARE® should be adjusted by either reducing the daily dose or reducing the dose frequency, and the patient should be monitored carefully for colchicine toxicity [see Clinical Pharmacology (12)].
7.3 HMG-CoA Reductase Inhibitors and Fibrates
Some drugs such as HMG-CoA reductase inhibitors and fibrates may increase the risk of myopathy when combined with MITIGARE®. Complaints of muscle pain or weakness could be an indication to check serum creatinine kinase levels for signs of myopathy.
7.4 Drug-Drug Interaction Studies
Four pharmacokinetic studies evaluated the effects of co-administration of voriconazole (200 mg BID), fluconazole (200 mg QD), cimetidine (800 mg BID), and propafenone (225 mg BID) on systemic levels of colchicine. Colchicine can be administered with these drugs at the tested doses without a need for dose adjustment. However, these results should not be extrapolated to other co-administered drugs [see Drug-Drug Interactions (7.1, 7.2) and Pharmacokinetics (12.3)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Use in Pregnancy
Pregnancy Category C. There are no adequate and well-controlled studies with MITIGARE® in pregnant women. Colchicine crosses the human placenta. Developmental studies in animals were not conducted with MITIGARE®, however published animal reproduction and development studies with colchicine demonstrated embryofetal toxicity, teratogenicity, and altered postnatal development at exposures within or above the clinical therapeutic range. Colchicine should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
8.3 Use in Nursing Mothers
Colchicine is excreted into human milk. Limited information suggests that infants exclusively breastfed receive less than 10 percent of the maternal weight-adjusted dose. While there are no published reports of adverse effects in breast-feeding infants of mothers taking colchicine, colchicine can affect gastrointestinal cell renewal and permeability. Caution should be exercised and breastfeeding infants should be observed for adverse effects when MITIGARE® is administered to a nursing woman.
8.4 Pediatric Use
Gout is rare in pediatric patients; the safety and effectiveness of MITIGARE® in pediatric patients has not been evaluated in controlled studies.
8.5 Geriatric Use
Because of the increased incidence of decreased renal function in the elderly population, and the higher incidence of other co-morbid conditions in the elderly population requiring the use of other medications, reducing the dosage of colchicine when elderly patients are treated with colchicine should be carefully considered.
8.6 Renal Impairment
No dedicated pharmacokinetic study has been conducted using MITIGARE® in patients with varying degrees of renal impairment. Colchicine is known to be excreted in urine in humans and the presence of severe renal impairment has been associated with colchicine toxicity. Urinary clearance of colchicine and its metabolites may be decreased in patients with impaired renal function. Dose reduction or alternatives should be considered for the prophylaxis of gout flares in patients with severe renal impairment. Colchicine is not effectively removed by hemodialysis. Patients who are undergoing hemodialysis should be monitored carefully for colchicine toxicity.
8.7 Hepatic Impairment
No dedicated pharmacokinetic study using MITIGARE® has been conducted in patients with varying degrees of hepatic impairment. Colchicine is known to be metabolized in humans and the presence of severe hepatic impairment has been associated with colchicine toxicity. Hepatic clearance of colchicine may be significantly reduced and plasma half-life prolonged in patients with chronic hepatic impairment.
Dose reduction or alternatives should be considered for the prophylaxis of gout flares in patients with severe hepatic impairment.
- 9 DRUG ABUSE AND DEPENDENCE
-
10 OVERDOSAGE
The dose of colchicine that would induce significant toxicity for an individual is unknown. Fatalities have been reported in patients after ingesting a dose as low as 7 mg over a 4-day period, while other patients have reportedly survived after ingesting more than 60 mg. A review of 150 patients who overdosed on colchicine found that those who ingested less than 0.5 mg/kg survived and tended to have milder adverse reactions, such as gastrointestinal symptoms, whereas those who ingested from 0.5 to 0.8 mg/kg had more severe adverse reactions, including myelosuppression. There was 100% mortality among patients who ingested more than 0.8 mg/kg.
- The first stage of acute colchicine toxicity typically begins within 24 hours of ingestion and includes gastrointestinal symptoms such as abdominal pain, nausea, vomiting, diarrhea, and significant fluid loss, leading to volume depletion. Peripheral leukocytosis may also be seen.
- Life-threatening complications occur during the second stage, which occurs 24 to 72 hours after drug administration, attributed to multi-organ failure and its associated consequences. Death usually results from respiratory depression and cardiovascular collapse. If the patient survives, recovery of multi-organ injury may be accompanied by rebound leukocytosis and alopecia starting about 1 week after the initial ingestion.
- Treatment of colchicine overdose should begin with gastric lavage and measures to prevent shock. Otherwise, treatment is symptomatic and supportive. No specific antidote is known. Colchicine is not effectively removed by hemodialysis [See Pharmacokinetics (12.3)].
-
11 DESCRIPTION
Colchicine is an alkaloid obtained from the plant colchicum autumnale.
The chemical name for colchicine is (S)-N-(5,6,7,9- tetrahydro-1,2,3,10-tetramethoxy-9 oxobenzol[a]heptalen-7-yl) acetamide. The structural formula is represented below:
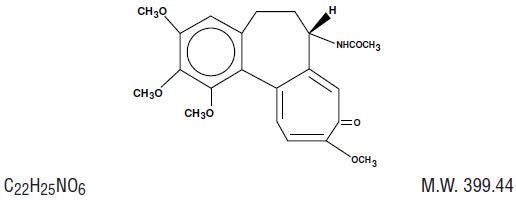
Colchicine consists of pale yellow scales or powder; it darkens on exposure to light. Colchicine is soluble in water, freely soluble in alcohol, and slightly soluble in ether.
MITIGARE® (colchicine) capsules are supplied for oral administration. Each capsule contains 0.6 mg Colchicine and the following inactive ingredients: colloidal silicon dioxide, lactose anhydrous, magnesium stearate, microcrystalline cellulose, and sodium starch glycolate. The capsule shell contains gelatin, purified water, titanium dioxide, erythrosine, Brilliant Blue FCF and Quinoline Yellow.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Colchicine’s effectiveness as a treatment for gout has been postulated to be due to its ability to block neutrophil-mediated inflammatory responses induced by monosodium urate crystals in synovial fluid. Colchicine disrupts the polymerization of β-tubulin into microtubules, thereby preventing the activation, degranulation, and migration of neutrophils to sites of inflammation. Colchicine also interferes with the inflammasome complex found in neutrophils and monocytes that mediates interleukin-1β (IL-1β) activation.
12.3 Pharmacokinetics
Absorption
In healthy adults, MITIGARE® when given orally reached a mean Cmax of 3 ng/mL in 1.3 h (range 0.7 to 2.5 h) after 0.6 mg single dose administration.
Absolute bioavailability is reported to be approximately 45%.
Administration with food has no effect on the rate or extent of colchicine absorption.
Colchicine is not effectively removed by hemodialysis.
Distribution
Colchicine has a mean apparent volume of distribution in healthy young volunteers of approximately 5 to 8 L/kg. Colchicine binding to serum protein is about 39%, primarily to albumin. Colchicine crosses the placenta and distributes into breast milk [see Pregnancy (8.1) and Nursing Mothers (8.3)].
Metabolism
A published in vitro human liver microsome study showed that about 16% of colchicine is metabolized to 2-O-demethylcolchicine and 3-O-demethylcolchicine (2- and 3-DMC, respectively) by CYP3A4. Glucuronidation is also believed to be a metabolic pathway for colchicine.
Excretion
In a published study in healthy volunteers, 40 to 65% of the total absorbed dose of colchicine (1 mg administered orally) was recovered unchanged in urine. Enterohepatic recirculation and biliary excretion are also believed to play a role in colchicine elimination. Colchicine is a substrate of P-gp and P-gp efflux is postulated to play an important role in colchicine disposition. Elimination half-life in humans was found to be 31 h (range 21.7 to 49.9 h).
Special Populations
There is no difference between men and women in the pharmacokinetic disposition of colchicine.
Pediatric Patients: Pharmacokinetics of colchicine was not evaluated in pediatric patients.
Elderly: Pharmacokinetics of colchicine have not been determined in elderly patients. A published report described the pharmacokinetics of 1 mg oral colchicine tablet in four elderly women compared to six young healthy males. The mean age of the four elderly women was 83 years (range 75 to 93), mean weight was 47 kg (38 to 61 kg) and mean creatinine clearance was 46 mL/min (range 25 to 75 mL/min). Mean peak plasma levels and AUC of colchicine were two times higher in elderly subjects compared to young healthy males. It is possible that the higher exposure in the elderly subjects was due to decreased renal function.
Renal impairment: Pharmacokinetics of colchicine in patients with mild and moderate renal impairment is not known. A published report described the disposition of colchicine (1 mg) in young adult men and women patients who had end-stage renal disease requiring dialysis compared to patients with normal renal function. Patients with end-stage renal disease had 75% lower colchicine clearance (0.17 vs. 0.73 L/hr/kg) and prolonged plasma elimination half-life (18.8 hrs vs. 4.4 hrs) as compared to subjects with normal renal function [see Renal Impairment (8.6)].
Hepatic impairment: Published reports on the pharmacokinetics of intravenous colchicine in patients with severe chronic liver disease, as well as those with alcoholic or primary biliary cirrhosis, and normal renal function suggest wide inter-patient variability. In some subjects with mild to moderate cirrhosis, the clearance of colchicine is significantly reduced and plasma half-life prolonged compared to healthy subjects. In subjects with primary biliary cirrhosis, no consistent trends were noted [see Hepatic Impairment (8.7)]. No pharmacokinetic data are available for patients with severe hepatic impairment (Child-Pugh C).
Drug Interactions
Pharmacokinetic studies evaluating changes in systemic levels of colchicine when co-administered with CYP3A4 inhibitors in healthy volunteers have been conducted with MITIGARE®. While voriconazole 200 mg BID for 5 days (considered a strong CYP3A4 inhibitor) and cimetidine 800 mg BID for 5 days (considered a weak CYP3A4 inhibitor) did not cause any changes in colchicine systemic levels, fluconazole 200 mg QD for 4 days with a 400 mg loading dose (considered a moderate CYP3A4 inhibitor) increased colchicine AUC by 40%. As voriconazole, cimetidine, and fluconazole are known as CYP3A4 inhibitors that do not inhibit P-gp, these studies show that CYP3A4 inhibition by itself may not lead to clinically significant increases in colchicine systemic levels in humans, and P-gp inhibition in addition to CYP3A4 inhibition may be necessary for clinically meaningful interactions of colchicine. However, based on published case reports that indicate the presence of colchicine toxicity when colchicine is co-administered with strong to moderate CYP3A4 inhibitors such as clarithromycin, erythromycin, grapefruit juice, etc., as well as the 40% increase in systemic levels of colchicine observed with concomitantly administered fluconazole (a moderate CYP3A4 inhibitor that is not known to inhibit P-gp) in a drug-drug interaction study, the drug-drug interaction potential of colchicine with strong or moderate CYP3A4 inhibitors that do not inhibit P-gp cannot be ruled out completely.
Co-administration of MITIGARE® with propafenone (a P-gp inhibitor) at 225 mg BID for 5 days, in a pharmacokinetic study in healthy volunteers, did not cause any changes in systemic levels of colchicine. This indicates that propafenone can be administered with MITIGARE® without any dose adjustment. However, these results should not be extrapolated to other P-gp inhibitors as colchicine is known to be a substrate for P‑gp and case reports of colchicine toxicity associated with the co-administration of P-gp inhibitors such as cyclosporine have been published.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Carcinogenicity studies of colchicine have not been conducted. Due to the potential for colchicine to produce aneuploid cells (cells with an unequal number of chromosomes), colchicine presents a theoretical increased risk of malignancy.Mutagenesis
Published studies demonstrated that colchicine was negative for mutagenicity in the bacterial reverse mutation assay. However, in vitro chromosomal aberration assays demonstrated the formation of micronuclei following colchicine treatment. Because published studies demonstrated that colchicine induces aneuploidy through the process of mitotic nondisjunction without structural DNA changes, colchicine is not considered clastogenic, although micronuclei are formed.Impairment of Fertility
There were no studies of the effects of MITIGARE® on fertility. However, published nonclinical studies have demonstrated that colchicine-induced disruption of microtubule formation affects meiosis and mitosis. Published reproductive studies with colchicine reported abnormal sperm morphology and reduced sperm counts in males, and interference with sperm penetration, second meiotic division, and normal cleavage in females.Case reports and epidemiology studies in human male subjects on colchicine therapy indicate that infertility from colchicine is rare. A case report indicated that azoospermia was reversed when therapy was stopped. Case reports and epidemiology studies in female subjects on colchicine therapy have not established a clear relationship between colchicine use and female infertility.
-
14 CLINICAL STUDIES
The evidence for the efficacy of colchicine in patients with chronic gout is derived from the published literature. Two randomized clinical trials assessed the efficacy of colchicine 0.6 mg twice a day for the prophylaxis of gout flares in patients with gout initiating treatment with urate lowering therapy. In both trials, treatment with colchicine decreased the frequency of gout flares.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
MITIGARE® (colchicine) capsules, 0.6 mg are No. 4 Dark Blue/Light Blue Hard Gelatin Capsules printed “West-ward 118” in white ink.
NDC: 59467-318-30: Bottle of 30 Capsules
NDC: 59467-318-01: Bottle of 100 Capsules
NDC: 59467-318-10: Bottle of 1000 Capsules
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Dosing Instructions
If a dose of MITIGARE® is missed, advise the patient to take the dose as soon as possible and then return to the normal dosing schedule. However, if a dose is skipped, the patient should not double the next dose.
Fatal Overdose
Advise the patient that fatal overdoses, both accidental and intentional, have been reported in adults and children who have ingested colchicine. MITIGARE® should be kept out of the reach of children.
Blood Dyscrasias
Advise patients that bone marrow depression with agranulocytosis, aplastic anemia, and thrombocytopenia may occur with MITIGARE®.
Drug and Food Interactions
Advise patients that many drugs or other substances may interact with MITIGARE® and some interactions could be fatal. Therefore, patients should report to their healthcare provider all of the current medications they are taking, and check with their healthcare provider before starting any new medications, including short-term medications such as antibiotics. Patients should also be advised to report the use of non-prescription medication or herbal products. Grapefruit and grapefruit juice may also interact and should not be consumed during treatment with MITIGARE®.
Neuromuscular Toxicity
Advise patients that muscle pain or weakness, tingling or numbness in fingers or toes may occur with MITIGARE® alone or when it is used with certain other drugs. Patients developing any of these signs or symptoms must discontinue MITIGARE® and seek medical evaluation immediately.
Mfd. for:
Hikma Americas Inc.
Memphis, TN 38120Mfd. by:
West-Ward Columbus Inc.
Columbus, OH 43228C50000042/02
Revised July 2019
-
Medication Guide
MITIGARE® (mīt-ah-gär-ay) Capsules
Rx only
What is the most important information I should know about MITIGARE®?
MITIGARE® can cause serious side effects or death if levels of MITIGARE® are too high in your body.
- Taking certain medicines with MITIGARE® can cause your level of MITIGARE® to be too high, especially if you have kidney or liver problems.
- Tell your healthcare provider about all your medical conditions, including if you have kidney or liver problems. Your dose of MITIGARE® may need to be changed.
- Even medicines that you take for a short period of time, such as antibiotics, can interact with MITIGARE® and cause serious side effects or death.
What is MITIGARE®?
MITIGARE® is a prescription medication used to prevent gout flares in adults.
It is not known if MITIGARE® is safe and effective for the treatment of:
- acute gout flares
MITIGARE® is not a pain medicine and it should not be taken to treat pain related to other conditions unless specifically for those conditions.
It is not known if MITIGARE® is safe and effective in children.
Who should not take MITIGARE®?
Do not take MITIGARE® if you have liver and kidney problems and you take certain other medicines. Serious side effects, including death, have been reported in these people even when taken as directed. See “What is the most important information I should know about MITIGARE®?”
What should I tell my healthcare provider before taking MITIGARE®?
Before you take MITIGARE®, tell your healthcare provider:
- about all of your medical conditions
- if you have kidney or liver problems
- if you are pregnant or plan to become pregnant. It is not known if MITIGARE® can harm your unborn baby. Talk to your healthcare provider if you are pregnant or plan to become pregnant.
- if you are breastfeeding or plan to breastfeed. MITIGARE® can pass into your breast milk and may harm your baby. Talk to your healthcare provider about the best way to feed your baby if you take MITIGARE®.
Tell your healthcare provider about all the medicines you take, including prescription, over-the-counter medicines, vitamins, or herbal supplements.
- Using MITIGARE® with certain other medicines can affect each other causing serious side effects and/or death.
- Do not take MITIGARE® with other medicines unless your healthcare provider tells you to.
- Know the medicines you take. Keep a list of your medicines with you to show your healthcare provider and pharmacist each time you get a new medicine.
- Especially tell your healthcare provider if you take:
- medicines that may affect how your liver works (CYP3A4 inhibitors)
- cyclosporine (Neoral®, Gengraf®, Sandimmune®)
- cholesterol lowering medicines
- antibiotics
Ask your healthcare provider or pharmacist if you are not sure if you take any of the medicines listed above. This is not a complete list of all the medicines that can affect MITIGARE®.
How should I take MITIGARE®?
- Take MITIGARE® exactly as your healthcare provider tells you to take it.
- MITIGARE® can be taken with or without food.
- If you take too much MITIGARE® call your healthcare provider or go to the nearest hospital emergency room right away.
- Do not stop taking MITIGARE® unless your healthcare provider tells you to.
- If you miss a dose of MITIGARE®, take it as soon as you remember. If it is almost time for your next dose, skip the missed dose. Take the next dose at your regular time. Do not take 2 doses at the same time.
- If you have a gout flare while taking MITIGARE®, tell your healthcare provider.
What should I avoid while taking MITIGARE®?
- Avoid eating grapefruit or drinking grapefruit juice while taking MITIGARE®. It can increase your chances of getting serious side effects.
What are the possible side effects of MITIGARE®?
MITIGARE® can cause serious side effects or death. See “What is the most important information I should know about MITIGARE®?”Get medical help right away, if you have:
- unusual bleeding or bruising
- increased infections
- weakness or fatigue
- muscle weakness or pain
- numbness or tingling in your fingers or toes
- pale or gray color to your lips, tongue, or palms of your hands
- severe diarrhea or vomiting
The most common side effects of MITIGARE® include abdominal pain, diarrhea, nausea, and vomiting.
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all of the possible side effects of MITIGARE®. For more information ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store MITIGARE®?
- Store MITIGARE® at room temperature between 68° to 77°F (20° to 25°C).
- Keep MITIGARE® in a tightly closed container.
- Keep MITIGARE® out of the light and away from moisture.
Keep MITIGARE® and all medicines out of the reach of children.
General information about the safe and effective use of MITIGARE®.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not take MITIGARE® for a condition for which it was not prescribed. Do not give MITIGARE® to other people, even if they have the same symptoms that you have. It may harm them.
This Medication Guide summarizes the most important information about MITIGARE®. If you would like more information, talk to your pharmacist or healthcare provider for information about MITIGARE® that is written for health professionals.
For more information, go to www.hikma-americas.com or call 1-800-962-8364.
What are the ingredients in MITIGARE® Capsules?
Active Ingredient: Colchicine
Inactive Ingredients: colloidal silicon dioxide, lactose anhydrous, magnesium stearate, microcrystalline cellulose and sodium starch glycolate. The capsule shell contains gelatin, purified water, titanium dioxide, erythrosine, Brilliant Blue FCF and Quinoline Yellow.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Mfd. for:
Hikma Americas Inc.
Memphis, TN 38120Mfd. by:
West-Ward Columbus Inc.
Columbus, OH 43228C50000042/02
Revised July 2019
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MITIGARE
colchicine capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59467-318 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength COLCHICINE (UNII: SML2Y3J35T) (COLCHICINE - UNII:SML2Y3J35T) COLCHICINE 0.6 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) GELATIN (UNII: 2G86QN327L) WATER (UNII: 059QF0KO0R) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C RED NO. 3 (UNII: PN2ZH5LOQY) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Product Characteristics Color BLUE (Dark/Light Blue) Score no score Shape CAPSULE Size 13mm Flavor Imprint Code West;ward;118 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59467-318-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 10/01/2014 2 NDC: 59467-318-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 10/01/2014 3 NDC: 59467-318-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 07/26/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204820 10/01/2014 Labeler - Hikma Specialty USA Inc. (078883518)
Trademark Results [MITIGARE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 MITIGARE 86224548 4761409 Live/Registered |
HIKMA PHARMACEUTICALS USA INC. 2014-03-18 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
