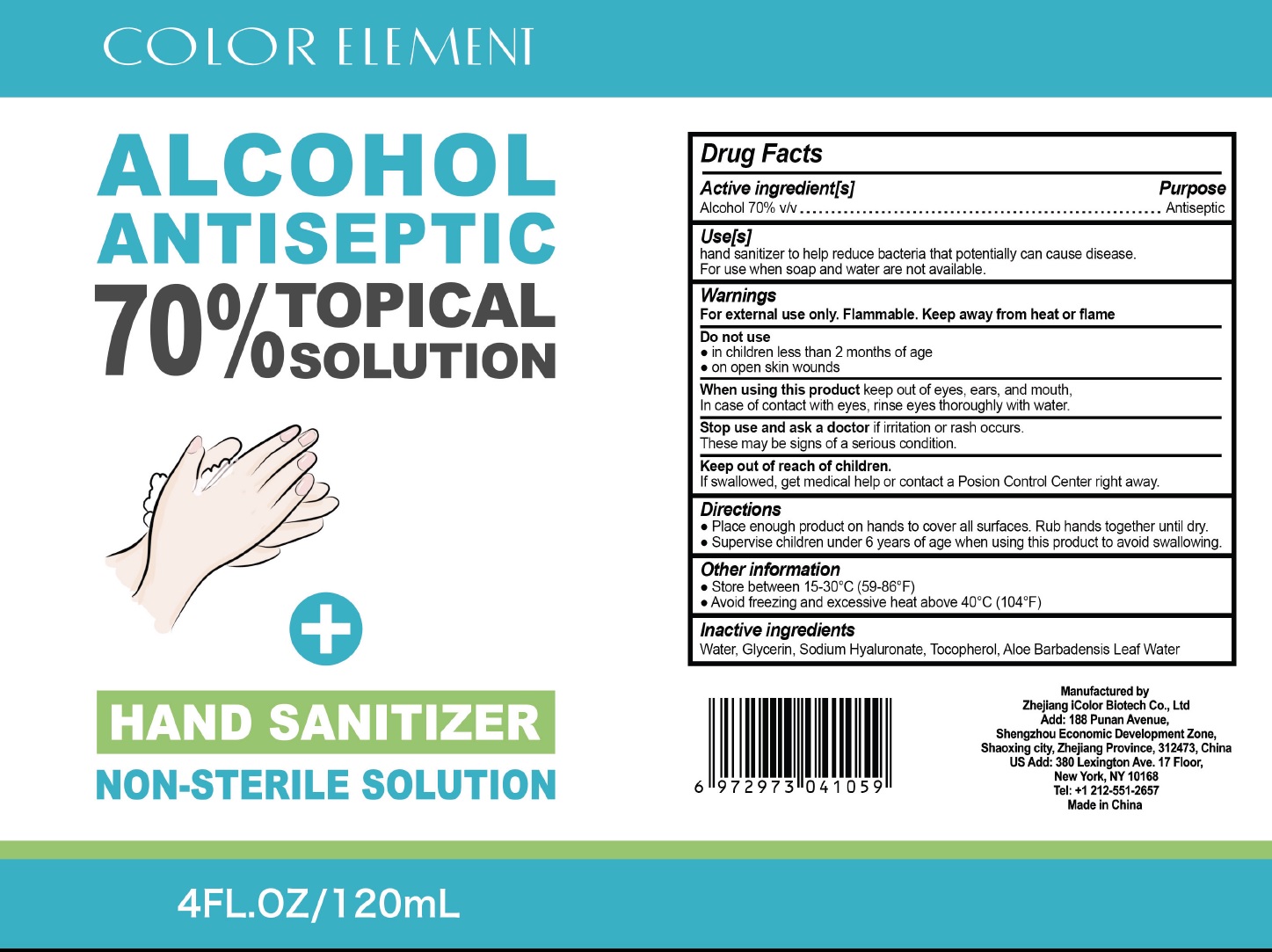
COLOR ELEMENT Alcohol Antiseptic Hand Sanitizer 70% Solution
COLOR ELEMENT Alcohol Antiseptic Hand Sanitizer 70 Solution by
Drug Labeling and Warnings
COLOR ELEMENT Alcohol Antiseptic Hand Sanitizer 70 Solution by is a Otc medication manufactured, distributed, or labeled by Zhejiang iColor Biotech Co., Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
COLOR ELEMENT ALCOHOL ANTISEPTIC HAND SANITIZER 70 SOLUTION- alcohol liquid
Zhejiang iColor Biotech Co., Ltd
----------
COLOR ELEMENT Alcohol Antiseptic Hand Sanitizer 70% Solution
Use[s]
hand sanitizer to help reduce bacteria that potentially can cause disease. For use when soap and water are not available.
Warnings
For external use only. Flammable. Keep away from heat or flame
Directions
- Place enough product on hands to cover all surfaces. Rub hands together until dry.
- Supervise children under 6 years of age when using this product to avoid swallowing.
| COLOR ELEMENT ALCOHOL ANTISEPTIC HAND SANITIZER 70 SOLUTION
alcohol liquid |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Zhejiang iColor Biotech Co., Ltd (554528308) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Zhejiang iColor Biotech Co., Ltd | 554528308 | manufacture(74934-019) | |
Revised: 1/2024
Document Id: 0fa01f65-1787-233b-e063-6294a90a62fb
Set id: cf332df0-caa5-4d87-acc1-2abddf007abe
Version: 2
Effective Time: 20240123