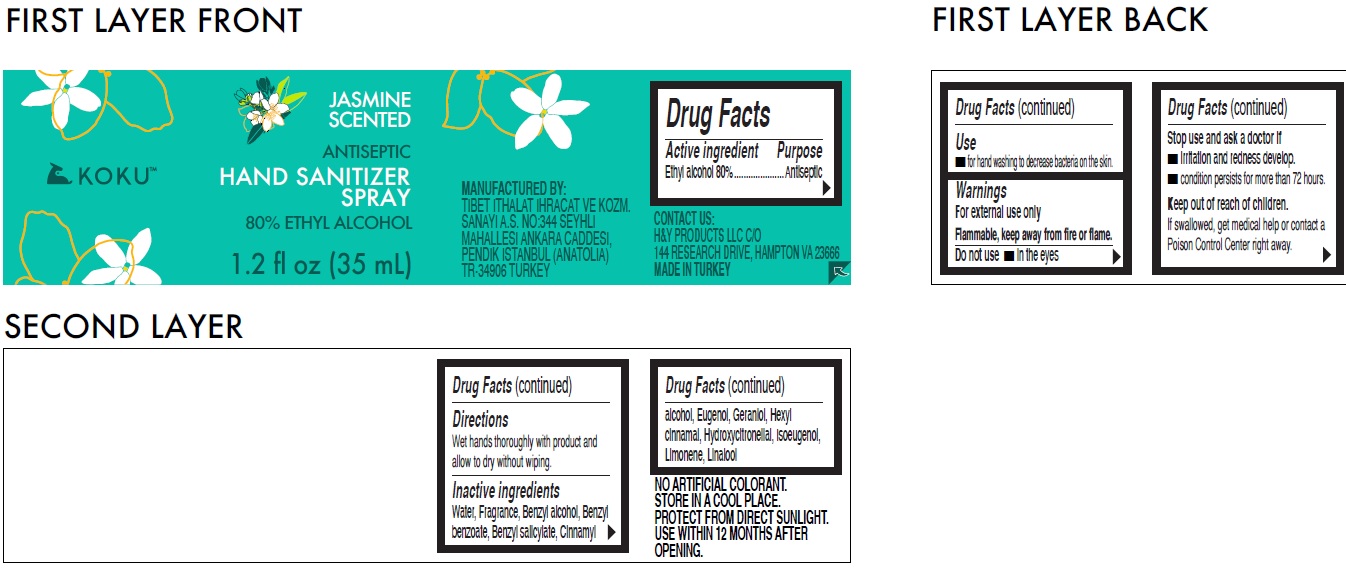
Jasmine Scented Antiseptic Hand Sanitizer Spray 80% Ethyl Alcohol
Jasmine Scented Antiseptic Hand Sanitizer 80 Ethyl Alcohol by
Drug Labeling and Warnings
Jasmine Scented Antiseptic Hand Sanitizer 80 Ethyl Alcohol by is a Otc medication manufactured, distributed, or labeled by H&Y Products LLC dba Kolonya Carre. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
JASMINE SCENTED ANTISEPTIC HAND SANITIZER 80 ETHYL ALCOHOL- alcohol liquid
H&Y Products LLC dba Kolonya Carre
----------
Jasmine Scented Antiseptic Hand Sanitizer Spray 80% Ethyl Alcohol
Warnings
For external use onlyFlammable, keep away from fire or flame.
| JASMINE SCENTED ANTISEPTIC HAND SANITIZER 80 ETHYL ALCOHOL
alcohol liquid |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - H&Y Products LLC dba Kolonya Carre (070644433) |
Revised: 12/2023
Document Id: 0daa3ebd-3919-4d0c-e063-6294a90a706d
Set id: d6105948-9ce9-4a49-b095-52c4a224e496
Version: 3
Effective Time: 20231229