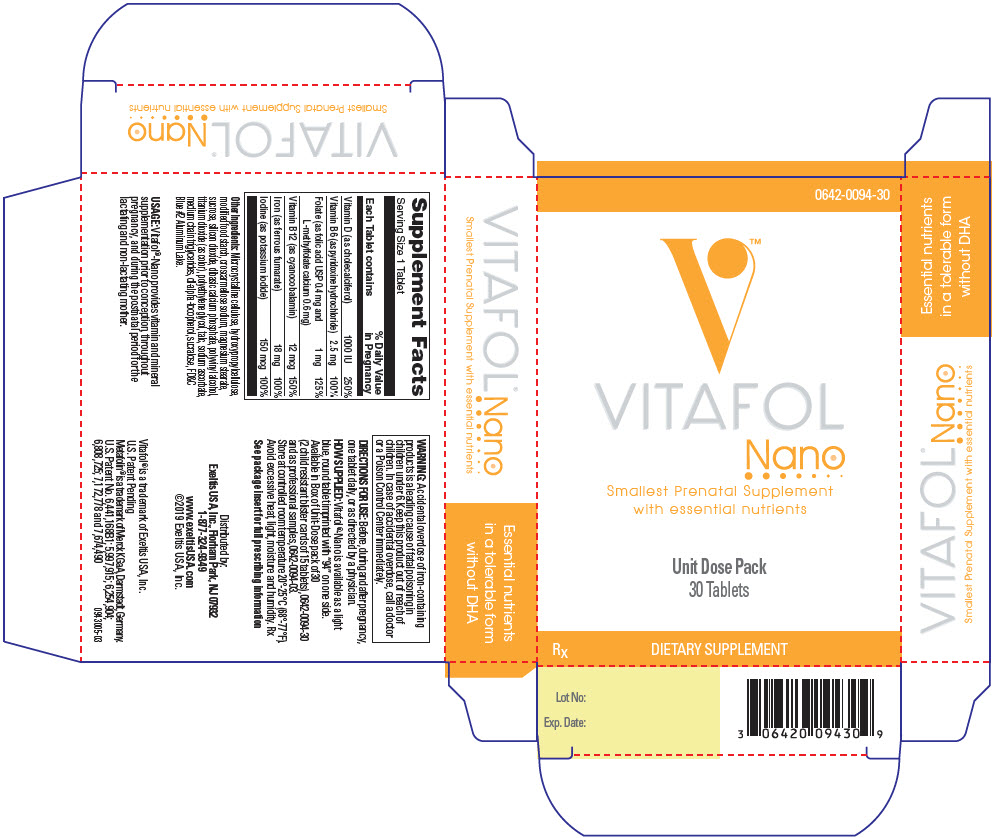
VITAFOL NANO- cholecalciferol, pyridoxine hydrochloride, folic acid, levomefolate calcium, cyanocobalamin, iron, and iodine tablet, coated
Vitafol by
Drug Labeling and Warnings
Vitafol by is a Prescription medication manufactured, distributed, or labeled by Exeltis USA, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
-
COMPOSITION
Amount per Tablet:
VITAMINS AND MINERALS
Vitamin D (as cholecalciferol) 1000 IU Vitamin B6 (as pyridoxine hydrochloride) 2.5 mg Folate (as Folic acid USP 0.4 mg and L-methylfolate calcium 0.6 mg, as Metafolin® CAS# 151533-22-1) 1 mg Vitamin B12 (as cyanocobalamin) 12 mcg Iron (as ferrous fumarate) 18 mg Iodine (as potassium iodide) 150 mcg Other Ingredients
Microcrystalline cellulose, hydroxypropylcellulose, modified food starch, croscarmellose sodium, magnesium stearate, sucrose, silicon dioxide, dibasic calcium phosphate, polyvinyl alcohol, titanium dioxide (as color), polyethylene glycol, talc, sodium ascorbate, medium chain triglycerides, dl-alpha-tocopherol, sucralose, FD&C Blue #2 Aluminum Lake.
- USAGE
-
CONTRAINDICATIONS
Vitafol®-Nano is contraindicated in patients with hypersensitivity to any of its components or color additives.
Folic acid is contraindicated in patients with untreated and uncomplicated pernicious anemia, and in those with anaphylactic sensitivity to folic acid.
Iron therapy is contraindicated in patients with hemochromatosis and patients with iron storage disease or the potential for iron storage disease due to chronic hemolytic anemia (e.g., inherited anomalies of hemoglobin structure or synthesis and/or red cell enzyme deficiencies, etc.), pyridoxine responsive anemia, or cirrhosis of the liver.
Cyanocobalamin is contraindicated in patients with sensitivity to cobalt or to cyanocobalamin (vitamin B12).
- BOXED WARNING (What is this?)
-
WARNINGS/PRECAUTIONS
Vitamin D supplementation should be used with caution in those with hypercalcemia or conditions that may lead to hypercalcemia such as hyperparathyroidism and those who form calcium-containing kidney stones. High doses of vitamin D can lead to elevated levels of calcium that reside in the blood and soft tissues. Bone pain, high blood pressure, formation of kidney stones, renal failure, and increased risk of heart disease can occur.
Prolonged use of iron salts may produce iron storage disease.
Folic acid, especially in doses above 0.1 mg daily, may obscure pernicious anemia, in that hematologic remission may occur while neurological manifestations remain progressive.
The use of folic acid doses above 1 mg daily may precipitate or exacerbate the neurological damage of vitamin B12 deficiency.
Avoid Overdosage. Keep out of the reach of children.
DRUG INTERATIONS
High doses of folic acid may result in decreased serum levels of the anticonvulsant drugs; carbamazepine, fosphenytoin, phenytoin, phenobarbitol, valproic acid. Folic acid may decrease a patient's response to methotrexate.
Vitamin D supplementation should not be given with large amounts of calcium in those with hypercalcemia or conditions that may lead to hypercalcemia such as hyperparathyroidism and those who form calcium-containing kidney stones.
Consult appropriate references for additional specific vitamin-drug interactions.
-
ADVERSE REACTIONS
Adverse reactions have been reported with specific vitamins and minerals, but generally at doses substantially higher than those in Vitafol®-Nano. However, allergic and idiosyncratic reactions are possible at any dose. Reported adverse events include skin ailments, gastrointestinal complaints, glucose abnormalities, and visual problems.
- DIRECTIONS FOR USE
- HOW SUPPLIED
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 30 Tablet Dose Pack Carton
-
INGREDIENTS AND APPEARANCE
VITAFOL NANO
cholecalciferol, pyridoxine hydrochloride, folic acid, levomefolate calcium, cyanocobalamin, iron, and iodine tablet, coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0642-0094 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHOLECALCIFEROL (UNII: 1C6V77QF41) (Cholecalciferol - UNII:1C6V77QF41) CHOLECALCIFEROL 1000 [iU] Pyridoxine Hydrochloride (UNII: 68Y4CF58BV) (Pyridoxine - UNII:KV2JZ1BI6Z) Pyridoxine Hydrochloride 2.5 mg Folic Acid (UNII: 935E97BOY8) (Folic Acid - UNII:935E97BOY8) Folic Acid 0.4 mg Levomefolate Calcium (UNII: A9R10K3F2F) (Levomefolic Acid - UNII:8S95DH25XC) Levomefolate Calcium 0.6 mg Cyanocobalamin (UNII: P6YC3EG204) (Cyanocobalamin - UNII:P6YC3EG204) Cyanocobalamin 12 ug Iron (UNII: E1UOL152H7) (Iron - UNII:E1UOL152H7) Iron 18 mg Iodine (UNII: 9679TC07X4) (Iodine - UNII:9679TC07X4) Iodine 150 ug Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) SUCROSE (UNII: C151H8M554) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) TALC (UNII: 7SEV7J4R1U) SODIUM ASCORBATE (UNII: S033EH8359) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) .ALPHA.-TOCOPHEROL, DL- (UNII: 7QWA1RIO01) SUCRALOSE (UNII: 96K6UQ3ZD4) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) Product Characteristics Color BLUE Score no score Shape ROUND Size 6mm Flavor Imprint Code EV0094 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0642-0094-30 30 in 1 BLISTER PACK; Type 0: Not a Combination Product 07/14/2014 2 NDC: 0642-0094-03 3 in 1 BLISTER PACK; Type 0: Not a Combination Product 07/14/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 07/14/2014 Labeler - Exeltis USA, Inc. (071170534)
Trademark Results [Vitafol]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 VITAFOL 88294273 not registered Live/Pending |
EXELTIS USA INC. 2019-02-08 |
 VITAFOL 86653118 4859841 Live/Registered |
Exeltis USA, Inc. 2015-06-05 |
 VITAFOL 73611865 1452861 Live/Registered |
EVERETT LABORATORIES, INC. 1986-07-28 |
 VITAFOL 72084824 0728602 Live/Registered |
VITA ZAHNFABRIK H. RAUTER, KG 1959-11-06 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.