133 Medline Retail Nasal Swabs
Medline by
Drug Labeling and Warnings
Medline by is a Otc medication manufactured, distributed, or labeled by Medline Industries, LP. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
MEDLINE- ethyl alcohol swab
Medline Industries, LP
----------
133 Medline Retail Nasal Swabs
Warnings
For external use only.
Flammable, keep away from fire or flame.
Directions
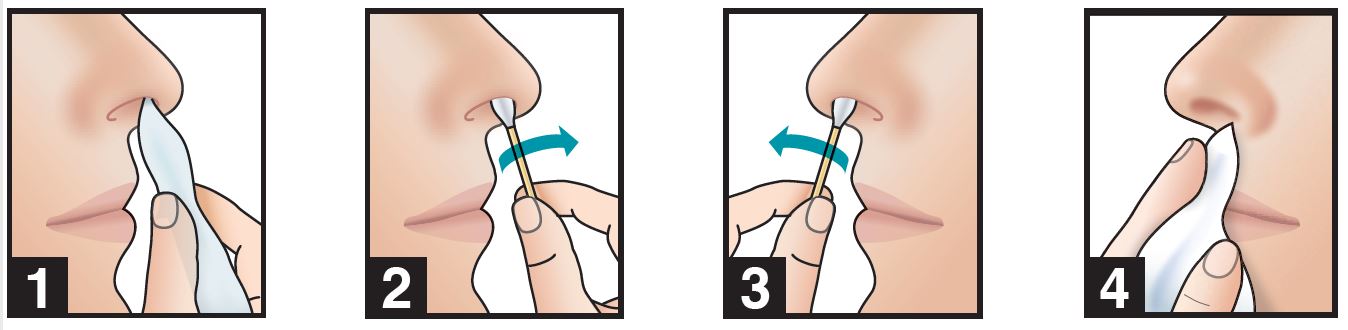
- 1. use a tissue to clean the inside of both nostrils, including the inside tip of nostril. Discard
- 2. insert swab comfortably into tip of right nostril and rotate for 30 seconds, covering all surfaces
- 3. using same swab, repeat step 2 with tip of left nostril
- 4. do not blow nose. If solution drips, gently wipe with a tissue
Inactive ingredients
ascorbyl palmitate, benzyl alcohol, BHT, ricinus communis (castor) seed oil, fragrance, isopropyl alcohol, menthol, polyaminopropyl biguanide, purified water
| MEDLINE
ethyl alcohol swab |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Medline Industries, LP (025460908) |
| Registrant - Medline Industries, LP (025460908) |
Revised: 11/2023
Document Id: 0b653332-6ea1-bb94-e063-6294a90a24c3
Set id: d670cda9-381e-9985-e053-2a95a90a7dc9
Version: 6
Effective Time: 20231130
Trademark Results [Medline]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 MEDLINE 86846058 5021107 Live/Registered |
IPEG, INC. 2015-12-11 |
 MEDLINE 86488830 5166882 Live/Registered |
Medline Industries, Inc. 2014-12-23 |
 MEDLINE 86488829 5171995 Live/Registered |
Medline Industries, Inc. 2014-12-23 |
 MEDLINE 86382367 not registered Dead/Abandoned |
IPEG, INC. 2014-09-02 |
 MEDLINE 79308544 not registered Live/Pending |
Singulus Technologies AG 2020-12-28 |
 MEDLINE 78790084 3301915 Live/Registered |
Medline Industries, Inc. 2006-01-12 |
 MEDLINE 78790077 3365696 Live/Registered |
Medline Industries, Inc. 2006-01-12 |
 MEDLINE 78790069 3343906 Live/Registered |
Medline Industries, Inc. 2006-01-12 |
 MEDLINE 78789639 3311898 Live/Registered |
Medline Industries, Inc. 2006-01-12 |
 MEDLINE 78476716 3311664 Live/Registered |
MEDLINE INDUSTRIES, INC. 2004-08-31 |
 MEDLINE 77978091 3710539 Live/Registered |
Medline Industries, Inc. 2008-09-03 |
 MEDLINE 77561183 3778944 Live/Registered |
Medline Industries, Inc. 2008-09-03 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.

