TOBRADEX ST- tobramycin / dexamethasone suspension/ drops
TOBRADEX ST by
Drug Labeling and Warnings
TOBRADEX ST by is a Prescription medication manufactured, distributed, or labeled by Alcon Laboratories, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use TOBRADEX® ST ophthalmic suspension safely and effectively. See full prescribing information for TOBRADEX® ST.
TOBRADEX® ST (tobramycin / dexamethasone ophthalmic suspension) 0.3%/0.05%
Initial U.S. Approval: 1988INDICATIONS AND USAGE
TOBRADEX® ST is a topical antibiotic and corticosteroid combination for steroid-responsive inflammatory ocular conditions for which a corticosteroid is indicated and where superficial bacterial ocular infection or a risk of bacterial ocular infection exists. (1)
DOSAGE AND ADMINISTRATION
Instill one drop into the conjunctival sac(s) every 4 to 6 hours. (2.1)
During the initial 24 to 48 hours, dosage may be increased to one drop every 2 hours. (2.1)
Frequency should be decreased gradually as warranted by improvement in clinical signs, but care should be taken not to discontinue therapy prematurely. (2.1)
DOSAGE FORMS AND STRENGTHS
TOBRADEX ST ophthalmic suspension contains 3 mg/mL tobramycin and 0.5 mg/mL dexamethasone. (3)
CONTRAINDICATIONS
TOBRADEX ST, as with other ophthalmic corticosteroids, is contraindicated in most viral diseases of the cornea and conjunctiva including epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, and varicella, and also in mycobacterial infection of the eye and fungal diseases of ocular structures. (4.1)
Hypersensitivity to any component of the medication (4.2)
WARNINGS AND PRECAUTIONS
Intraocular pressure (IOP) increase-Prolonged use of corticosteroids may result in glaucoma with damage to the optic nerve, defects in visual acuity and fields of vision. If this product is used for 10 days or longer, IOP should be monitored. (5.1).
Sensitivity to topically applied aminoglycosides may occur. (5.2)
Cataracts- Use of corticosteroids may result in posterior subcapsular cataract formation. (5.3)
Delayed healing- The use of steroids after cataract surgery may delay healing and increase the incidence of bleb formation. In those diseases causing thinning of the cornea or sclera, perforations have been known to occur with the use of topical steroids. The initial prescription and renewal of the medication order should be made by a physician only after examination of the patient with the aid of magnification such as slit lamp biomicroscopy and, where appropriate, fluorescein staining. (5.4)
Bacterial infections- Prolonged use of corticosteroids may suppress the host response and thus increase the hazard of secondary ocular infections. In acute purulent conditions, steroids may mask infection or enhance existing infection. If signs and symptoms fail to improve after 2 days, the patient should be re-evaluated. (5.5)
Viral infections- Employment of a corticosteroid medication in the treatment of patients with a history of herpes simplex requires great caution. Use of ocular steroids may prolong the course and may exacerbate the severity of many viral infections of the eye (including herpes simplex). (5.6)
Fungal infections- Fungal infections of the cornea are particularly prone to develop coincidentally with long-term local steroid application. Fungus invasion must be considered in any persistent corneal ulceration where a steroid has been used or is in use. (5.7)
If product is used in combination with systemic aminoglycoside antibiotics the patient should be monitored for total serum concentration of tobramycin. (5.8)
ADVERSE REACTIONS
Most common adverse reactions to topical ocular tobramycin are hypersensitivity and localized ocular toxicity, including eye pain, eyelids pruritus, eyelid edema, and conjunctival hyperemia. The reactions due to the steroid component are increases in intraocular pressure with possible development of glaucoma. (6)
(6)
To report SUSPECTED ADVERSE REACTIONS, contact Alcon Laboratories, Inc. at 1-800-757-9195 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. (6)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 7/2011
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosing
2.2 Prescription Guideline
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
4.1 Nonbacterial Etiology
4.2 Hypersensitivity
5 WARNINGS AND PRECAUTIONS
5.1 IOP increase
5.2 Aminoglycoside sensitivity
5.3 Cataracts
5.4 Delayed Healing
5.5 Bacterial infections
5.6 Viral infections
5.7 Fungal infections
5.8 Use with systemic aminoglycosides
6 ADVERSE REACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Nursing Mothers
8.3 Pediatric Use
8.4 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
17.1 Storage and Handling
17.2 Avoid Contamination
17.3 Contact Lens Wear
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
TOBRADEX® ST ophthalmic suspension is indicated for steroid-responsive inflammatory ocular conditions for which a corticosteroid is indicated and where superficial bacterial ocular infection or a risk of bacterial ocular infection exists.
Ocular steroids are indicated in inflammatory conditions of the palpebral and bulbar conjunctiva, cornea and anterior segment of the globe where the inherent risk of steroid use in certain infective conjunctivitides is accepted to obtain a diminution in edema and inflammation. They are also indicated in chronic anterior uveitis and corneal injury from chemical, radiation or thermal burns, or penetration of foreign bodies.
The use of a combination drug with an anti-infective component is indicated where the risk of superficial ocular infection is high or where there is an expectation that potentially dangerous numbers of bacteria will be present in the eye.
The particular anti-infective drug in this product is active against the following common bacterial eye pathogens: Staphylococci, including S. aureus and S. epidermidis (coagulase-positive and coagulase-negative), including penicillin-resistant isolates. Streptococci, including some Group A and other beta-hemolytic species, some nonhemolytic species, and some Streptococcus pneumoniae, Pseudomonas aeruginosa, Escherichia coli, Klebsiella pneumoniae, Enterobacter aerogenes, Proteus mirabilis, Morganella morganii, most Proteus vulgaris isolates, Haemophilus influenzae, H. aegyptius, Moraxella lacunata, Acinetobacter calcoaceticus and some Neisseria species.
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosing
Instill one drop into the conjunctival sac(s) every four to six hours. During the initial 24 to 48 hours, the dosage may be increased to one drop every two hours. Frequency should be decreased gradually as warranted by improvement in clinical signs. Care should be taken not to discontinue therapy prematurely.
2.2 Prescription Guideline
Not more than 20 mL should be prescribed initially and the prescription should not be refilled without further evaluation as outlined in WARNINGS AND PRECAUTIONS (5).
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
4.1 Nonbacterial Etiology
TOBRADEX ST, as with other ophthalmic corticosteroids, is contraindicated in most viral diseases of the cornea and conjunctiva including epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, and varicella, and also in mycobacterial infection of the eye and fungal diseases of ocular structures. (4.1)
-
5 WARNINGS AND PRECAUTIONS
5.1 IOP increase
Prolonged use of corticosteroids may result in glaucoma with damage to the optic nerve, defects in visual acuity and fields of vision. If this product is used for 10 days or longer, IOP should be monitored.
5.4 Delayed Healing
The use of steroids after cataract surgery may delay healing and increase the incidence of bleb formation. In those diseases causing thinning of the cornea or sclera, perforations have been known to occur with the use of topical steroids. The initial prescription and renewal of the medication order should be made by a physician only after examination of the patient with the aid of magnification such as slit lamp biomicroscopy and, where appropriate, fluorescein staining.
5.5 Bacterial infections
Prolonged use of corticosteroids may suppress the host response and thus increase the hazard of secondary ocular infections. In acute purulent conditions, steroids may mask infection or enhance existing infection. If signs and symptoms fail to improve after 2 days, the patient should be re-evaluated.
5.6 Viral infections
Employment of a corticosteroid medication in the treatment of patients with a history of herpes simplex requires great caution. Use of ocular steroids may prolong the course and may exacerbate the severity of many viral infections of the eye (including herpes simplex).
-
6 ADVERSE REACTIONS
Adverse reactions have occurred with steroid/anti-infective combination drugs which can be attributed to the steroid component, the anti-infective component, or the combination. Exact incidence figures are not available.
The most frequent adverse reactions to topical ocular tobramycin (TOBREX®) are hypersensitivity and localized ocular toxicity, including eye pain, eyelids pruritus, eyelid edema, and conjunctival hyperemia. These reactions occur in less than 4% of patients. Similar reactions may occur with the topical use of other aminoglycoside antibiotics.
The reactions due to the steroid component are: increased intraocular pressure (IOP) with possible development of glaucoma, and infrequent optic nerve disorder; subcapsular cataract; and impaired healing.
Secondary Infection.
The development of secondary infection has occurred after use of combinations containing steroids and antimicrobials. Fungal infections of the cornea are particularly prone to develop coincidentally with long-term applications of steroids. The possibility of fungal invasion must be considered in any persistent corneal ulceration where steroid treatment has been used. Secondary bacterial ocular infection following suppression of host responses also occurs.
Non-ocular adverse events occurring at an incidence of 0.5% to 1% included headache and increased blood pressure.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnanacy Category C. Corticosteroids have been shown to be teratogenic in animal studies. Ocular administration of 0.1% dexamethasone resulted in 15.6% and 32.3% incidence of fetal anomalies in two groups of pregnant rabbits. Fetal growth retardation and increased mortality rates have been observed in rats with chronic dexamethasone therapy. Reproduction studies have been performed in rats and rabbits with tobramycin at doses up to 100 mg/kg/day (equivalent to human doses of 16 and 32 mg/kg/day, respectively) and have revealed no evidence of impaired fertility or harm to the fetus. There are no adequate and well controlled studies in pregnant women. TOBRADEX® ST ophthalmic suspension should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
8.2 Nursing Mothers
Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in human milk. Because many drugs are excreted in human milk, caution should be exercised when TOBRADEX® ST is administered to a nursing woman.
-
11 DESCRIPTION
TOBRADEX® ST (tobramycin / dexamethasone ophthalmic suspension) 0.3%/0.05% is a sterile, isotonic, white, aqueous antibiotic and steroid suspension with a pH of approximately 5.7 and an osmolality of approximately 290 mOsm/kg.
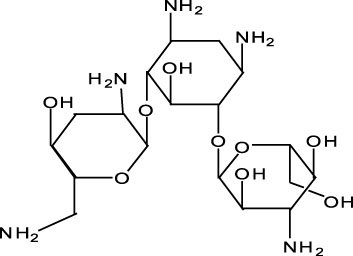
The chemical name of tobramycin is O-3-amino-3-deoxy-α-D-glucopyranosyl-(14)-O-[2,6-diamino-2,3,6-trideoxy-α-D-ribo-hexopyranosyl-(16)]-2-deoxy- L-streptamine. It has a molecular formula of C18H37N5O9 and a molecular weight of 467.52. The chemical structure is:
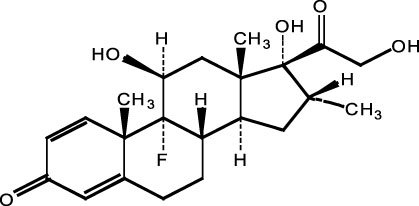
The chemical name of dexamethasone is 9-fluoro-11β,17,21-trihydroxy-16α-methylpregna-1,4-diene-3,20-dione. It has a molecular formula of C22H29FO5 and a molecular weight of 392.47. The chemical structure is:
Each mL of TOBRADEX® ST contains: Actives: tobramycin 3 mg and dexamethasone 0.5 mg. Preservative: benzalkonium chloride 0.1 mg. Inactives: xanthan gum, tyloxapol, edetate disodium, sodium chloride, propylene glycol, sodium sulfate, hydrochloric acid and/or sodium hydroxide (to adjust pH) and purified water.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Dexamethasone is a potent corticoid. Corticoids suppress the inflammatory response to a variety of agents and they can delay or slow healing. Since corticoids may inhibit the body's defense mechanism against infection, a concomitant antimicrobial drug may be used when this inhibition is considered to be clinically significant.
Tobramycin is an antibacterial drug. It inhibits the growth of bacteria by inhibiting protein synthesis. Tobramycin is included in this combination product to provide action against susceptible bacteria.
12.3 Pharmacokinetics
In a multi-center, double-masked, parallel-group, randomized, single-dose pharmacokinetic study in male and female cataract surgery patients, mean dexamethasone concentrations following administration of TOBRADEX ST were similar to dexamethasone concentrations following administration of TOBRADEX (tobramycin /dexamethasone ophthalmic suspension) 0.3%/0.1%. Aqueous humor concentrations reached a mean peak of 33.7 ng/mL 2 hours following single-dose administration of TOBRADEX ST.
No data are available on the extent of systemic absorption of dexamethasone or tobramycin from TOBRADEX ST ophthalmic suspension. Following multiple-dose (QID for 2 days) bilateral ocular administration of TOBRADEX (Tobramycin 0.3%/Dexamethasone 0.1% ophthalmic suspension) in healthy male and female volunteers, peak plasma concentrations of dexamethasone were less than 1 ng/mL and occurred within 2 hours post-dose across all subjects.
12.4 Microbiology
The antibiotic component (tobramycin) in the combination is included to provide action against susceptible bacteria. In vitro studies have demonstrated that tobramycin is active against susceptible isolates of the following bacteria: Staphylococcus aureus (includes penicillin-resistant isolates), Staphylococcus epidermidis (includes penicillin-resistant isolates), Streptococci, including some Group A other beta-hemolytic species, some nonhemolytic species, and some Streptococcus pneumoniae. Acinetobacter calcoaceticus, Enterobacter aerogenes, Escherichia coli, Haemophilus influenzae, Haemophilus aegypticus, Klebsiella pneumoniae, Moraxella lacunata, Morganella morganii, Neisseria perflava, Neisseria sicca, Proteus mirabilis, Proteus vulgaris, Pseudomonas aeruginosa.
In vitro bacterial studies demonstrate that in some cases bacteria resistant to gentamicin are susceptible to tobramycin.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been conducted to evaluate the carcinogenic or mutagenic potential. No impairment of fertility was noted in studies of subcutaneous tobramycin in rats at doses of 50 and 100 mg/kg/day (equivalent to human doses of 8 and 16 mg/kg/day, at least 2 orders of magnitude greater than the topical ocular dose).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
TOBRADEX® ST is supplied as a 2.5 mL, 5 mL, or 10 mL suspension in a 4 mL, 8 mL or 10 mL natural polyethylene DROP-TAINER® bottle with a natural polyethylene dispenser tip and a pink polypropylene overcap. Tamper evidence is provided with a shrink band around the closure and neck area of the bottle.
NDC: 0065-0652-25: 2.5 mL
NDC: 0065-0652-05: 5 mL
NDC: 0065-0652-10: 10 mL
Storage
Store at 2° to 25°C (36° to 77°F).
Protect from light.
- 17 PATIENT COUNSELING INFORMATION
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TOBRADEX ST
tobramycin / dexamethasone suspension/ dropsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0065-0652 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TOBRAMYCIN (UNII: VZ8RRZ51VK) (TOBRAMYCIN - UNII:VZ8RRZ51VK) TOBRAMYCIN 3 mg in 1 mL DEXAMETHASONE (UNII: 7S5I7G3JQL) (DEXAMETHASONE - UNII:7S5I7G3JQL) DEXAMETHASONE 0.5 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) XANTHAN GUM (UNII: TTV12P4NEE) TYLOXAPOL (UNII: Y27PUL9H56) EDETATE DISODIUM (UNII: 7FLD91C86K) SODIUM CHLORIDE (UNII: 451W47IQ8X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM SULFATE (UNII: 0YPR65R21J) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0065-0652-25 2.5 mL in 1 BOTTLE; Type 0: Not a Combination Product 09/07/2010 09/07/2010 2 NDC: 0065-0652-05 1 in 1 CARTON 09/07/2010 2 5 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 3 NDC: 0065-0652-10 10 mL in 1 BOTTLE; Type 0: Not a Combination Product 09/07/2010 09/07/2010 4 NDC: 0065-0652-01 1 in 1 CARTON 09/09/2010 4 1 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA050818 09/07/2010 Labeler - Alcon Laboratories, Inc. (008018525)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.