ADSOL RED CELL PRESERVATION SOLUTION SYSTEM IN PLASTIC CONTAINER (PL 146 PLASTIC) (anticoagulant citrate phosphate dextrose- cpd solution and adsol preservation solution kit
ADSOL Red Cell Preservation Solution System in Plastic Container (PL 146 Plastic) by
Drug Labeling and Warnings
ADSOL Red Cell Preservation Solution System in Plastic Container (PL 146 Plastic) by is a Prescription medication manufactured, distributed, or labeled by Fenwal, Inc., Fenwal International. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
Instructions for Blood Collection Using CPD OPTIPAC™ Unit with an Integrally Attached Container of ADSOL™ Red Cell Preservation Solution
Rx Only
Opti-System
Use aseptic technique
Caution: Do not use unless the solutions are clear.
- 1. Identify BLOOD-PACK™ OPTIPAC™ unit using appropriate donor identification system.
- 2. Adjust donor scale to desired collection weight/volume.
- 3. Suspend primary container from donor scale as far as possible below donor arm and clamp donor tubing with hemostat.
- 4. Apply pressure to donor’s arm and disinfect site of venipuncture.
- 5. If blood pressure cuff is used, inflate to approximately 60 mm Hg, or use tourniquet as applicable.
- 6. Remove needle cover per instructions below:
- a) Hold the needle hub upwards. With the other hand, grasp the base of the needle cover (Figure 1), twist approximately 1/4 turn to break tamper evident seal (Figure 2).
- b) Remove needle cover (Figure 3), be careful not to drag cover across the needle point (Figure 4).
- 7. Perform venipuncture, appropriately secure donor needle and/or tubing and release hemostat.
- 8. Mix blood and anticoagulant at several intervals during collection and immediately after collection.
- 9. Collect labeled volume of blood. Anticoagulant volume is sufficient for labeled volume ± 10%.
- 10. Apply hemostat to donor tubing.
- 11. If it is necessary, obtain whole anticoagulated blood samples for typing or crossmatching by returning donor tubing blood to the primary bag, mixing and allowing tubing to refill. Repeat once. Seal donor tubing at the desired intervals in order to obtain the desired total blood samples.
- 12. Centrifuge primary and secondary bags in order to prepare CPD Red Blood Cells.
- 13. Refer to the OPTIPRESS™ Blood Components Automated Extractor User Manual, in order to prepare the components.
- 14. After separation, seal transfer tubing in three places near the primary bag and cut middle seal taking care to avoid fluid splatter.
NOTE: In OPTIPAC Quadruple BLOOD-PACK unit, the empty transfer unit may be used for preparation of other components.- 15. Fill empty tubing of OPTIPAC AS-1 for red blood cells by stripping the tubing and mixing the AS-1 containter, twice, in order to ensure that the tubing is correctly filled. Seal on "X" marks on tubing in order to obtain red blood cells samples.
- 16. Mix Adsol Solution and red cells thoroughly.
- 17. Store suspended Red Blood Cells between 1 and 6 °C.
For further processing, use standard techniques for component processing.
Sterile, non-pyrogenic fluid path. Sterilized by steam. Single use only. Dispose of container appropriately.
Store at Controlled Room Temperature
USP Definition of “Controlled Room Temperature”
United States Pharmacopeia, General Notices.
United States Pharmacopeial Convention, Inc.
12601 Twinbrook Parkway, Rockville, MD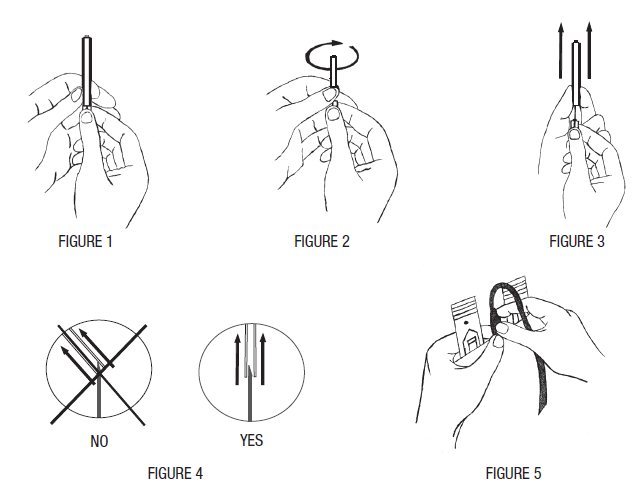
 Manufactured by:
Manufactured by:
Fenwal International, Inc.
Road 357, Km. 0.8
Maricao, PR 00606Made in USA
07-19-05-015 REV: A
07/2010Imported and distributed in Indonesia by:
PT.Medquest Jaya Global
Menara Salemba 6th Floor
Jl.Salemba Raya Kav 5-5A
Jakarta-Indonesia 10440
Reg. No.: DEPKES RI AKL 20209902359Imported and distributed in Thailand by:
Fenwal (Thailand) Ltd.
17th Fl. Thanapoom Tower
1550 New Petchburi Rd., Makasan
Rajthevi, Bangkok 10400
Thailand
Reg. No.:Imported and distributed in Philippines by:
Medlink Marketing
Unit 404 Vicar’s Building,
No.31 Visayas Ave.,
Vasra, Quezon City 1100
Philippines
Reg. No.: DVR-5805 – Manufacturer
– ManufacturerFENWAL, ADSOL, BLOOD-PACK, OPTIPAC and OPTIPRESS are trademarks of Fenwal, Inc.
© 2010 Fenwal, Inc. All rights reserved.
-
PACKAGE/LABEL DISPLAY PANEL
Code A4R7703
5 Units
Fenwal™
Quadruple BLOOD-PACK™ OPTIPAC™ Unit CPD/ADSOL™ OPTI-SYSTEM™
Rx only
Quadruple BLOOD-PACK OPTIPAC unit consisting of a primary pack containing 63 mL of CPD anticoagulant solution for collection of 450 mL of blood, one TRANSFER-PACK containing 100 mL of ADSOL solution and two TRANSFER-PACK containers without solution. 16 gauge needle.
Each 63 mL of CPD solution contains: 1.66 g, Sodium citrate (dihydrate), USP, 1.61 g Dextrose (monohydrate), USP, 188 mg Citric acid (anhydrous), USP, 140 mg Monobasic sodium phosphate (monohydrate), USP. Water for Injection USP to 63 mL
Each 100 mL of ADSOL solution contains: 2.20 g Dextrose (monohydrate), USP - 900 mg Sodium Chloride (monohydrate,) USP - 750 mg Mannitol, USP - 27.0 mg Adenine, USP. Water for injection USP to 100 mL.
See instructions for use. Sterile, non-pyrogenic fluid path. Steam sterilized. Single use only. Do not vent. Do not use if there is visible evidence of deterioration. Dispose of container properly.
Store at Controlled Room Temperature
(refer to direction insert).Unused units in open foil pouch may be kept up to 60 days by folding and securing open end of foil pouch to prevent possible loss of moisture.
Direct handling of product surfaces prior to extended storage in the foil pouch may result in mold growth.
Units removed from the foil pouch must be used within 4 days (96 hours). Units out of the foil pouch for longer than 4 days must be discarded.
 Manufactured by:
Manufactured by:
Fenwal International, Inc.
Road 357, Km. 0.8
Maricao, PR 00606Made in USA
Imported and distributed in Thailand by:
Fenwal (Thailand) Ltd.
17th Fl. Thanapoom Tower
1550 New Petchburi Rd., Makasan
Rajthevi, Bangkok 10400
ThailandImported and distributed in Indonesia by:
PT.Medquest Jaya Global
Menara Salemba 6th Floor
Jl.Salemba Raya Kav 5-5A
Jakarta-Indonesia 10440
Reg. No.: DEPKES RI AKL 20209902359FENWAL, BLOOD-PACK, ADSOL, OPTIPAC and OPTI-SYSTEM are trademarks of Fenwal, Inc.
07-28-05-406 REV: A
Lot No.:
Exp. Date:

-
INGREDIENTS AND APPEARANCE
ADSOL RED CELL PRESERVATION SOLUTION SYSTEM IN PLASTIC CONTAINER (PL 146 PLASTIC)
anticoagulant citrate phosphate dextrose (cpd) solution and adsol preservation solution kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0942-6502 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0942-6502-04 1 in 1 KIT Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BAG 63 mL Part 2 1 BAG 100 mL Part 1 of 2 CPD
citrate phosphate dextrose solutionProduct Information Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Trisodium Citrate Dihydrate (UNII: B22547B95K) (Anhydrous Citric Acid - UNII:XF417D3PSL) Anhydrous Citric Acid 1.66 g in 63 mL Dextrose Monohydrate (UNII: LX22YL083G) (ANHYDROUS DEXTROSE - UNII:5SL0G7R0OK) Dextrose Monohydrate 1.61 g in 63 mL Anhydrous Citric Acid (UNII: XF417D3PSL) (Anhydrous Citric Acid - UNII:XF417D3PSL) Anhydrous Citric Acid 188 mg in 63 mL Sodium Phosphate, Monobasic, Monohydrate (UNII: 593YOG76RN) (PHOSPHATE ION - UNII:NK08V8K8HR) Sodium Phosphate, Monobasic, Monohydrate 140 mg in 63 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 63 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA BN811104 01/11/2011 Part 2 of 2 ADSOL RED CELL PRESERVATION SOLUTION SYSTEM
adsol red cell preservation solution solutionProduct Information Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Dextrose Monohydrate (UNII: LX22YL083G) (ANHYDROUS DEXTROSE - UNII:5SL0G7R0OK) Dextrose Monohydrate 2.2 g in 100 mL Sodium Chloride (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37) Sodium Chloride 900 mg in 100 mL Mannitol (UNII: 3OWL53L36A) (Mannitol - UNII:3OWL53L36A) Mannitol 750 mg in 100 mL Adenine (UNII: JAC85A2161) (Adenine - UNII:JAC85A2161) Adenine 27 mg in 100 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 100 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA BN811104 01/11/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA BN811104 01/11/2011 Labeler - Fenwal, Inc. (794519020) Establishment Name Address ID/FEI Business Operations Fenwal International, Inc. 091164590 MANUFACTURE(0942-6502)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.