AMPICILLIN SODIUM AND SULBACTAM SODIUM injection, powder, for solution
ampicillin sodium and sulbactam sodium by
Drug Labeling and Warnings
ampicillin sodium and sulbactam sodium by is a Prescription medication manufactured, distributed, or labeled by Mylan Institutional LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
SPL UNCLASSIFIED SECTION
PHARMACY BULK PACKAGE NOT FOR DIRECT INFUSION
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ampicillin and sulbactam for injection and other antibacterial drugs, ampicillin and sulbactam for injection should be used only to treat infections that are proven or strongly suspected to be caused by bacteria.
-
DESCRIPTION
Ampicillin and Sulbactam for Injection, USP is an injectable antibacterial combination consisting of the semisynthetic antibacterial ampicillin sodium and the beta-lactamase inhibitor sulbactam sodium for intravenous administration and intramuscular administration.
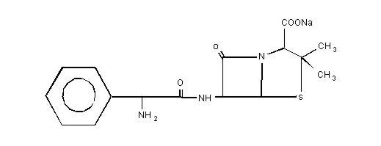
Ampicillin sodium is derived from the penicillin nucleus, 6-aminopenicillanic acid. Chemically, it is monosodium (2S, 5R, 6R)-6-[(R)-2-amino-2-phenylacetamido]-3, 3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate and has a molecular weight of 371.39. Its chemical formula is C16H18N3NaO4S. The structural formula is:
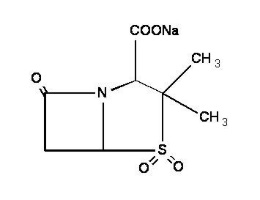
Sulbactam sodium is a derivative of the basic penicillin nucleus. Chemically, sulbactam sodium is sodium penicillinate sulfone; sodium (2S, 5R)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo [3.2.0] heptane-2-carboxylate 4,4-dioxide. Its chemical formula is C8H10NNaO5S with a molecular weight of 255.22. The structural formula is:
Ampicillin and Sulbactam for Injection, USP, ampicillin sodium/sulbactam sodium parenteral combination, is available as a white to off-white dry powder for reconstitution. Ampicillin and sulbactam for injection dry powder is freely soluble in aqueous diluents to yield pale yellow to yellow solutions containing ampicillin sodium and sulbactam sodium equivalent to 250 mg ampicillin per mL and 125 mg sulbactam per mL. The pH of the solutions is between 8.0 and 10.0.
Dilute solutions (up to 30 mg ampicillin and 15 mg sulbactam per mL) are essentially colorless to pale yellow. The pH of dilute solutions remains the same.
Ampicillin and Sulbactam for Injection, USP pharmacy bulk package is a bottle containing a sterile preparation of ampicillin sodium and sulbactam sodium for parenteral use that contains many single doses. The Pharmacy Bulk Package is for use in a pharmacy admixture setting; it provides many single doses of ampicillin and sulbactam for injection for addition to suitable parenteral fluids in the preparation of admixtures for intravenous infusion.
(See DIRECTIONS FOR USE – Directions for Proper Use of Pharmacy Bulk Package.)
Each 15 g ampicillin and sulbactam for injection Pharmacy Bulk Package bottle contains ampicillin sodium and sulbactam sodium equivalent to 10 g ampicillin and 5 g sulbactam. The sodium content per 1.5 gram is 115.1 mg and 5 mEq.
-
CLINICAL PHARMACOLOGY
General
Immediately after completion of a 15-minute intravenous infusion of ampicillin and sulbactam for injection, peak serum concentrations of ampicillin and sulbactam are attained. Ampicillin serum levels are similar to those produced by the administration of equivalent amounts of ampicillin alone. Peak ampicillin serum levels ranging from 109 to 150 mcg/mL are attained after administration of 2,000 mg of ampicillin plus 1,000 mg sulbactam and 40 to 71 mcg/mL after administration of 1,000 mg ampicillin plus 500 mg sulbactam. The corresponding mean peak serum levels for sulbactam range from 48 to 88 mcg/mL and 21 to 40 mcg/mL, respectively. After an intramuscular injection of 1,000 mg ampicillin plus 500 mg sulbactam, peak ampicillin serum levels ranging from 8 to 37 mcg/mL and peak sulbactam serum levels ranging from 6 to 24 mcg/mL are attained.
The mean serum half-life of both drugs is approximately 1 hour in healthy volunteers.
Approximately 75% to 85% of both ampicillin and sulbactam are excreted unchanged in the urine during the first 8 hours after administration of ampicillin and sulbactam for injection to individuals with normal renal function. Somewhat higher and more prolonged serum levels of ampicillin and sulbactam can be achieved with the concurrent administration of probenecid.
In patients with impaired renal function the elimination kinetics of ampicillin and sulbactam are similarly affected, hence the ratio of one to the other will remain constant whatever the renal function. The dose of ampicillin and sulbactam for injection in such patients should be administered less frequently in accordance with the usual practice for ampicillin (see DOSAGE and ADMINISTRATION section).
Ampicillin has been found to be approximately 28% reversibly bound to human serum protein and sulbactam approximately 38% reversibly bound.
The following average levels of ampicillin and sulbactam were measured in the tissues and fluids listed:
TABLE 1 Concentration of Ampicillin and Sulbactam in Various Body Tissues and Fluids Fluid or Tissue Dose
(grams)
Ampicillin/SulbactamConcentration
(mcg/mL or mcg/g)
Ampicillin/SulbactamPeritoneal Fluid
0.5/0.5 IV
7/14
Blister Fluid (Cantharides)
0.5/0.5 IV
8/20
Tissue Fluid
1/0.5 IV
8/4
Intestinal Mucosa
0.5/0.5 IV
11/18
Appendix
2/1 IV
3/40
Penetration of both ampicillin and sulbactam into cerebrospinal fluid in the presence of inflamed meninges has been demonstrated after IV administration of ampicillin and sulbactam for injection.
The pharmacokinetics of ampicillin and sulbactam in pediatric patients receiving ampicillin and sulbactam for injection are similar to those observed in adults. Immediately after a 15-minute infusion of 50 to 75 mg ampicillin and sulbactam for injection/kg body weight, peak serum and plasma concentrations of 82 to 446 mcg ampicillin/mL and 44 to 203 mcg sulbactam/mL were obtained. Mean half-life values were approximately 1 hour.
-
MICROBIOLOGY
Ampicillin is similar to benzyl penicillin in its bactericidal action against susceptible organisms during the stage of active multiplication. It acts through the inhibition of cell wall mucopeptide biosynthesis. Ampicillin has a broad spectrum of bactericidal activity against many gram-positive and gram-negative aerobic and anaerobic bacteria. (Ampicillin is, however, degraded by beta-lactamases and therefore the spectrum of activity does not normally include organisms which produce these enzymes).
A wide range of beta-lactamases found in microorganisms resistant to penicillins and cephalosporins have been shown in biochemical studies with cell free bacterial systems to be irreversibly inhibited by sulbactam. Although sulbactam alone possesses little useful antibacterial activity except against the Neisseriaceae, whole organism studies have shown that sulbactam restores ampicillin activity against beta-lactamase producing strains. In particular, sulbactam has good inhibitory activity against the clinically important plasmid mediated beta-lactamases most frequently responsible for transferred drug resistance. Sulbactam has no effect on the activity of ampicillin against ampicillin susceptible strains.
The presence of sulbactam in the ampicillin and sulbactam for injection formulation effectively extends the antibacterial spectrum of ampicillin to include many bacteria normally resistant to it and to other beta-lactam antibacterials. Thus, ampicillin and sulbactam for injection possesses the properties of a broad-spectrum antibacterial and a beta-lactamase inhibitor.
While in vitro studies have demonstrated the susceptibility of most strains of the following organisms, clinical efficacy for infections other than those included in the INDICATIONS and USAGE section has not been documented.
Gram-Positive Bacteria: Staphylococcus aureus (beta-lactamase and non-beta-lactamase producing), Staphylococcus epidermidis (beta-lactamase and non-beta-lactamase producing), Staphylococcus saprophyticus (beta-lactamase and non-beta-lactamase producing), Streptococcus faecalis† (Enterococcus), Streptococcus pneumoniae† (formerly D. pneumoniae), Streptococcus pyogenes†, Streptococcus viridans†.
Gram-Negative Bacteria: Hemophilus influenzae (beta-lactamase and non-beta-lactamase producing), Moraxella (Branhamella) catarrhalis (beta-lactamase and non-beta-lactamase producing), Escherichia coli (beta-lactamase and non-beta-lactamase producing), Klebsiella species (all known strains are beta-lactamase producing), Proteus mirabilis (beta-lactamase and non-beta-lactamase producing), Proteus vulgaris, Providencia rettgeri, Providencia stuartii, Morganella morganii, and Neisseria gonorrhoeae (beta-lactamase and non-beta-lactamase producing).
Anaerobes: Clostridium species, †Peptococcus species, †Peptostreptococcus species, Bacteroides species, including B. fragilis.
† These are not beta-lactamase producing strains and, therefore, are susceptible to ampicillin alone.
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
-
INDICATIONS AND USAGE
Ampicillin and Sulbactam for Injection, USP is indicated for the treatment of infections due to susceptible strains of the designated microorganisms in the conditions listed below.
Skin and Skin Structure Infections caused by beta-lactamase producing strains of Staphylococcus aureus, Escherichia coli,1 Klebsiella spp.1 (including K. pneumoniae1), Proteus mirabilis,1 Bacteroides fragilis,1 Enterobacter spp.,1 and Acinetobacter calcoaceticus.1
NOTE: For information on use in pediatric patients see PRECAUTIONS–Pediatric Use and CLINICAL STUDIES sections.
Intra-Abdominal Infections caused by beta-lactamase producing strains of Escherichia coli, Klebsiella spp. (including K. pneumoniae1), Bacteroides spp. (including B. fragilis), and Enterobacter spp.1
Gynecological Infections caused by beta-lactamase producing strains of Escherichia coli,1 and Bacteroides spp.1 (including B. fragilis1).
While ampicillin and sulbactam for injection, USP is indicated only for the conditions listed above, infections caused by ampicillin-susceptible organisms are also amenable to treatment with ampicillin and sulbactam for injection, USP due to its ampicillin content. Therefore, mixed infections caused by ampicillin-susceptible organisms and beta-lactamase producing organisms susceptible to ampicillin and sulbactam for injection, USP should not require the addition of another antibacterial.
Appropriate culture and susceptibility tests should be performed before treatment in order to isolate and identify the organisms causing infection and to determine their susceptibility to ampicillin and sulbactam for injection, USP.
Therapy may be instituted prior to obtaining the results from bacteriological and susceptibility studies when there is reason to believe the infection may involve any of the beta-lactamase producing organisms listed above in the indicated organ systems. Once the results are known, therapy should be adjusted if appropriate.
To reduce the development of drug-resistant bacteria and maintain effectiveness of ampicillin and sulbactam for injection, USP and other antibacterial drugs, ampicillin and sulbactam for injection, USP should be used only to treat infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
- 1 Efficacy for this organism in this organ system was studied in fewer than 10 infections.
-
CONTRAINDICATIONS
The use of ampicillin and sulbactam for injection is contraindicated in individuals with a history of serious hypersensitivity reactions (e.g., anaphylaxis or Stevens-Johnson syndrome) to ampicillin, sulbactam or to other beta-lactam antibacterial drugs (e.g., penicillins and cephalosporins).
Ampicillin and sulbactam for injection is contraindicated in patients with a previous history of cholestatic jaundice/hepatic dysfunction associated with ampicillin and sulbactam for injection.
-
WARNINGS
Hypersensitivity
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients on penicillin therapy. These reactions are more apt to occur in individuals with a history of penicillin hypersensitivity and/or hypersensitivity reactions to multiple allergens. There have been reports of individuals with a history of penicillin hypersensitivity who have experienced severe reactions when treated with cephalosporins. Before therapy with a penicillin, careful inquiry should be made concerning previous hypersensitivity reactions to penicillins, cephalosporins, and other allergens. If an allergic reaction occurs, ampicillin and sulbactam for injection should be discontinued and the appropriate therapy instituted.
Hepatotoxicity
Hepatic dysfunction, including hepatitis and cholestatic jaundice has been associated with the use of ampicillin and sulbactam for injection. Hepatic toxicity is usually reversible; however, deaths have been reported. Hepatic function should be monitored at regular intervals in patients with hepatic impairment.
Severe Cutaneous Adverse Reactions
Ampicillin and sulbactam for injection may cause severe skin reactions, such as toxic epidermal necrolysis (TEN), Stevens-Johnson syndrome (SJS), dermatitis exfoliative, erythema multiforme, and Acute generalized exanthematous pustulosis (AGEP). If patients develop a skin rash they should be monitored closely and ampicillin and sulbactam for injection discontinued if lesions progress (see CONTRAINDICATIONS and ADVERSE REACTIONS sections).
Clostridium difficile-Associated Diarrhea
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including ampicillin and sulbactam for injection, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
-
PRECAUTIONS
General
A high percentage of patients with mononucleosis who receive ampicillin develop a skin rash. Thus, ampicillin class antibacterials should not be administered to patients with mononucleosis. In patients treated with ampicillin and sulbactam for injection the possibility of superinfections with mycotic or bacterial pathogens should be kept in mind during therapy. If superinfections occur (usually involving Pseudomonas or Candida), the drug should be discontinued and/or appropriate therapy instituted.
Prescribing ampicillin and sulbactam for injection in the absence of proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Information for Patients
Patients should be counseled that antibacterial drugs including ampicillin and sulbactam for injection should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When ampicillin and sulbactam for injection is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by ampicillin and sulbactam for injection or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibacterials which usually ends when the antibacterial is discontinued. Sometimes after starting treatment with antibacterials, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibacterial. If this occurs, patients should contact their physician as soon as possible.
Drug Interactions
Probenecid decreases the renal tubular secretion of ampicillin and sulbactam. Concurrent use of probenecid with ampicillin and sulbactam for injection may result in increased and prolonged blood levels of ampicillin and sulbactam. The concurrent administration of allopurinol and ampicillin increases substantially the incidence of rashes in patients receiving both drugs as compared to patients receiving ampicillin alone. It is not known whether this potentiation of ampicillin rashes is due to allopurinol or the hyperuricemia present in these patients. There are no data with ampicillin and sulbactam for injection and allopurinol administered concurrently. Ampicillin and sulbactam for injection and aminoglycosides should not be reconstituted together due to the in vitro inactivation of aminoglycosides by the ampicillin component of ampicillin and sulbactam for injection.
Drug/Laboratory Test Interactions
Administration of ampicillin and sulbactam for injection will result in high urine concentration of ampicillin. High urine concentrations of ampicillin may result in false positive reactions when testing for the presence of glucose in urine using Clinitest™, Benedict's Solution or Fehling's Solution. It is recommended that glucose tests based on enzymatic glucose oxidase reactions (such as Clinistix™ or Testape™) be used. Following administration of ampicillin to pregnant women, a transient decrease in plasma concentration of total conjugated estriol, estriol-glucuronide, conjugated estrone and estradiol has been noted. This effect may also occur with ampicillin and sulbactam for injection.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate carcinogenic or mutagenic potential.
Pregnancy
Reproduction studies have been performed in mice, rats, and rabbits at doses up to ten (10) times the human dose and have revealed no evidence of impaired fertility or harm to the fetus due to ampicillin and sulbactam for injection. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed. (see –PRECAUTIONS-Drug/Laboratory Test Interactions section).
Labor and Delivery
Studies in guinea pigs have shown that intravenous administration of ampicillin decreased the uterine tone, frequency of contractions, height of contractions, and duration of contractions. However, it is not known whether the use of ampicillin and sulbactam for injection in humans during labor or delivery has immediate or delayed adverse effects on the fetus, prolongs the duration of labor, or increases the likelihood that forceps delivery or other obstetrical intervention or resuscitation of the newborn will be necessary.
Nursing Mothers
Low concentrations of ampicillin and sulbactam are excreted in the milk; therefore, caution should be exercised when ampicillin and sulbactam for injection is administered to a nursing woman.
Pediatric Use
The safety and effectiveness of ampicillin and sulbactam for injection have been established for pediatric patients one year of age and older for skin and skin structure infections as approved in adults. Use of ampicillin and sulbactam for injection in pediatric patients is supported by evidence from adequate and well-controlled studies in adults with additional data from pediatric pharmacokinetic studies, a controlled clinical trial conducted in pediatric patients and post-marketing adverse events surveillance. (see CLINICAL PHARMACOLOGY, INDICATIONS AND USAGE, ADVERSE REACTIONS, DOSAGE AND ADMINISTRATION, and CLINICAL STUDIES sections).
The safety and effectiveness of ampicillin and sulbactam for injection have not been established for pediatric patients for intra-abdominal infections.
-
ADVERSE REACTIONS
Adult Patients
Ampicillin and sulbactam for injection is generally well tolerated. The following adverse reactions have been reported in clinical trials.
Local Adverse Reactions
Pain at IM injection site – 16%
Pain at IV injection site – 3%
Thrombophlebitis – 3%
Phlebitis – 1.2%
Systemic Adverse Reactions
The most frequently reported adverse reactions were diarrhea in 3% of the patients and rash in less than 2% of the patients.
Additional systemic reactions reported in less than 1% of the patients were: itching, nausea, vomiting, candidiasis, fatigue, malaise, headache, chest pain, flatulence, abdominal distension, glossitis, urine retention, dysuria, edema, facial swelling, erythema, chills, tightness in throat, substernal pain, epistaxis and mucosal bleeding.
Pediatric Patients
Available safety data for pediatric patients treated with ampicillin and sulbactam for injection demonstrate a similar adverse events profile to those observed in adult patients. Additionally, atypical lymphocytosis has been observed in one pediatric patient receiving ampicillin and sulbactam for injection.
Adverse Laboratory Changes
Adverse laboratory changes without regard to drug relationship that were reported during clinical trials were:
Hepatic: Increased AST (SGOT), ALT (SGPT), alkaline phosphatase, and LDH.
Hematologic: Decreased hemoglobin, hematocrit, RBC, WBC, neutrophils, lymphocytes, platelets and increased lymphocytes, monocytes, basophils, eosinophils, and platelets.
Blood Chemistry: Decreased serum albumin and total proteins.
Renal: Increased BUN and creatinine.
Urinalysis: Presence of RBC's and hyaline casts in urine.
Postmarketing Experience
In addition to adverse reactions reported from clinical trials, the following have been identified during post-marketing use of ampicillin and sulbactam for injection or other products containing ampicillin. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to a combination of their seriousness, frequency, or potential causal connection to ampicillin and sulbactam for injection.
Blood and Lymphatic System Disorders: Hemolytic anemia, thrombocytopenic purpura, and agranulocytosis have been reported. These reactions are usually reversible on discontinuation of therapy and are believed to be hypersensitivity phenomena. Some individuals have developed positive direct Coombs Tests during treatment with ampicillin and sulbactam for injection, as with other beta-lactam antibacterials.
Gastrointestinal Disorders:Abdominal pain, cholestatic hepatitis, cholestasis, hyperbilirubinemia, jaundice, abnormal hepatic function, melena, gastritis, stomatitis, dyspepsia, black “hairy” tongue, and Clostridium difficile associated diarrhea (see CONTRAINDICATIONS and WARNINGS sections).
General Disorders and Administration Site Conditions: Injection site reaction
Immune System Disorders: Serious and fatal hypersensitivity (anaphylactic) reactions (See WARNINGS section).
Nervous System Disorders: Convulsion and dizziness
Renal and Urinary Disorders: Tubulointerstitial nephritis
Respiratory, Thoracic and Mediastinal Disorders: Dyspnea
Skin and Subcutaneous Tissue Disorders: Toxic epidermal necrolysis, Stevens-Johnson syndrome, angioedema, Acute generalized exanthematous pustulosis (AGEP), erythema multiforme, exfoliative dermatitis, and urticaria (see CONTRAINDICATIONS and WARNINGS sections).
To report SUSPECTED ADVERSE REACTIONS, contact Mylan at 1-877-446-3679 (1-877-4-INFO-RX) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch for voluntary reporting of adverse reactions.
-
OVERDOSAGE
Neurological adverse reactions, including convulsions, may occur with the attainment of high CSF levels of beta-lactams. Ampicillin may be removed from circulation by hemodialysis. The molecular weight, degree of protein binding and pharmacokinetics profile of sulbactam suggest that this compound may also be removed by hemodialysis.
-
CLINICAL STUDIES
Skin and Skin Structure Infections in Pediatric Patients
Data from a controlled clinical trial conducted in pediatric patients provided evidence supporting the safety and efficacy of ampicillin and sulbactam for injection for the treatment of skin and skin structure infections. Of 99 pediatric patients evaluable for clinical efficacy, 60 patients received a regimen containing intravenous ampicillin and sulbactam for injection, and 39 patients received a regimen containing intravenous cefuroxime. This trial demonstrated similar outcomes (assessed at an appropriate interval after discontinuation of all antimicrobial therapy) for ampicillin and sulbactam for injection - and cefuroxime-treated patients:
TABLE 4 Therapeutic Regimen
Clinical Success
Clinical Failure
Ampicillin and Sulbactam
51/60 (85%)
9/60 (15%)
Cefuroxime
34/39 (87%)
5/39 (13%)
Most patients received a course of oral antimicrobials following initial treatment with intravenous administration of parenteral antimicrobials. The study protocol required that the following three criteria be met prior to transition from intravenous to oral antimicrobial therapy: (1) receipt of a minimum of 72 hours of intravenous therapy; (2) no documented fever for prior 24 hours; and (3) improvement or resolution of the signs and symptoms of infection.
The choice of oral antimicrobial agent used in this trial was determined by susceptibility testing of the original pathogen, if isolated, to oral agents available. The course of oral antimicrobial therapy should not routinely exceed 14 days.
-
DOSAGE AND ADMINISTRATION
The pharmacy bulk package is for preparation of solutions for IV infusion only. Ampicillin and sulbactam should be administered by slow intravenous injection over at least 10 to 15 minutes or can also be delivered in greater dilutions with 50 to 100 mL of a compatible diluent as an intravenous infusion over 15 to 30 minutes.
The recommended adult dosage of ampicillin and sulbactam for injection is 1.5 g (1 g ampicillin as the sodium salt plus 0.5 g sulbactam as the sodium salt) to 3 g (2 g ampicillin as the sodium salt plus 1 g sulbactam as the sodium salt) every six hours. This 1.5 to 3 g range represents the total of ampicillin content plus the sulbactam content of ampicillin and sulbactam for injection, and corresponds to a range of 1 g ampicillin/0.5 g sulbactam to 2 g ampicillin/1 g sulbactam. The total dose of sulbactam should not exceed 4 grams per day.
Pediatric Patients 1 Year of Age or Older
The recommended daily dose of ampicillin and sulbactam for injection in pediatric patients is 300 mg per kg of body weight administered via intravenous infusion in equally divided doses every 6 hours. This 300 mg/kg/day dosage represents the total ampicillin content plus the sulbactam content of ampicillin and sulbactam for injection, and corresponds to 200 mg ampicillin/100 mg sulbactam per kg per day. The safety and efficacy of ampicillin and sulbactam for injection administered via intramuscular injection in pediatric patients have not been established. Pediatric patients weighing 40 kg or more should be dosed according to adult recommendations, and the total dose of sulbactam should not exceed 4 grams per day. The course of intravenous therapy should not routinely exceed 14 days. In clinical trials, most children received a course of oral antimicrobials following initial treatment with intravenous ampicillin and sulbactam for injection (see CLINICAL STUDIES section).
Impaired Renal Function
In patients with impairment of renal function the elimination kinetics of ampicillin and sulbactam are similarly affected, hence the ratio of one to the other will remain constant whatever the renal function. The dose of ampicillin and sulbactam for injection in such patients should be administered less frequently in accordance with the usual practice for ampicillin and according to the following recommendations:
TABLE 5
Ampicillin and Sulbactam for Injection Dosage Guide for Patients with Renal Impairment
- Creatinine Clearance
(mL/min/1.73m2)
- Ampicillin/Sulbactam
- Half-Life (Hours)
Recommended
Ampicillin and Sulbactam for Injection Dosage
≥30
1
1.5 to 3 g q 6h to q 8h
15 to 29
5
1.5 to 3 g q 12h
5 to 14
9
1.5 to 3 g q 24h
When only serum creatinine is available, the following formula (based on sex, weight, and age of the patient) may be used to convert this value into creatinine clearance. The serum creatinine should represent a steady-state of renal function.
Males weight (kg) × (140 – age)
72 × serum creatinine
Females 0.85 × above value
- COMPATIBILITY, RECONSTITUTION AND STABILITY
-
Intravenous Administration
Directions for Proper Use of Pharmacy Bulk Package
Ampicillin and sulbactam for injection sterile powder for intravenous use may be reconstituted with any of the compatible diluents described in this insert. Solutions should be allowed to stand after dissolution to allow any foaming to dissipate in order to permit visual inspection for complete solubilization. Ampicillin and sulbactam concentrations between 3 and 45 mg (2 to 30 mg ampicillin/1 to 15 mg sulbactam/mL) are recommended for intravenous use.
The 15 gram bottle may be reconstituted with either 92 mL Sterile Water for Injection or 0.9% Sodium Chloride Injection. The diluent should be added in two separate aliquots in a suitable work area, such as a laminar flow hood. Add 50 mL of solution, shake to dissolve. Then add an additional 42 mL and shake. The solution should be allowed to stand after dissolution to allow any foaming to dissipate in order to permit visual inspection for complete solubilization. The resultant solution will have a final concentration of approximately 100 mg/mL ampicillin and 50 mg/mL sulbactam. The closure may be penetrated only one time after reconstitution, if needed, using a suitable sterile transfer device or dispensing set that allows for measured dispensing of the contents.
After reconstitution, use within two hours if stored at room temperature, or within four hours if stored under refrigeration.
Reconstituted Bulk Solution Should Not be Used For Direct Infusion
If the reconstituted bulk solution is stored for less than one hour at room temperature (20°C/68°F) prior to further dilution, the use periods indicated in Table 6 apply for the diluted solutions.
If the bulk solution is stored for one to two hours at room temperature (20°C/68°F) and then diluted with Sterile Water for Injection or 0.9% Sodium Chloride Injection to the following concentrations, the use periods indicated in Table 7 apply.
Any unused portions of solution that remain after the indicated time periods should be discarded.
Table 6 Diluent Maximum Concentration
(mg/mL)
Ampicillin and Sulbactam for Injection (Ampicillin/Sulbactam)Use Periods Sterile Water for Injection
45 (30/15)
45 (30/15)
30 (20/10)8 hrs at 21°C
48 hrs at 4°C
72 hrs at 4°C0.9% Sodium Chloride Injection, (USP)
45 (30/15)
45 (30/15)
30 (20/10)8 hrs at 21°C
48 hrs at 4°C
72 hrs at 4°C5% Dextrose Injection
30 (20/10)
30 (20/10)
3 (2/1)2 hrs at 21°C
4 hrs at 4°C
2 hrs at 21°CLactated Ringer's Injection
45 (30/15)
45 (30/15)8 hrs at 21°C
24 hrs at 4°CM/6 Sodium Lactate Injection
45 (30/15)
45 (30/15)8 hrs at 21°C
12 hrs at 4°C5% Dextrose in 0.45% Saline
3 (2/1)
15 (10/5)4 hrs at 21°C
4 hrs at 4°C10% Invert Sugar
3 (2/1)
30 (20/10)4 hrs at 21°C
3 hrs at 4°CTable 7
IV Solution
Maximum Concentration (mg/mL)
Ampicillin and Sulbactam for Injection (Ampicillin/Sulbactam)
Use Period
Sterile Water for Injection, USP
45 (30/15)
4 hrs at 21°C
45 (30/15)
24 hrs at 4°C
0.9% Sodium Chloride Injection, USP
45 (30/15)
4 hrs at 21°C
45 (30/15)
24 hrs at 4°C
Animal Pharmacology
While reversible glycogenosis was observed in laboratory animals, this phenomenon was dose- and time-dependent and is not expected to develop at the therapeutic doses and corresponding plasma levels attained during the relatively short periods of combined ampicillin/sulbactam therapy in man.
-
HOW SUPPLIED
Ampicillin and Sulbactam for Injection, USP, a sterile white to off-white dry powder, is available in Pharmacy Package bottle containing ampicillin sodium and sulbactam sodium equivalent to 10 g ampicillin and 5 g sulbactam × 1 (NDC: 67457-649-10).
Ampicillin and Sulbactam for Injection, USP sterile powder is to be stored at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]prior to reconstitution.
-
SPL UNCLASSIFIED SECTION
Clinitest™ is a registered trademark of Miles, Inc.
Clinistix™ is a registered trademark of Bayer Corporation.
Testape™ is a registered trademark of Eli Lilly Company.
Rx only
Manufactured for:
Mylan Institutional LLC
Rockford, IL 61103 U.S.A.
Manufactured by:
MITIM S.r.l.
Via Cacciamali n°34-36-38 25125 Brescia, ITALY (premises)
Via Rodi n° 27 Brescia, ITALY (headquarters)
JUNE 2018
-
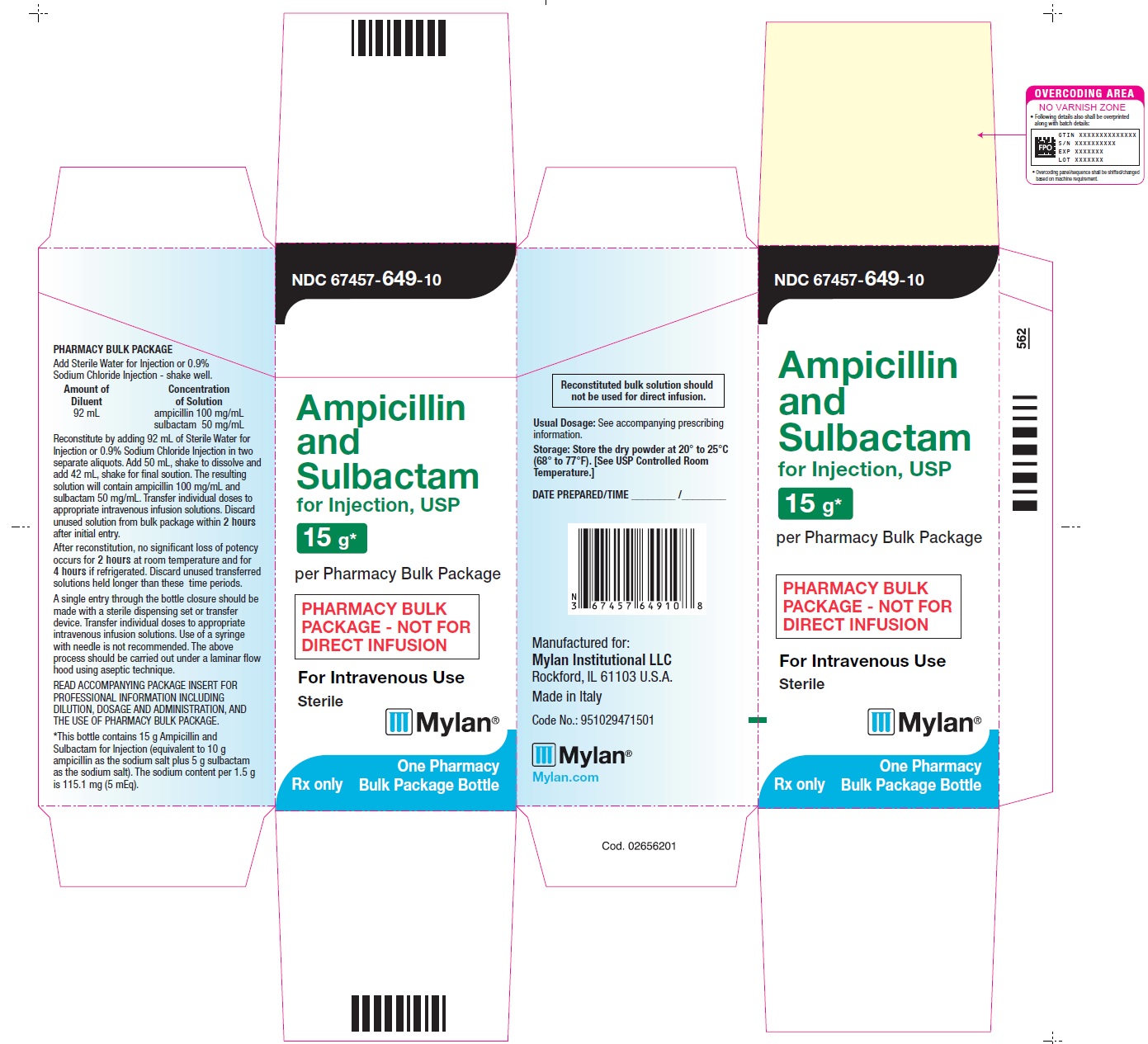
Package/Label Display Panel
NDC: 67457-649-10
Ampicillin and Sulbactam for Injection, USP
15 g* per Pharmacy Bulk Package
PHARMACY BULK PACKAGE - NOT FOR DIRECT INFUSION
Sterile
For Intravenous Use
Rx only
One Pharmacy Bulk Package Bottle
-
INGREDIENTS AND APPEARANCE
AMPICILLIN SODIUM AND SULBACTAM SODIUM
ampicillin sodium and sulbactam sodium injection, powder, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 67457-649 Route of Administration INTRAMUSCULAR, INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AMPICILLIN SODIUM (UNII: JFN36L5S8K) (AMPICILLIN - UNII:7C782967RD) AMPICILLIN 100 mg in 1 mL SULBACTAM SODIUM (UNII: DKQ4T82YE6) (SULBACTAM - UNII:S4TF6I2330) SULBACTAM 50 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 67457-649-10 1 in 1 CARTON 04/08/2014 1 92 mL in 1 VIAL, PHARMACY BULK PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA202197 04/08/2014 Labeler - Mylan Institutional LLC (790384502)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.