EPINEPHrine Chloride Solution(Epinephrine Nasal Solution, USP)10mg/10mL (1 mg/mL)Vasoconstrictor For Topical ApplicationRx Only
Epinephrine Chloride Solution by
Drug Labeling and Warnings
Epinephrine Chloride Solution by is a Prescription medication manufactured, distributed, or labeled by BPI Labs, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
EPINEPHRINE CHLORIDE SOLUTION- epinephrine nasal solution solution
BPI Labs, LLC
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
EPINEPHrine Chloride Solution
(Epinephrine Nasal Solution, USP)
10mg/10mL (1 mg/mL)
Vasoconstrictor For Topical Application
Rx Only
1 INDICATIONS AND USAGE
For use as a nasal decongestant. Apply locally as drops or spray, or with a sterile swab, as required.
2 DOSAGE AND ADMINISTRATION
Instructions for diluting
| To make Epinephrine Chloride Solution | Epinephrine Chloride Solution 1 mg/mL | Isotonic Salt Solution |
| 0.5 mg/mL | 1 mL | 1 mL |
| 0.2 mg/mL | 1 mL | 4 mL |
| 0.1 mg/mL | 1 mL | 9 mL |
Note: Do not use the solution if it is pinkish or darker than yellow or if it contains a precipitate.
3 WARNINGS AND PRECAUTIONS
Caution: Administer with caution to elderly people. Do not use if high blood pressure, heart disease, diabetes, or thyroid disease is present unless directed by a physician. If prompt relief is not obtained, consult a physician. Administer to pediatric patients under six years of age only on the advice of a physician.
4 DESCRIPTION
Each mL contains 1 mg Epinephrine as the hydrochloride, dissolved in isotonic sodium chloride solution with 0.5% chlorobutanol (chloroform derivative) as a preservative and not more than 0.05% sodium metabisulfite as an antioxidant.
EPINEPHrine nasal solution, USP must not be brought into contact with oxidizing agents.The solution is also quickly decomposed by iron salts and alkalies.
5 ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact BPI Labs, LLC at (727) 471-0850 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
6 HOW SUPPLIED/STORAGE AND HANDLING
Each carton contains one vial of 10 mg/10 mL (1 mg/mL) of Epinephrine Chloride Solution (Epinephrine Nasal Solution,USP) in an amber glass vial.
NDC: 54288-123-01 10 mL Vial
This product is sterile until opened.
Keep this and all drugs out of the reach of children.
Store between 20° to 25°C (68° to 77°F). (See USP Controlled Room Temperature.) Do not refrigerate or freeze, Protect from light.
Do not use if cap seal is broken or missing.
Manufactured by:
BPI Labs, LLC
12393 Belcher Rd S, Suite 450
Largo, FL 33773
PACKAGE LABEL PRINCIPAL DISPLAY PANEL
Carton:
Epinephrine Chloride Solution
(Epinephrine Nasal Solution, USP)
10 mg/10 mL (1 mg/mL)
Vasoconstrictor for Topical Application
10 mL Vial
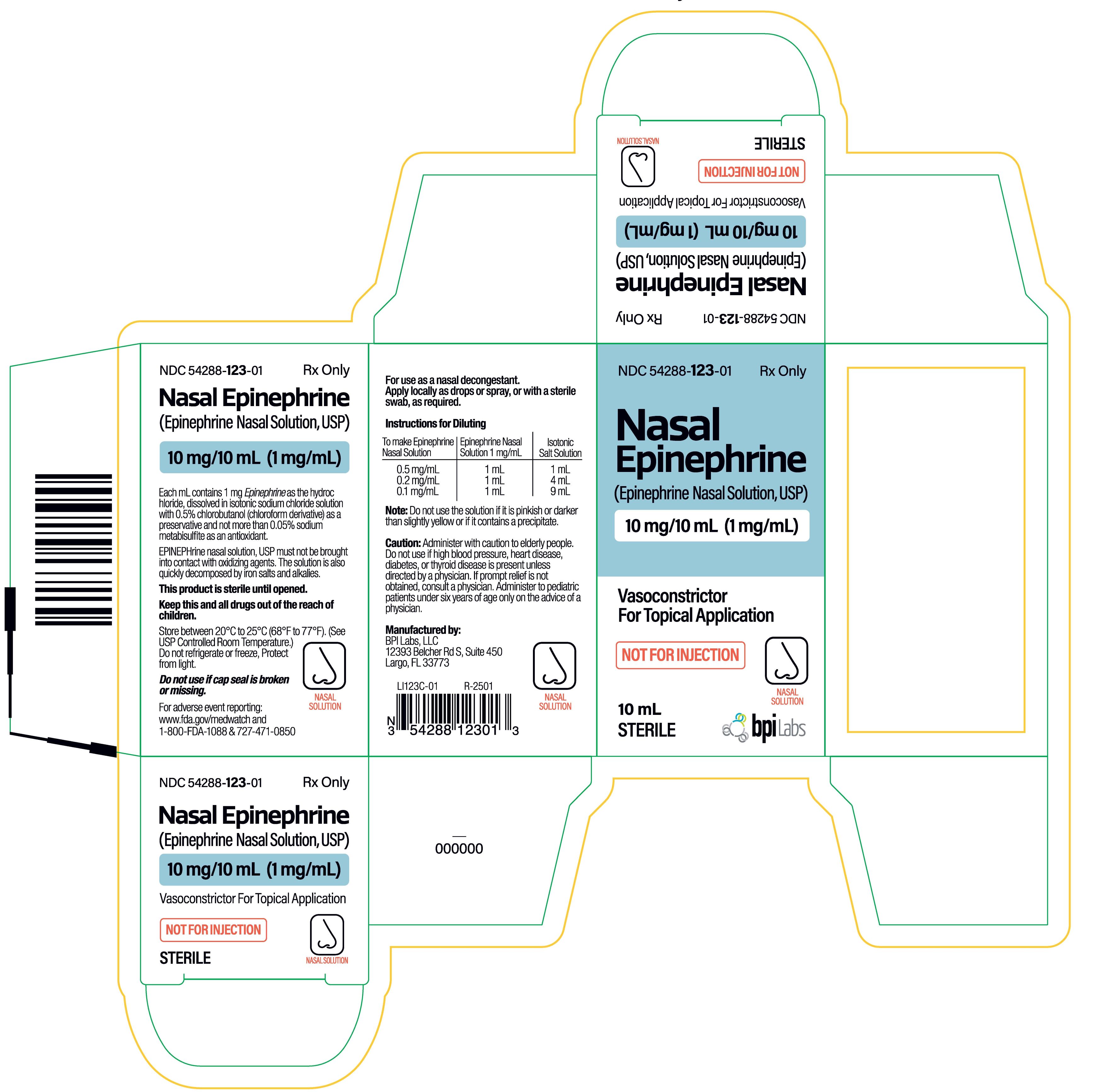
Label:
Epinephrine Chloride Solution
(Epinephrine Nasal Solution,USP)
10 mg/10 mL (1 mg/mL)
Vasoconstrictor for Topical Application
10 mL Vial

| EPINEPHRINE CHLORIDE SOLUTION
epinephrine nasal solution solution |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - BPI Labs, LLC (078627620) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| BPI Labs, LLC | 078627620 | manufacture(54288-123) , label(54288-123) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.