SOLUCION INTERSOL- solucion aditiva para plaquetas 3 solution
Solucion InterSol by
Drug Labeling and Warnings
Solucion InterSol by is a Prescription medication manufactured, distributed, or labeled by Fenwal, Inc., Fenwal International, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
Spanish Language
Solución InterSol Solución aditiva para plaquetas 3
500 mL
Solamente Rx
Para ser usada exclusivamente con el separador AMICUS.
Almacenar a temperatura ambiente controlada. Proteger de la congelación. Evitar el calor excesivo.
Definición de "Temperatura ambiente controlada":
"Una temperatura mantenida termostáticamente entre 20 °C y 25 °C (68 °F y 77 °F) prevalente por lo general en el ambiente habitual de trabajo, la cual resulta en una temperatura cinética media calculada de no más de 25 °C, y que permite desviaciones de entre 15 °C y 30 °C (59 °F y 86 °F) experimentadas en farmacias, hospitales y almacenes. Se permiten elevaciones pasajeras de temperatura no superiores a los 40 °C, siempre que la temperatura cinética media permanezca en el intervalo permitido y que dichas elevaciones no duren más de 24 horas... La temperatura cinética media es un valor calculado que puede emplearse como la temperatura de almacenamiento isotérmica que simula los efectos no isotérmicos de las variaciones de temperatura de almacenamiento."
Referencia: United States Pharmacopeia, Avisos generales. United States Pharmacopeial Convention, Inc. 12601 Twinbrook Parkway, Rockville, MD.
Desechar los desperdicios en un contenedor apropiado de biopeligrosos o según los requisitos de regulaciones locales.
Indicaciones y uso:
La solución InterSol es una solución isotónica diseñada para sustituir una parte del plasma utilizado en el almacenamiento de plaquetas de aféresis leucorreducidas derivadas de AMICUS en las condiciones estándar de los bancos de sangre. No existe ningún efecto terapéutico directo derivado de la fórmula. La solución nunca se debe infundir directamente en el paciente.
Las plaquetas de InterSol son concentrados de plaquetas de aféresis leucorreducidas que se almacenan en una solución que, nominalmente, contiene 65% de InterSol y 35% de plasma. Las plaqueta de InterSol que se preparan dentro del intervalo indicado en la Tabla 1 pueden almacenarse por hasta 5 días a entre 20 y 24 °C, con agitación continua.
Tabla 1

Dosis y administración:
La solución InterSol solo se puede usar con el sistema separador AMICUS utilizando un kit de aféresis AMICUS con un conector de la solución aditiva de plaquetas (PAS). Para obtener instrucciones completes sobre el uso de la solución InterSol con el separador AMICUS, consulte el Manual del operador de AMICUS.
Antes de usar la solución InterSol, verificar que el contenedor plástico no tenga fugas apretando la bolsa con firmeza. Si existen fugas, desechar la solución.
Para conectar la solución InterSol al kit de aféresis AMICUS:
1) Cerrar la abrazadera de la línea del conector de PAS.
2) Retirar la tapa protectora del puerto de membrana en el contenedor de solución InterSol.
3) Introducir la espiga en la línea del conector de PAS hacia dentro del puerto que tiene una membrana en el contenedor de solución InterSol hasta que quede bien asentada.
4) Para agregar solución InterSol al producto de plaquetas, seguir las instrucciones incluidas en el Manual del operador de AMICUS.
Presentaciones y concentraciones de la dosis:
La solución InterSol viene en la forma de 500 mL de solución apirogéna esterilizada en un contenedor plástico que no es de PVC y que incluye una vía de fluido apirogéna esterilizada.
Cada 100 mL contienen 305 mg de fosfato de sodio dibásico, anhidro, USP; 93 mg de fosfato de sodio monobásico, monohidratado, USP; 318 mg de citrato de sodio, dihidratado, USP; 442 mg de acetato de sodio, trihidratado, USP; 452 mg de cloruro de sodio, USP; agua para inyección, USP, en cantidad suficiente.
Contraindicaciones:
La solución InterSol se añade a los concentrados de plaquetas leucorreducidas derivadas de AMICUS después de haber completado el proceso de aféresis. No es para infusión intravenosa directa. Se desconoce que existan contraindicaciones asociadas con el uso de la solución InterSol para la preparación de plaquetas InterSol.
Advertencias y precauciones:
Solución InterSol NO ES PARA INFUSIÓN INTRAVENOSA DIRECTA.
No utilizar si existen partículas o si la solución está turbia.
No usar si el contenedor está dañado, tiene fugas o muestra signos de deterioro.
No ventilar.
No volver a usar. Desechar la solución InterSol parcialmente usada o sin usar.
Proteger contra objetos puntiagudos.
Comprobar que la solución InterSol se haya acoplado firmemente a la línea del conector PAS a fin de evitar desconexiones y fugas.
Reacciones adversas:
La solución InterSol se añade a los concentrados de plaquetas leucorreducidas derivadas de AMICUS después de haber completado el proceso de aféresis. No es para infusión intravenosa directa. No es de esperarse que la solución InterSol cause reacciones adversas más allá de las que normalmente están asociadas con la transfusión de plaquetas.
Abuso / dependencia de drogas:
La solución InterSol se utiliza como una solución de almacenamiento para los concentrados de plaquetas y no tiene efectos farmacológicos.
Sobredosis:
La solución InterSol se utiliza como una solución de almacenamiento para los concentrados de plaquetas y no es para infusión intravenosa directa.
Descripción:
La solución InterSol es una solución isotónica diseñada para reemplazar una parte del plasma utilizado en el almacenamiento de plaquetas. La solución contiene elementos constituyentes que son de existencia natural y que están presentes en muchos sistemas celulares: acetato de sodio, que actúa como nutriente; citrato de sodio para evitar la activación y la aglomeración de plaquetas; fosfato de sodio que actúa como buffer; y cloruro de sodio para efectos de osmolaridad. La solución InterSol no tiene un efecto farmacológico “in vivo”, sino que tiene la función de proporcionar el entorno y los nutrientes adecuados que sustituyen una parte del plasma que se utiliza normalmente para el almacenamiento de las plaquetas.
Farmacología clínica:
La solución InterSol se utiliza como una solución de almacenamiento para los concentrados de plaquetas y no es para infusión intravenosa directa. Esta solución no tiene efectos farmacológicos.
Estudios clínicos:
Evaluaciones funcionales y bioquímicas "in vitro"
Los concentrados de plaquetas con InterSol (n=70) que se prepararon utilizando el separador AMICUS y se han almacenado por 5 días exhibieron, en el día 5, un pH cuyos valores de media y valor promedio fueron, respectivamente, 7,2 y 7,2 ± 0,1 (el rango fue de 6,9 a 7,5), con un límite inferior no paramétrico de 6,9 con una tolerancia de 95%/95%.
En la Tabla 2 se presentan evaluaciones complementarias "in vitro" de plaquetas con InterSol y de plaquetas leucorreducidas derivadas de AMICUS que están almacenadas en plasma al 100%.
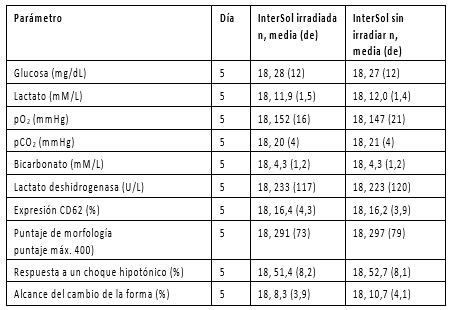
Se realizó una comparación entre plaquetas con InterSol no irradiadas y plaquetas con InterSol irradiadas (n=18) a 2500 cGray o a 2800 cGray (dependiendo de los procedimientos de la institución). Al final del almacenamiento, en el día 5, la producción de plaquetas de los productos irradiados fue de concentrados de entre 2,5 x 1011 y 3,5 x 1011 e incluyó un producto con una concentración <2,5 x 1011 y dos productos con concentraciones >3,5 x 1011. Las pruebas realizadas “in vitro” no predicen el rendimiento “in vivo”, pero esto no fue evaluado. En la Tabla 3 se presenta el resumen de los datos estadísticos.
Recuperación y supervivencia "in vivo" en sujetos sanos
La evaluación "in vivo" de plaquetas con InterSol en el día 5 (n=33), en comparación con un control con plaquetas nuevas dio como resultado una recuperación porcentual promedio de 46,4 ± 11,9 por ciento y 58,0 ± 10,7 por ciento y una supervivencia promedio de 5,7 ± 1,4 días y 8,0 ± 1,4 días, respectivamente. Los datos "in vivo" recolectados fueron utilizados para calcular el límite superior de un intervalo de confianza bilateral de 95% de la recuperación porcentual promedio, expresado por la expresión algebraica (0,66 x nuevas – 5 días), y de la supervivencia porcentual (días), expresado por la expresión (0,58 x nuevas – 5 días). El límite superior de los intervalos de confianza bilaterales de 95% para la recuperación y la supervivencia en el día 5 fue de – 4,6 y de –0,6, respectivamente, y cumplió con el requisito de ser menor que 0.
Estudio de eventos adversos (EA) relacionados con la transfusión después de que el producto ha salido al mercado
Se realizó en 6 instituciones un estudio retrospectivo a etiqueta abierta y no aleatorio de revisión de registros médicos a fin de demostrar que el índice global de EA relacionados con la transfusión en pacientes que reciben transfusiones de plaquetas con InterSol no fue mayor que el doble del índice observado en pacientes que recibieron plaquetas de aféresis almacenadas en plasma al 100% (plaquetas con plasma). El estudio clasificó las reacciones adversas a la transfusión según las definiciones descritas por “Biovigilance Component” del sistema “National Healthcare Safety Network” (NHSN). A todas las instituciones participantes se les exigió tener sistemas instalados para la observación de EA relacionados con la transfusión y para registrar cuando lo observado no era ningún EA relacionado con la transfusión. Un comité independiente de casos clínicos (CEC, Clinical Events Committee), sin ningún conocimiento del tipo de plaquetas y de la institución donde se realizó el estudio, se encargó de adjudicar los eventos registrados.
A los pacientes se les prescribieron transfusiones de plaquetas en acatamiento de la metodología normal de la institución. El tipo de plaqueta que recibió el paciente fue determinado por el inventario de la institución al momento en que se prescribió la transfusión. El personal de la institución observó al receptor de la transfusión de plaquetas en acatamiento de sus procedimientos normales. Los síntomas y las evidencias de una reacción que, posiblemente, estuvo relacionada con la transfusión se registraron utilizando los sistemas de preparación de informes existentes. En el análisis final se incluyó un total de 14 005 transfusiones procedentes de 6 instituciones donde se realizó el estudio. Se administraron un total de 4160 transfusiones de plaquetas con InterSol a un total de 1444 pacientes y se administraron 9845 transfusiones de plaquetas con plasma a un total de 2202 pacientes. Se registraron 165 reacciones adversas que fueron adjudicadas por el comité CEC. De ese número, 23 eventos fueron asociados con plaquetas con InterSol y 142 eventos fueron asociados con plaquetas con plasma.
En términos globales, un 1,13% de todas las transfusiones resultó en un EA. El porcentaje de las transfusiones de plaqueta con InterSol que resultaron en EA fue 0,55%, mientras que un 1,37% de las transfusiones de plaquetas con plasma resultaron en EA. El límite de confianza superior de 97,5% correspondiente al riesgo relativo de EA relacionados con la transfusión y asociados con plaquetas con InterSol, en relación con los correspondientes a plaquetas con plasma, fue de 0,66. Esto indica que se cumplió el objetivo del estudio de excluir la duplicación de EA relacionados con la transfusión para plaqueta con InterSol frente a plaquetas con plasma.
La mayoría de las 165 reacciones que fueron adjudicadas por el comité CEC fueron reacciones alérgicas (n=93) o reacciones febriles no hemolíticas a las transfusiones (“Febrile Non-Hemolytic Transfusions Reactions”, FNHTR, n=56), lo que significa, respectivamente, 0,66% y 0,40% del total de transfusiones. Hubo reacciones alérgicas asociadas con 0,29% de las transfusiones de plaquetas con InterSol y con 0,82% de las transfusiones de plaquetas con plasma. Los eventos FNHTR fueron asociados con 0,17% de las transfusiones de plaquetas con InterSol y con 0,50% de las transfusiones de plaquetas con plasma.
Ninguna transfusión de plaquetas con InterSol fue asociada con más de una sola reacción. Siete (7) transfusiones de plaquetas con plasma (0,07% del total de transfusiones) fueron asociadas, cada una, con dos 2) reacciones adversas independientes. Dos (2) reacciones adversas de las plaquetas con InterSol y cinco (5) reacciones adversas de las plaquetas con plasma fueron clasificadas como graves. Todas las demás reacciones fueron clasificadas como no graves. Todas las reacciones adversas fueron registradas con un efecto final “menor o sin secuelas”.
No se identificaron tendencias adversas en cuanto a seguridad después de la revisión de todos los EA por parte del Comité independiente de casos clínicos (Clinical Events Committee).
Tabla 2: Evaluaciones "in vitro"
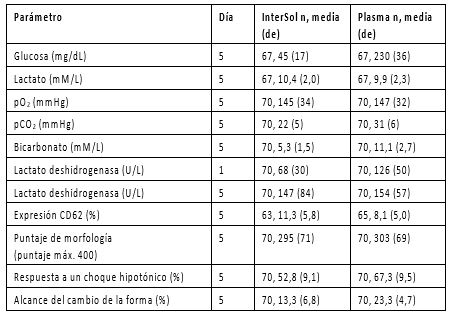
Tabla 3: Evaluaciones "in vitro" — Plaquetas irradiadas y sin irradiar
Cómo se suministra/Manipulación y almacenamiento:
500 mL de solución esterilizada en un contenedor plástico que no es de PVC y que incluye una vía de fluido apirogéna esterilizada. El contenedor de la solución InterSol viene con una cubierta de plástico ventilada que funciona como una cubierta antipolvo para el contenedor. La cubierta antipolvo no es una barrera estéril y no es un elemento que indique la fecha de caducidad del producto InterSol.
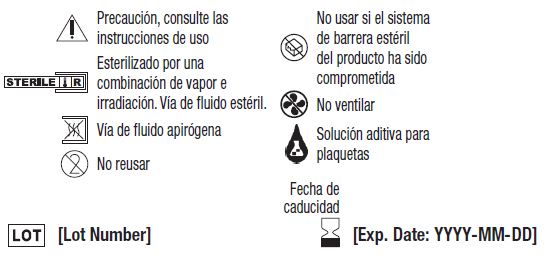
Símbolos con sus definiciones:


Fresenius Kabi AG
61346 Bad Homburg / Germany
Tel.: +49 (0) 61 72 / 686-0
www.fresenius-kabi.com

Fenwal International Inc.
Road 357 K.M. 0.8
Maricao, Puerto Rico 00606
Importado y distribuido por:
Fresenius Kabi México S.A. de C.V.
Av. Paseo del Norte No. 5300-A,
Colonia San Juan de Ocotán,
C.P. 45010, Zapopan, Jalisco, México
Reg. No.: [Pending Number]
© 2019 Fresenius Kabi AG. Todos los derechos reservados
-
INDICATIONS & USAGE
Rx only
For use with the AMICUS Separator only.
Store at Controlled Room Temperature. Protect from freezing. Avoid excessive heat. Definition of "Controlled Room Temperature":
"A temperature maintained thermostatically that encompasses the usual and customary working environment of 20° to 25°C (68° to 77°F); that results in a mean kinetic temperature calculated to be not more than 25°C; and that allows for excursions between 15°C and 30°C (59° and 86°F) that are experienced in pharmacies, hospitals, and warehouses. Provided the mean kinetic temperature remains in the allowed range, transient spikes up to 40°C are permitted as long as they do not exceed 24 hours ... The mean kinetic temperature is a calculated value that may be used as an isothermal storage temperature that simulates the non isothermal effects of storage temperature variations."
Reference: United States Pharmacopeia, General Notices. United States Pharmacopeial Convention, Inc. 12601 Twinbrook Parkway, Rockville, MD.
Dispose of waste in appropriate biohazard container or according to local regulatory requirements.Indications and Usage:
InterSol solution is an isotonic solution designed to replace a proportion of the plasma used in the storage of AMICUS™-derived leukoreduced apheresis platelets under standard blood banking conditions. There is no direct therapeutic effect to be expected from the formulation. The solution should never be infused directly into a patient.InterSol platelets are leukocyte-reduced apheresis platelet concentrates that are stored in a mix of 65% InterSol and 35% plasma, nominal. InterSol platelets prepared within the range of Table 1 may be stored for up to 5 days at 20-24°C, with continuous agitation.

-
DOSAGE & ADMINISTRATION
Dosage and Administration:
InterSol solution may only be used with the AMICUS Separator System using an AMICUS Apheresis Kit with a Platelet Additive Solution (PAS) Connector. For full instructions on the use of InterSol solution with the AMICUS separator, see the AMICUS Operator’s Manual.Prior to use of InterSol solution, check the plastic container for leaks by squeezing the bag firmly. If leaks are found, discard solution.
To connect the InterSol solution to the AMICUS Apheresis Kit:
1) Close the clamp on the PAS Connector line.
2) Remove the protective cap from the membrane port on the InterSol solution container.
3) Insert the spike on the PAS Connector line into the membrane port on the InterSol solution container until firmly seated.
4) For the addition of InterSol solution to the platelet product, follow the instructions provided in the AMICUS Separator Operator’s Manual. -
DOSAGE FORMS & STRENGTHS
Dosage Forms and Strengths:
InterSol solution is provided as a 500 mL sterile and non-pyrogenic solution in a non-PVC plastic container with a sterile and non-pyrogenic fluid path. Each 100 mL contains 305 mg Dibasic Sodium Phosphate, Anhydrous, USP; 93 mg Monobasic Sodium Phosphate, Monohydrate, USP; 318 mg Sodium Citrate, Dihydrate, USP; 442 mg Sodium Acetate, Trihydrate, USP; 452 mg Sodium Chloride, USP; Water for Injection, USP quantity sufficient. -
CONTRAINDICATIONS
Contraindications:
InterSol solution is added to AMICUS-derived leukoreduced platelet concentrates after the apheresis procedure is complete. It is not for direct intravenous infusion. There are no known contraindications associated with the use of InterSol solution for the preparation of InterSol platelets. -
WARNINGS AND PRECAUTIONS
Warnings and Precautions:
- InterSol solution is NOT FOR DIRECT INTRAVENOUS INFUSION.
- Do not use if particulate matter is present or if the solution is cloudy.
- Do not use if the container is damaged, leaking or if there is any visible sign of deterioration.
- Do not vent.
- Do not reuse. Discard unused or partially used InterSol solution.
- Protect from sharp objects.
- Verify that the InterSol solution has been securely attached to the PAS Connector line to avoid disconnection and leaks.
-
ADVERSE REACTIONS
Adverse Reactions:
InterSol solution is added to AMICUS-derived leukoreduced platelet concentrates after the apheresis procedure is complete. It is not for direct intravenous infusion. InterSol solution is not expected to cause adverse events other than those normally associated with platelet transfusion. - DRUG ABUSE AND DEPENDENCE
- OVERDOSAGE
-
DESCRIPTION
Description:
InterSol solution is an isotonic solution designed to replace a proportion of the plasma used in the storage of platelets. The solution contains constituents that are naturally occurring components present in many cellular systems: sodium acetate as a nutrient, sodium citrate to prevent platelet clumping and activation, sodium phosphate for buffering and sodium chloride for osmolarity. InterSol solution does not have a pharmacological effect in vivo, but rather acts to provide the appropriate environment and nutrients in lieu of a portion of the plasma normally used for the storage of platelets. - CLINICAL PHARMACOLOGY
-
CLINICAL STUDIES
Clinical Studies:
In Vitro Biochemical and Functional Evaluations
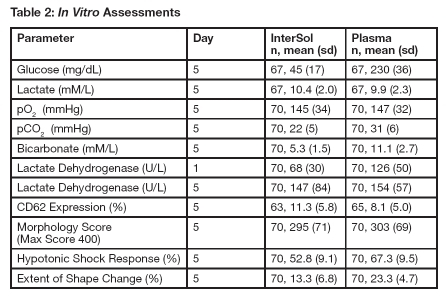
InterSol platelet concentrates (n=70) prepared using the AMICUS separator and stored for 5 days showed a median and mean pH value on Day 5 of 7.2 and 7.2 ± 0.1 (range: 6.9-7.5), respectively, with a lower non-parametric 95%/95% tolerance limit of 6.9.Supplemental in vitro assessments of InterSol platelets and AMICUS-derived leukoreduced platelets stored in 100% plasma are presented in Table 2.
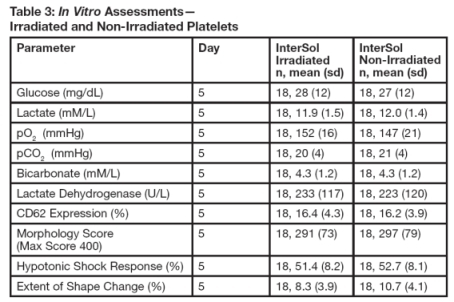
InterSol platelets irradiated (n=18) at either 2500 or 2800 cGray (dependent on site procedures) were compared to non-irradiated InterSol platelets. At the end of storage on Day 5 the platelet yields of the irradiated products were concentrated between 2.5 x 10^11 to 3.5 x 10^11 and included one product with a yield less than2.5 x 10^11 and two products with yields greater than 3.5 x 10^11. In vitro testing is not predictive of in vivo performance, which was not evaluated. Summary statistics are presented in Table 3.
In Vivo Recovery and Survival in Healthy Subjects
In vivo evaluation of InterSol platelets at Day 5 (n=33) compared to a fresh platelet control resulted in a mean percent recovery of 46.4 ± 11.9 percent and 58.0 ± 10.7 percent and mean survival of 5.7 ± 1.4 days and 8.0 ± 1.4 days, respectively. The in vivo data collected were used to calculate the upper limit of a two-sided 95% confidence interval of the mean percent recovery of the algebraic expression (0.66 x Fresh – 5 Day), and of the mean percent survival (days) of the algebraic expression (0.58 x Fresh – 5 Day). The upper bound of the two-sided 95% confidence intervals for recovery and survival on Day 5 was –4.6 and –0.6 respectively and met the requirement of less than 0.
Post-Market Transfusion-Related Adverse Events (AE) Study
An open label, non-randomized, retrospective medical record review study was performed in 6 centers to demonstrate that the overall rate of transfusion-related AEs in patients receiving InterSol Platelet transfusions was not more than double the rate in patients receiving apheresis platelets stored in 100% plasma (Plasma Platelets). The study categorized adverse transfusion reactions according to the definitions outlined by the Biovigilance Component of the National Healthcare Safety Network (NHSN) System. All sites that participated were required to have systems in place to observe transfusion-related AEs and to record when no transfusion-related AE was observed. An independent Clinical Events Committee (CEC) blinded to platelet type and study site adjudicated the reported events.
Patients were prescribed platelet transfusions per each site’s standard practice. The type of platelet unit the patient received was based on the site’s inventory at the time the transfusion was ordered. Site personnel observed the platelet transfusion recipient following their standard procedures. Signs and symptoms of a potential transfusion-related reaction were noted using existing reporting systems. A total of 14,005 transfusions from 6 study sites were included in the final analysis. A total of 4,160 InterSol Platelet transfusions were given to a total of 1,444 patients, and 9,845 Plasma Platelet transfusions were given to 2,202 patients. There were 165 CEC-adjudicated adverse reactions reported. Of those, 23 events were associated with InterSol Platelets and 142 events were associated with Plasma Platelets.
Overall, 1.13% of all transfusions resulted in an AE. The percentage of InterSol Platelet transfusions which led to AEs was 0.55%, while 1.37% of Plasma Platelet transfusions resulted in AEs. The 97.5% upper confidence limit for the relative risk of transfusion-related AE associated with InterSol Platelets relative to Plasma Platelets was 0.66, indicating that the study objective of ruling out a doubling of transfusion-related AEs for InterSol vs. Plasma Platelets was met.
The majority of the 165 CEC-adjudicated reactions were Allergic (n=93) or Febrile Non-Hemolytic Transfusions Reactions (FNHTR, n=56), at 0.66% and 0.40% of total transfusions, respectively. There were allergic reactions associated with 0.29% of InterSol Platelet transfusions and 0.82% of Plasma Platelet transfusions. FNHTR events were associated with 0.17% of InterSol Platelet transfusions and 0.50% of Plasma Platelet transfusions.
No InterSol Platelet transfusion was associated with more than one reaction. Seven (7) Plasma Platelet transfusions (0.07% of total transfusions) were associated with two separate adverse reactions each. Two (2) InterSol Platelet and five (5) Plasma Platelet adverse reactions were classified as severe. All other reactions were classified as non-severe. All adverse reactions were reported with an outcome of “Minor or No Sequelae”.
No adverse safety trends were identified after review of all the AEs by the independent Clinical Events Committee.
-
HOW SUPPLIED
How Supplied/Storage and Handling:
500 mL sterile solution in a non-PVC plastic container with a sterile, non-pyrogenic fluid path. The InterSol solution container is supplied in a vented plastic overwrap covering that serves as a dust cover for the container. The dust cover is not a sterility barrier and is not an element that defines the expiration date of the InterSol product.

© 2019 Fresenius Kabi AG. All rights reserved.
Fresenius Kabi AG
61346 Bad Homburg / Germany
Tel.: +49 (0) 61 72 / 686-0
www.fresenius-kabi.com
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
L6B7850
NDC No. 0942-9603-12
Solamente Rx
500 mL
Solución InterSol Solución aditiva para plaquetas 3
Para ser usada exclusivamente con el separador AMICUS. Almacenar a temperatura ambiente controlada. Proteger de la congelación. Evitar el calor excesivo. Deseche la porción no utilizada. Cada 100 mL contienen 305 mg de fosfato de sodio dibásico, anhidro, USP; 93 mg de fosfato de sodio monobásico, monohidratado, USP; 318 mg de citrato de sodio, dihidratado, USP; 442 mg de acetato de sodio, trihidratado, USP; 452 mg de cloruro de sodio, USP; agua para inyección, USP, en cantidad suficiente.
NO PARA INFUSIÓN INTRAVENOSA DIRECTA.

Fabricado para
Fresenius Kabi AG
61346 Bad Homburg / Germany
Tel.: +49 (0) 61 72 / 686-0

Fabricado por
Fenwal International Inc.
Road 357 K.M. 0.8
Maricao, Puerto Rico 00606
Made in US
Importado y Distribuido por:
Uruguay BIOERIX SA
Simón Bolívar 1472-
Tel/Fax 27081624
Dirección Técnica: Q.F. Mariela Ricca
Importado y distribuido por:
Fresenius Kabi México S.A. de C.V.
Av. Paseo del Norte No. 5300-A,
Colonia San Juan de Ocotán,
C.P. 45010, Zapopan, Jalisco, México
Reg. No.: [Pending Number]
L6B7850
NDC No. 0942-9603-12
Rx Only
500 mL
InterSol Solution Platelet Additive Solution 3
For use with the AMICUS separator only. Store at Controlled Room Temperature. Protect from freezing. Avoid excessive heat.
Discard unused portion. Each 100 mL contains 305 mg Dibasic Sodium Phosphate, Anhydrous, USP; 93 mg Monobasic Sodium Phosphate, Monohydrate, USP; 318 mg Sodium Citrate, Dihydrate, USP; 442 mg Sodium Acetate, Trihydrate, USP; 452 mg Sodium Chloride, USP; Water for Injection, USP quantity sufficient. NOT FOR DIRECT INTRAVENOUS INFUSION.


Manufacturer
Fresenius Kabi AG
61346 Bad Homburg / Germany
Tel.: +49 (0) 61 72 / 686-0

Manufacturing facility/
Manufacture by
Fenwal International Inc.
Road 357 K.M. 0.8
Maricao, Puerto Rico 00606
Made in US
Imported and distributed by:
Uruguay BIOERIX SA
Simón Bolívar 1472-
Tel/Fax 27081624
Dirección Técnica: Q.F. Mariela Ricca
Imported and distributed by:
Fresenius Kabi México S.A. de C.V.
Av. Paseo del Norte No. 5300-A,
Colonia San Juan de Ocotán,
C.P. 45010, Zapopan, Jalisco, México
Reg. No.: [Pending Number]

-
INGREDIENTS AND APPEARANCE
SOLUCION INTERSOL
solucion aditiva para plaquetas 3 solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0942-9603 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Sodium Chloride (UNII: 451W47IQ8X) (CHLORIDE ION - UNII:Q32ZN48698) Sodium Chloride 452 mg in 100 mL SODIUM ACETATE (UNII: 4550K0SC9B) (ACETATE ION - UNII:569DQM74SC) SODIUM ACETATE 442 mg in 100 mL Trisodium Citrate Dihydrate (UNII: B22547B95K) (Anhydrous Citric Acid - UNII:XF417D3PSL) Anhydrous Citric Acid 318 mg in 100 mL SODIUM PHOSPHATE, DIBASIC (UNII: GR686LBA74) (PHOSPHATE ION - UNII:NK08V8K8HR) SODIUM PHOSPHATE, DIBASIC 305 mg in 100 mL Sodium Phosphate, Monobasic, Monohydrate (UNII: 593YOG76RN) (PHOSPHATE ION - UNII:NK08V8K8HR) Sodium Phosphate, Monobasic, Monohydrate 93 mg in 100 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0942-9603-12 500 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA BN080041 10/25/2012 Labeler - Fenwal, Inc. (794519020) Establishment Name Address ID/FEI Business Operations Fenwal International, Inc. 091164590 MANUFACTURE(0942-9603)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.