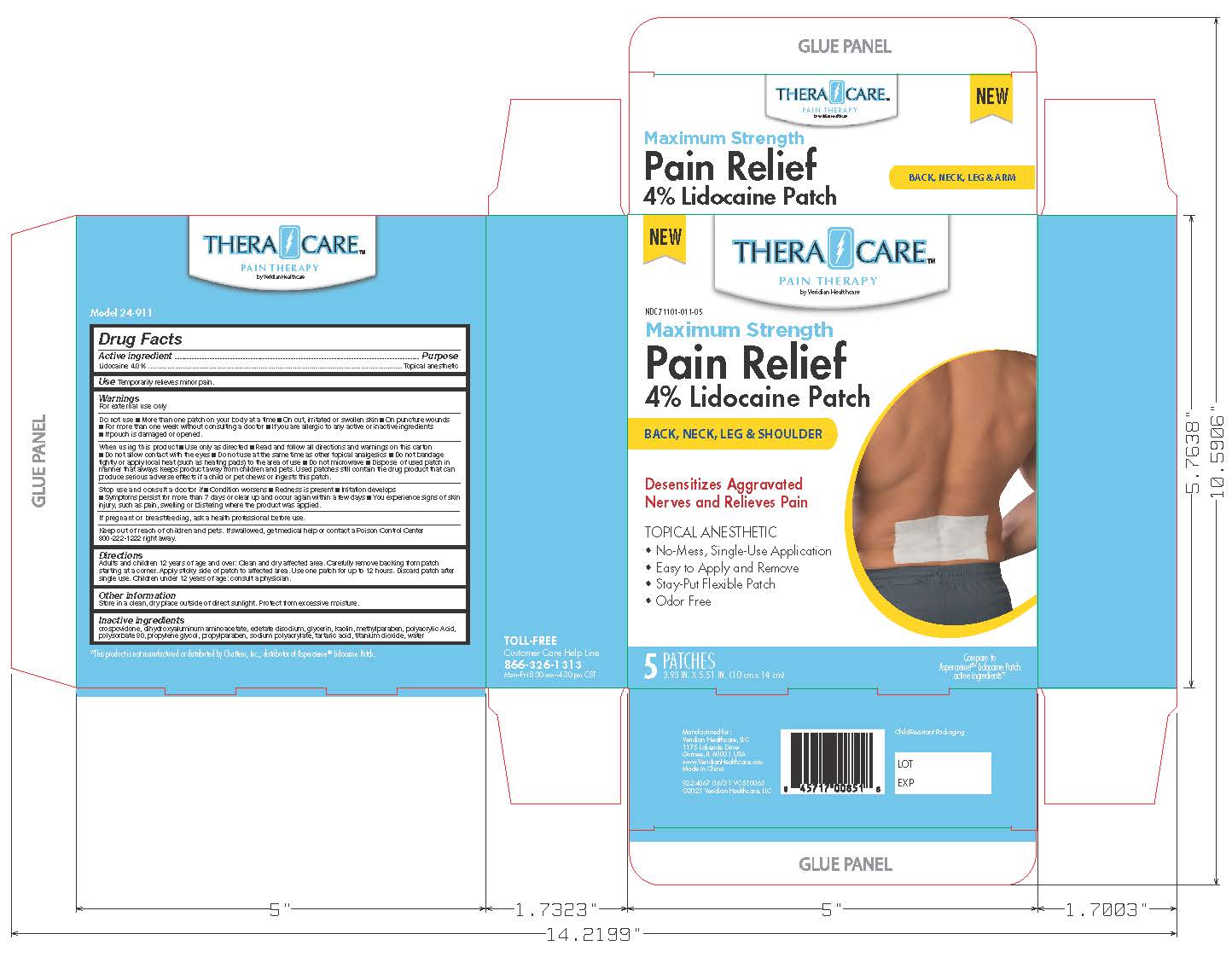
THERACARE PAIN RELIEF LIDOCAINE- lidocaine patch
TheraCare Pain Relief Lidocaine by
Drug Labeling and Warnings
TheraCare Pain Relief Lidocaine by is a Otc medication manufactured, distributed, or labeled by Veridian Healthcare, Shanghai Chuangshi Medical Technology (Group) Co., Ltd.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active Ingredients
- Purpose
- Idications and Usage
- Warnings
- Do not Use
-
When using this product
■ use only as directed
■ read and follow all directions and warnings on this carton
■ do not allow contact with the eyes
■ do not use at the same time as other topical analgesics
■ do not bandage tightly or apply local heat (such as heating pads) to the area of use
■ do not microwave
■ dispose of used patch in manner that always keeps product away from children and pets. Used patches still contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch.
- Stop use and consult a doctor
- PREGNANCY OR BREAST FEEDING
- Keep out of reach of children and pets.
- Dosage and Administration
- Other Safety Information
- Inactive Ingredients
- Questions
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
THERACARE PAIN RELIEF LIDOCAINE
lidocaine patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 71101-011 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 4 g in 100 g Inactive Ingredients Ingredient Name Strength DIHYDROXYALUMINUM AMINOACETATE ANHYDROUS (UNII: 1K713C615K) GLYCERIN (UNII: PDC6A3C0OX) KAOLIN (UNII: 24H4NWX5CO) METHYLPARABEN (UNII: A2I8C7HI9T) POLYACRYLIC ACID (250000 MW) (UNII: 9G2MAD7J6W) POLYSORBATE 80 (UNII: 6OZP39ZG8H) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) CROSPOVIDONE (UNII: 2S7830E561) SODIUM POLYACRYLATE (8000 MW) (UNII: 285CYO341L) TARTARIC ACID (UNII: W4888I119H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) WATER (UNII: 059QF0KO0R) EDETATE DISODIUM (UNII: 7FLD91C86K) Product Characteristics Color Score Shape RECTANGLE Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 71101-011-05 5 in 1 CARTON 10/01/2021 1 9 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 10/01/2021 Labeler - Veridian Healthcare (830437997) Establishment Name Address ID/FEI Business Operations Shanghai Chuangshi Medical Technology (Group) Co., Ltd. 546872672 manufacture(71101-011)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.