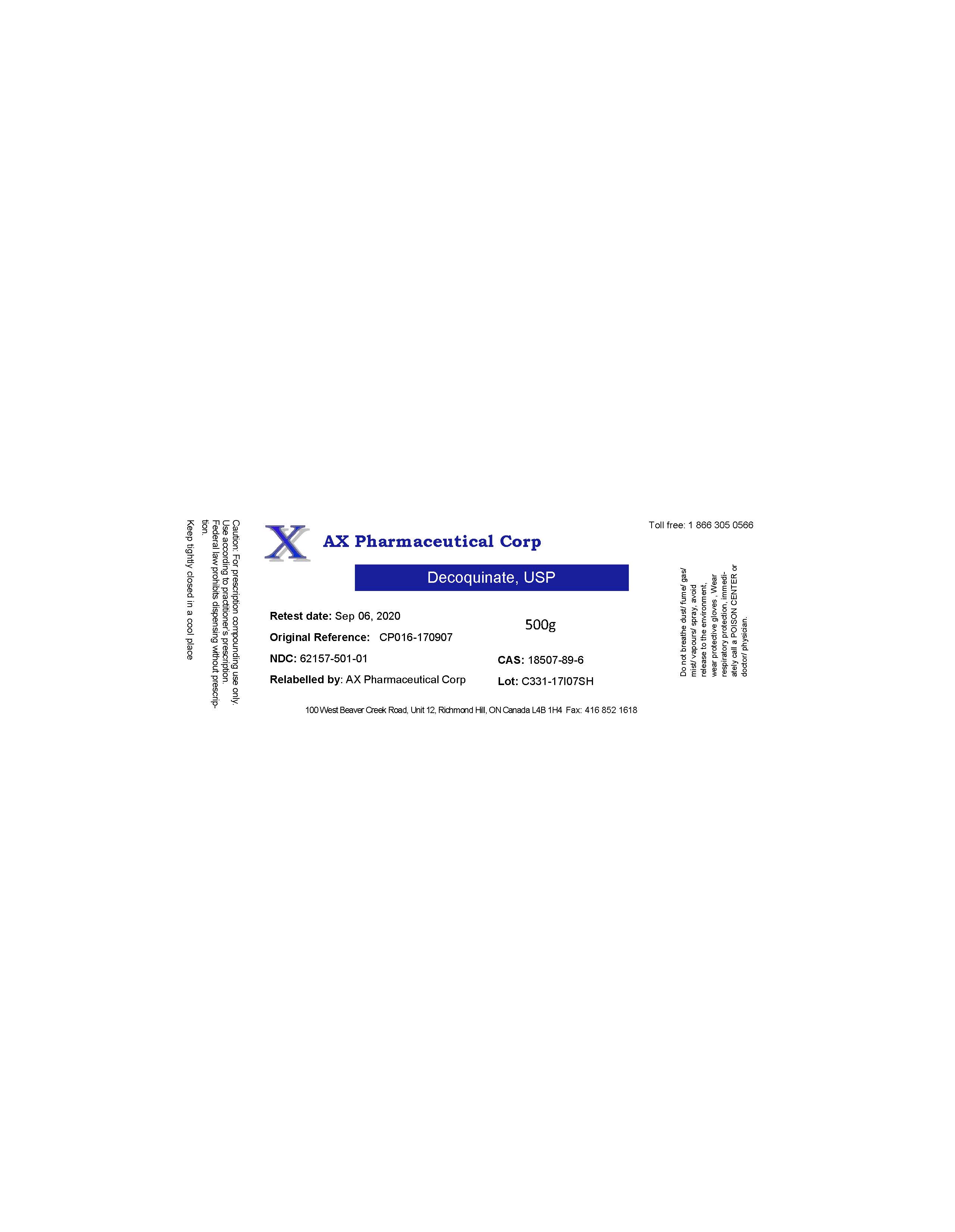
AX PHARMACEUTICAL CORP- decoquinate powder
AX Pharmaceutical Corp by
Drug Labeling and Warnings
AX Pharmaceutical Corp by is a Other medication manufactured, distributed, or labeled by AX Pharmaceutical Corp. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
AX PHARMACEUTICAL CORP
decoquinate powderProduct Information Product Type BULK INGREDIENT Item Code (Source) NDC: 62157-501 Route of Administration NOT APPLICABLE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DECOQUINATE (UNII: 534I52PVWH) (DECOQUINATE - UNII:534I52PVWH) DECOQUINATE 495 g in 500 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Product Characteristics Color yellow Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 62157-501-01 500 g in 1 JAR 12/05/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date bulk ingredient for animal drug compounding 12/05/2017 Labeler - AX Pharmaceutical Corp (202924858)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.