DOLLAR GENERAL MAXIMUM STRENGTH URINARY PAIN RELIEF- phenazopyridine hydrochloride tablet
Dollar General Maximum Strength Urinary Pain Relief by
Drug Labeling and Warnings
Dollar General Maximum Strength Urinary Pain Relief by is a Otc medication manufactured, distributed, or labeled by Dolgencorp, Inc., Reese Pharmaceutical Co. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
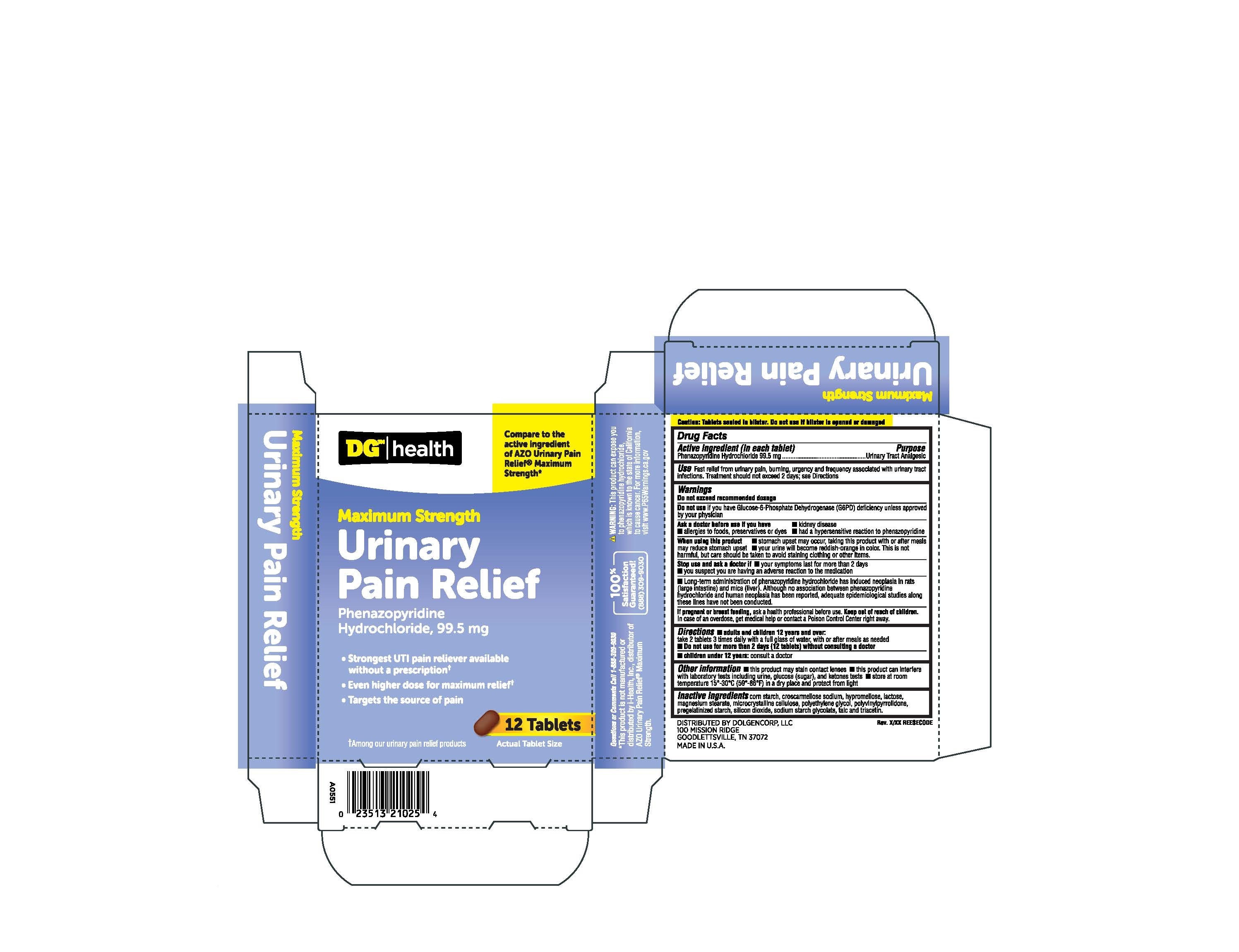
- Active ingredient (in each tablet)
- Purpose
- Warnings
- Ask doctor before use if you have
- When using this product
- Stop use and ask doctor if
- If pregnant or breast feeding,
- Keep out of reach of children
- Use
- Inactive ingredients
- Directions
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DOLLAR GENERAL MAXIMUM STRENGTH URINARY PAIN RELIEF
phenazopyridine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 55910-631 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENAZOPYRIDINE HYDROCHLORIDE (UNII: 0EWG668W17) (PHENAZOPYRIDINE - UNII:K2J09EMJ52) PHENAZOPYRIDINE HYDROCHLORIDE 99.5 mg Inactive Ingredients Ingredient Name Strength LACTOSE (UNII: J2B2A4N98G) MAGNESIUM SILICATE (UNII: 9B9691B2N9) Product Characteristics Color brown Score no score Shape OVAL Size 9mm Flavor Imprint Code p99 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 55910-631-12 1 in 1 CARTON 10/03/2019 1 12 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC: 55910-631-24 2 in 1 CARTON 03/12/2020 2 12 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/03/2019 Labeler - Dolgencorp, Inc. (068331990) Registrant - Reese Pharmaceutical Co (004172052) Establishment Name Address ID/FEI Business Operations Reese Pharmaceutical Co 004172052 relabel(55910-631) , repack(55910-631)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.