PULMOZYME- dornase alfa solution
Pulmozyme by
Drug Labeling and Warnings
Pulmozyme by is a Prescription medication manufactured, distributed, or labeled by Genentech, Inc., Roche Singapore Technical Operations Pte. Ltd. (RSTO), Roche Diagnostics GmbH, Genentech, Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use PULMOZYME safely and effectively. See full prescribing information for PULMOZYME.
PULMOZYME® (dornase alfa) inhalation solution, for inhalation use
Initial U.S. Approval: 1993INDICATIONS AND USAGE
PULMOZYME is a recombinant DNase enzyme indicated in conjunction with standard therapies for the management of cystic fibrosis (CF) patients to improve pulmonary function. (1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Inhalation Solution: 2.5 mg/2.5 mL clear, colorless solution in single-use ampules. (3)
CONTRAINDICATIONS
PULMOZYME is contraindicated in patients with known hypersensitivity to dornase alfa, Chinese Hamster Ovary cell products, or any component of the product. (4)
WARNINGS AND PRECAUTIONS
None. (5)
ADVERSE REACTIONS
The most common adverse reactions (occurring in ≥3% of patients treated with PULMOZYME over placebo) seen in clinical trials in CF patients were: voice alteration, pharyngitis, rash, laryngitis, chest pain, conjunctivitis, rhinitis, decrease in FVC of ≥10%, fever, and dyspnea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Genentech at 1-888-835-2555 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 1/2018
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
2.2 Directions for Use
2.3 Storage and Handling
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
PULMOZYME® (dornase alfa) is indicated for daily administration in conjunction with standard therapies for the management of cystic fibrosis (CF) patients to improve pulmonary function.
In CF patients with an FVC ≥ 40% of predicted, daily administration of PULMOZYME has also been shown to reduce the risk of respiratory tract infections requiring parenteral antibiotics.
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
The recommended dosage for use in most cystic fibrosis patients is one 2.5 mg single-use ampule inhaled once daily using a recommended jet nebulizer/compressor system or eRapid™ Nebulizer System.
Some patients may benefit from twice daily administration [see Clinical Studies (14)].
2.2 Directions for Use
Administer PULMOZYME via the eRapid Nebulizer System or via a jet nebulizer connected to an air compressor with an adequate air flow and equipped with a mouthpiece or suitable face mask (see Table 1). No data are currently available to support the administration of PULMOZYME with other nebulizer systems.
Do not dilute or mix PULMOZYME with other drugs in the nebulizer. Mixing of PULMOZYME with other drugs could lead to adverse physicochemical and/or functional changes in PULMOZYME or the admixed compound.
Table 1. Recommended Jet Nebulizers/Compressors and Nebulizer Systems - * Patients who are unable to inhale or exhale orally throughout the entire nebulization period may use the PARI BABY™ nebulizer.
- † Consisting of the eRapid™ Nebulizer Handset with eBase™ Controller.
Jet Nebulizer Compressor Hudson T Up-draft II® with Pulmo-Aide® Marquest Acorn II® with Pulmo-Aide® PARI LC® Plus with PARI PRONEB® *PARI BABY™ with PARI PRONEB® Durable Sidestream® with MOBILAIRE™ Durable Sidestream® with Porta-Neb® Nebulizer System eRapid™ Nebulizer System† The patient should follow the manufacturer's instructions on the use and maintenance of the equipment, including cleaning and disinfection procedures.
When PULMOZYME is administered with the eRapid Nebulizer System, advise patients to replace the handset after 90 uses, regardless of whether the EasyCare cleaning aid is used. Since delivery data are not available for PULMOZYME administered with the eRapid handset beyond 90 administrations, delivery of the appropriate therapeutic dose of PULMOZYME cannot be assured beyond 90 administrations. The eRapid Nebulizer System should only be used by adults and children who can use a mouthpiece, and not by younger children who need a mask to take PULMOZYME.
2.3 Storage and Handling
Store PULMOZYME ampules in their protective foil pouch under refrigeration and protected from light. Refrigerate ampules during transport and do not expose to room temperatures for a total time of 24 hours.
Each PULMOZYME ampule should be squeezed prior to use in order to check for leaks. Discard ampules if the solution is cloudy or discolored. Once opened, the entire contents of the ampule must be used or discarded.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
- 5 WARNINGS AND PRECAUTIONS
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to PULMOZYME in 902 patients, with exposures ranging from 2 weeks of daily administration up to once or twice daily administration for six months. PULMOZYME was studied in both placebo-controlled and uncontrolled trials (n=804 and n=98). The population of patients in placebo-controlled trials was with FVC ≥ 40% of predicted (n=643) or with more advanced pulmonary disease, FVC < 40% of predicted (n=161). The population in the uncontrolled trial included 98 pediatric patients with CF ranging from 3 months to 10 years of age. More than half of the patients received PULMOZYME 2.5 mg by inhalation once a day (n=581), while the rest of patients (n=321) received PULMOZYME 2.5 mg by inhalation twice a day.
Placebo-Controlled Trials
Trial 1: Trial 1 was a randomized, placebo-controlled clinical trial in patients with FVC ≥ 40% of predicted. In this trial, over 600 patients received PULMOZYME once or twice daily for six months. The most common adverse reaction (risk difference ≥5%) was voice alteration. The proportion of most adverse events was similar for patients on PULMOZYME and on placebo, probably reflecting the sequelae of the underlying lung disease. In most cases reactions that were increased were mild, transient in nature, and did not require alterations in dosing. Few patients experienced adverse reactions resulting in permanent discontinuation from PULMOZYME, and the proportion of discontinuations were similar for placebo (2%) and PULMOZYME (3%). Adverse reactions occurring in a higher proportion (greater than 3%) of PULMOZYME treated patients than in placebo-treated patients are listed in Table 2.
Trial 2: Trial 2 was a randomized, placebo-controlled trial in patients with more advanced pulmonary disease (FVC < 40% of predicted) who were treated for 12 weeks. In this trial, the safety profile of PULMOZYME was similar to that reported in patients with less advanced pulmonary disease (FVC ≥ 40% of predicted). Adverse reactions that were reported in this trial with a higher proportion (greater than 3%) in the PULMOZYME treated patients are listed in Table 2.
Table 2. Adverse Reactions Increased 3% or More in PULMOZYME Treated Patients Over Placebo in CF Clinical Trials Adverse Reactions
(of any severity or seriousness)Trial 1
CF Patients with FVC ≥ 40% of predicted treated for 24 weeksTrial 2
CF Patients with FVC <40% of predicted treated for 12 weeksPlacebo
n=325Pulmozyme QD
n=322Pulmozyme BID
n=321Placebo
n=159Pulmozyme QD
n=161- * Single measurement only, does not reflect overall FVC changes.
- † Total reports of dyspnea (regardless of severity or seriousness) had a difference of less than 3% in Trial 2.
Voice alteration 7% 12% 16% 6% 18% Pharyngitis 33% 36% 40% 28% 32% Rash 7% 10% 12% 1% 3% Laryngitis 1% 3% 4% 1% 3% Chest Pain 16% 18% 21% 23% 25% Conjunctivitis 2% 4% 5% 0% 1% Rhinitis Differences were less than 3% 24% 30% FVC decrease of ≥ 10% of predicted* 17% 22% Fever 28% 32% Dyspepsia 0% 3% Dyspnea (when reported as serious) Differences were less than 3% 12%† 17%† Mortality rates observed in controlled trials were similar for the placebo and PULMOZYME treated patients. Causes of death were consistent with progression of cystic fibrosis and included apnea, cardiac arrest, cardiopulmonary arrest, cor pulmonale, heart failure, massive hemoptysis, pneumonia, pneumothorax, and respiratory failure.
Uncontrolled Trial
Trial 3: The safety of PULMOZYME, 2.5 mg by inhalation, was studied with 2 weeks of daily administration in 98 pediatric patients with cystic fibrosis 3 months to 10 years of age (65 aged 3 months to < 5 years, 33 aged 5 to ≤ 10 years). The PARI BABY™ reusable nebulizer (which uses a facemask instead of a mouthpiece) was utilized in patients unable to demonstrate the ability to inhale or exhale orally throughout the entire treatment period (54/65, 83% of the younger and 2/33, 6% of the older patients). Overall, the nature of adverse reactions was similar to that seen in the placebo-controlled trials. The number of patients reporting cough was higher in the younger age group as compared to the older age group (29/65, 45% compared to 10/33, 30%) as was the number reporting moderate to severe cough (24/65, 37% as compared to 6/33, 18%). The number of patients reporting rhinitis was higher in the younger age group as compared to the older age group (23/65, 35% compared to 9/33, 27%) as was the number reporting rash (4/65, 6% as compared to 0/33).
Allergic Reactions
There have been no reports of anaphylaxis attributed to the administration of PULMOZYME. Urticaria, mild to moderate, and mild skin rash have been observed and have been transient. Within all of the studies, a small percentage (average of 2-4%) of patients treated with PULMOZYME developed serum antibodies to PULMOZYME. None of these patients developed anaphylaxis, and the clinical significance of serum antibodies to PULMOZYME is unknown.
6.2 Postmarketing Experience
Postmarketing spontaneous reports and prospectively collected safety data from observational studies confirm the safety profile to be as described in clinical trials [see Adverse Reactions (6.1)].
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk summary
There are no adequate and well-controlled studies with PULMOZYME in pregnant women. However, animal reproduction studies have been conducted with dornase alfa. In these studies, no evidence of fetal harm was observed in rats and rabbits at doses of dornase alfa up to approximately 600 times the maximum recommended human dose (MRHD).
The background risk of major birth defects and miscarriage for the cystic fibrosis population is unknown. However, the background risk in the U.S. general population of major birth defects is 2-4% and of miscarriage is 15-20% of clinically recognized pregnancies.
Data
Animal Data
Reproductive studies have been performed in rats and rabbits at intravenous doses of dornase alfa up to 10 mg/kg/day (approximately 600 times the MRHD in adults). In a combined embryo-fetal development and pre- and post-natal development study, no evidence of maternal toxicity, embryotoxicity, or teratogenicity was observed when dornase alfa was administered to dams throughout organogenesis (Gestation days 6 to 17). Dornase alfa did not elicit adverse effects on fetal or neonatal growth when administered to dams throughout most of gestation and delivery (Gestation days 6 to 25) and nursing (Post-partum days 6 to 21).
A pharmacokinetic study in Cynomolgus monkeys found no detectable levels of dornase alfa in fetal blood or amniotic fluid on gestation day 150 (end of gestation) from mothers that were administered an intravenous bolus dose (0.1 mg/kg) followed by an intravenous infusion dose (0.080 mg/kg) over a 6-hour period during pregnancy.
8.2 Lactation
Risk Summary
It is not known whether PULMOZYME is present in human milk. In a pharmacokinetic study in Cynomolgus monkeys, levels of dornase alfa detected in milk were less than 0.1% of the maternal serum concentration at 24 hours after dosing [intravenous bolus dose (0.1 mg/kg) of dornase alfa followed by an intravenous infusion (0.080 mg/kg/hr) over a 6-hour period] on post-partum day 14. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for PULMOZYME and any potential adverse effects on the breastfed child from PULMOZYME or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of PULMOZYME have been established in pediatric patients 5 years of age and older [see Adverse Reactions (6) and Clinical Studies (14)]. The safety of PULMOZYME, 2.5 mg by inhalation, was studied with 2 weeks of daily administration in 65 patients with cystic fibrosis aged 3 months to < 5 years [see Adverse Reactions (6.1)]. While clinical trial data are limited in pediatric patients younger than 5 years of age, the use of PULMOZYME should be considered for pediatric CF patients who may experience potential benefit in pulmonary function or who may be at risk of respiratory tract infection.
-
10 OVERDOSAGE
Single-dose inhalation studies in rats and monkeys at doses up to 180-times higher than doses routinely used in clinical studies are well tolerated. Single dose oral administration of PULMOZYME in doses up to 200 mg/kg are also well tolerated by rats.
Cystic fibrosis patients have received up to 20 mg BID for up to 6 days and 10 mg BID intermittently (2 weeks on/2 weeks off drug) for 168 days. These doses were well tolerated.
-
11 DESCRIPTION
Dornase alfa is a recombinant human deoxyribonuclease I (rhDNase), an enzyme which selectively cleaves DNA. The protein is produced by genetically engineered Chinese Hamster Ovary (CHO) cells containing DNA encoding for the native human protein, deoxyribonuclease I (DNase). The product is purified by column chromatography and tangential flow filtration. The purified glycoprotein contains 260 amino acids with an approximate molecular weight of 37,000 daltons. The primary amino acid sequence is identical to that of the native human enzyme.
PULMOZYME (dornase alfa) inhalation solution is administered by inhalation of an aerosol mist produced by a compressed air driven nebulizer or an approved nebulizer system [see Clinical Studies (14) and Dosage and Administration (2)]. PULMOZYME is a sterile, clear, colorless, highly purified solution in single-use ampules. Each ampule delivers 2.5 mL of the solution to the nebulizer bowl. Each mL of aqueous solution contains 1 mg dornase alfa, calcium chloride dihydrate (0.15 mg) and sodium chloride (8.77 mg). The solution contains no preservative. The nominal pH of the solution is 6.3.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
PULMOZYME is recombinant human deoxyribonuclease I (rhDNase), an enzyme which selectively cleaves DNA. In preclinical in vitro studies, PULMOZYME hydrolyzes the DNA in sputum of CF patients and reduces sputum viscoelasticity. In CF patients, retention of viscous purulent secretions in the airways contributes both to reduced pulmonary function and to exacerbations of infection. Purulent pulmonary secretions contain very high concentrations of extracellular DNA released by degenerating leukocytes that accumulate in response to infection.
12.3 Pharmacokinetics
When 2.5 mg PULMOZYME was administered by inhalation to eighteen CF patients, mean sputum concentrations of 3 µg/mL DNase were measurable within 15 minutes. Mean sputum concentrations declined to an average of 0.6 µg/mL two hours following inhalation. Inhalation of up to 10 mg TID of PULMOZYME by 4 CF patients for six consecutive days did not result in a significant elevation of serum concentrations of DNase above normal endogenous levels. After administration of up to 2.5 mg of PULMOZYME twice daily for six months to 321 CF patients, no accumulation of serum DNase was noted. Dornase alfa is expected to be metabolized by proteases present in biological fluids. A human intravenous dose study suggested an elimination half-life of 3-4 hours for dornase alfa.
PULMOZYME, 2.5 mg by inhalation, was administered daily to 98 patients aged 3 months to ≤ 10 years, and bronchoalveolar lavage (BAL) fluid was obtained within 90 minutes of the first dose. BAL DNase concentrations were detectable in all patients but showed a broad range, from 0.007 to 1.8 µg/mL. Over an average of 14 days of exposure, serum DNase concentrations (mean ± s.d.) increased by 1.1 ± 1.6 ng/mL for the 3 months to < 5 year age group and by 0.8 ± 1.2 ng/mL for the 5 to ≤ 10 year age group. The relationship between BAL or serum DNase concentration and adverse experiences and clinical outcomes is unknown.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
PULMOZYME produced no treatment-related increases in the incidence of tumors in a lifetime study in Sprague Dawley rats that were administered inhaled doses up to 0.246 mg/kg/day (approximately 30 times the MRHD in adults). There was no increase in the development of benign or malignant neoplasms and no occurrence of unusual tumor types in rats after lifetime exposure.
PULMOZYME tested negative in the following genotoxicity assays: the in vitro Ames assay, in vitro mouse lymphoma assay, and in vivo mouse bone marrow micronucleus assay. No evidence of impairment of fertility was observed in male and female rats that received intravenous doses up to 10 mg/kg/day (approximately 600 times the MRHD in adults).
-
14 CLINICAL STUDIES
Trial in CF Patients with FVC ≥40% of Predicted
PULMOZYME has been evaluated in a randomized, placebo-controlled trial of clinically stable cystic fibrosis patients, 5 years of age and older, with baseline forced vital capacity (FVC) greater than or equal to 40% of predicted and receiving standard therapies for cystic fibrosis. Patients were treated with placebo (325 patients), 2.5 mg of PULMOZYME once a day (322 patients), or 2.5 mg of PULMOZYME twice a day (321 patients) for six months administered via a Hudson T Up-draft II® nebulizer with a Pulmo-Aide® compressor.
Both doses of PULMOZYME resulted in significant reductions in the number of patients experiencing respiratory tract infections requiring use of parenteral antibiotics compared with the placebo group. Administration of PULMOZYME reduced the relative risk of developing a respiratory tract infection by 27% and 29% for the 2.5 mg daily dose and the 2.5 mg twice daily dose, respectively (see Table 3). The data suggest that the effects of PULMOZYME on respiratory tract infections in older patients ( > 21 years) may be smaller than in younger patients, and that twice daily dosing may be required in the older patients. Patients with baseline FVC > 85% may also benefit from twice a day dosing (see Table 3). The reduced risk of respiratory infection observed in PULMOZYME treated patients did not directly correlate with improvement in FEV1 during the initial two weeks of therapy.
Within 8 days of the start of treatment with PULMOZYME, mean FEV1 increased 7.9% in those treated once a day and 9.0% in those treated twice a day compared to the baseline values. The overall mean FEV1 during long-term therapy increased 5.8% from baseline at the 2.5 mg daily dose level and 5.6% from baseline at the 2.5 mg twice daily dose level. Placebo recipients did not show significant mean changes in pulmonary function testing (see Figure 1).
For patients 5 years of age or older with baseline FVC greater than or equal to 40%, administration of PULMOZYME decreased the incidence of occurrence of first respiratory tract infection requiring parenteral antibiotics, and improved mean FEV1, regardless of age or baseline FVC.
Table 3. Incidence of First Respiratory Tract Infection Requiring Parenteral Antibiotics in Patients with FVC ≥40% of Predicted Placebo
N=3252.5 mg QD
N=3222.5 mg BID
N=321Percent of Patients Infected 43% 34% 33% Relative Risk (vs placebo) 0.73 0.71 p-value (vs placebo) 0.015 0.007 Subgroup by Age and Baseline FVC Placebo
% (N)2.5 mg QD
% (N)2.5 mg BID
% (N)Age 5-20 years 42% (201) 25% (199) 28% (184) 21 years and older 44% (124) 48% (123) 39% (137) Baseline FVC 40-85% Predicted 54% (194) 41% (201) 44% (203) > 85% Predicted 27% (131) 21% (121) 14% (118) Figure 1. Mean Percent Change from Baseline FEV1 in Patients with FVC ≥40% of Predicted 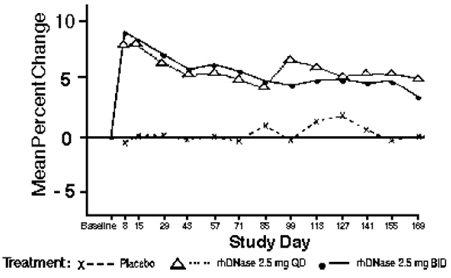
Trial in CF Patients with FVC <40% of Predicted
PULMOZYME has also been evaluated in a second randomized, placebo-controlled trial in clinically stable patients with baseline FVC < 40% of predicted. Patients were enrolled and treated with placebo (162 patients) or PULMOZYME 2.5 mg QD (158 patients) for twelve weeks. In patients who received PULMOZYME, there was an increase in mean change (as percent of baseline) compared to placebo in FEV1 (9.4% vs. 2.1%, p < 0.001) and in FVC (12.4% vs. 7.3%, p < 0.01). PULMOZYME did not significantly reduce the risk of developing a respiratory tract infection requiring parenteral antibiotics (54% of PULMOZYME patients vs. 55% of placebo patients had experienced a respiratory tract infection by 12 weeks, relative risk = .93, p = 0.62).
The effect of PULMOZYME on exercise tolerance has not been established in adult and pediatric patients.
Other Studies
Clinical trials have indicated that PULMOZYME therapy can be continued or initiated during an acute respiratory exacerbation.
Short-term dose ranging studies demonstrated that doses in excess of 2.5 mg BID did not provide further improvement in FEV1. Patients who have received drug on a cyclical regimen (i.e., administration of PULMOZYME 10 mg BID for 14 days, followed by a 14 day wash out period) showed rapid improvement in FEV1 with the initiation of each cycle and a return to baseline with each PULMOZYME withdrawal.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
PULMOZYME (dornase alfa) inhalation solution is a sterile, clear, colorless solution supplied in:
- 30 unit cartons containing 5 foil pouches of 6 single-use ampules. Each 2.5 mL ampule contains 2.5 mg of dornase alfa (1 mg/mL): NDC: 50242-100-40.
Storage and Handling
Store PULMOZYME under refrigeration (2°C to 8°C/36°F to 46°F) in their protective foil to protect from light. Do not use beyond the expiration date stamped on the ampule. Store unused ampules in their protective foil pouch under refrigeration. Refrigerate PULMOZYME during transport and do not expose to room temperatures for a total time of 24 hours.
-
17 PATIENT COUNSELING INFORMATION
Advise patients to read the FDA-approved Patient Labeling (Instructions for Use).
Storage and Handling Information
Instruct patients on the proper techniques to store and handle PULMOZYME. PULMOZYME must be stored in the refrigerator at 2 to 8°C (36 to 46°F) and protected from light. It should be kept refrigerated during transport and should not be exposed to room temperatures for a total time of 24 hours.
Advise patients to squeeze each ampule prior to use in order to check for leaks. The solution should be discarded if it is cloudy or discolored. Once opened, the entire contents of the ampule must be used or discarded.
Instruct patients in the proper use and maintenance of the jet nebulizer/compressor system or eRapid Nebulizer System used in PULMOZYME delivery.
Instruct patients not to dilute or mix PULMOZYME with other drugs in the nebulizer. Mixing of PULMOZYME with other drugs could lead to adverse physicochemical and/or functional changes in PULMOZYME or the admixed compound.
Use with the eRapid Nebulizer System
Instruct patients and caregivers to read and follow the directions in both the PULMOZYME Instructions for Use and in the Manufacturer's eRapid Nebulizer System Instruction Booklet.
Instruct patients and caregivers to clean the handset, including the medication reservoir, medicine cap, aerosol head, and mouthpiece, after each use. Instruct patients and caregivers to disinfect the handset, including the medication reservoir, medicine cap, aerosol head, and mouthpiece, after each day of use.
Instruct patients to replace the handset after 90 uses, regardless of whether the EasyCare cleaning aid is used. Since delivery data are not available for PULMOZYME administered with the eRapid handset beyond 90 administrations, delivery of the appropriate therapeutic dose of PULMOZYME cannot be assured beyond 90 administrations.
- SPL UNCLASSIFIED SECTION
-
Instructions for Use
PULMOZYME® (PULL-muh-zyme)
(dornase alfa)
Inhalation Solution
Instructions for Use with Jet Nebulizers and Compressors
See the other side of this Instructions for Use for information on use of Pulmozyme with the ultrasonic eRapid™ Nebulizer System
Read this Instructions for Use before you start taking Pulmozyme and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or your treatment.
A nebulizer and a compressor are used together to give a dose of Pulmozyme. A nebulizer changes the Pulmozyme liquid medicine into a fine mist you inhale by breathing through a mouthpiece. A compressor gives the nebulizer power and makes the nebulizer work.
Pulmozyme should only be used with the approved nebulizers and compressors listed below, or with the eRapid Nebulizer System (see other side).
Do not use any other inhaled medicines in the nebulizer at the same time. Keep all other inhaled medication systems completely separate from Pulmozyme.
Do not use a mask. Use the mouthpiece provided with each nebulizer kit.
If your child cannot breathe in or breathe out by mouth, you may use the PARI BABY™ reusable nebulizer, but you should discuss it with your doctor first. The PARI BABY™ nebulizer is the same as the PARI LC Plus Jet system, except the mouthpiece is replaced by a tight fitting face mask connected to an elbow piece.
Follow the steps on this side of the sheet to administer Pulmozyme using the following jet nebulizer systems.
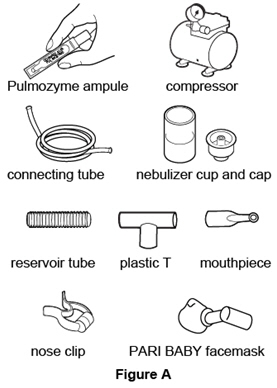
Nebulizer Compressor Hudson T Up-draft II® with Pulmo-Aide® Marquest Acorn II® with Pulmo-Aide® PARI LC® Plus with PARI PRONEB® PARI BABY™ with PARI PRONEB® Durable Sidestream® with MOBILAIRE™ Durable Sidestream® with Porta-Neb® Supplies you will need to give a dose of Pulmozyme (See Figure A):
- 1 ampule of Pulmozyme
- Compressor
- Nebulizer cup and screw-on or snap-on cap
- Plastic T connector (not needed for Sidestream nebulizer or PARI BABY)
- Flexible aerosol tube (not needed for Sidestream nebulizer or PARI BABY)
- Clean mouthpiece or PARI BABY facemask
- Long connecting tube
- Nose clip (optional, not needed for PARI BABY)
Preparing the jet nebulizer and compressor:
Step 1. Clean a flat table surface and wash your hands.- Clean a flat table surface.
- Wash your hands well with soap and water before using the Pulmozyme ampule and nebulizer. This helps prevent infection (See Figure B).
Step 2. Gather the nebulizer and test the compressor. - Place the nebulizer parts on a clean, flat table surface within reach.
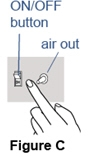
- Test the compressor by turning it on and putting your finger in front of the "air out" or "air" port to feel air flowing. Turn off the compressor (See Figure C).
- For the MOBILAIRE™ compressor only, turn the pressure control knob fully to the right for the highest pressure output.
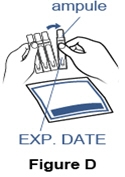
Step 3. Gather the Pulmozyme ampule and check the expiration date. - Remove 1 foil pouch of Pulmozyme from the refrigerator. Open the foil pouch and remove 1 ampule of Pulmozyme. Put the remaining ampules back in the foil pouch and return them to the refrigerator.
- Check the expiration date printed on the ampule (See Figure D).
Do not use the Pulmozyme ampule if the expiration date has passed.
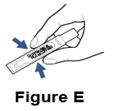
Step 4. Check the Pulmozyme ampule. - Check the ampule for leaks by turning it upside down and gently squeezing (See Figure E).
Do not use the ampule if it is leaking. Throw it away and get a new one. - Check the Pulmozyme liquid in the ampule and make sure it is clear and free of particles.
Do not use Pulmozyme if the liquid is cloudy or discolored. Take the Pulmozyme back to the pharmacy, hospital, or clinic that gave you the medicine.
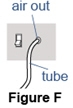
Step 5. Attach the tube to the compressor. - Attach the long connecting tube to the "air out" or "air" port on the compressor (See Figure F).
Step 6. Attach the mouthpiece. - Skip to Step 7 if you use the Sidestream nebulizer or the PARI BABY nebulizer.
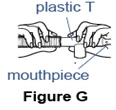
- Push the mouthpiece into the wider end of the plastic T. Attach the flex tube to the other end of the T (See Figure G).
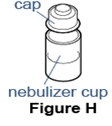
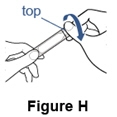
Step 7. Remove the cap from the cup. - Unscrew or unsnap the cap from the nebulizer cup (See Figure H).
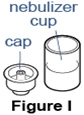
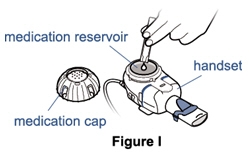
- Put the nebulizer cup on the table face up and place the cap upside down on a clean surface (See Figure I).
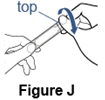
Step 8. Open the Pulmozyme ampule. - Hold the tab at the bottom of the Pulmozyme ampule firmly. Twist off the top. Do not squeeze the body of the ampule (See Figure J).
Step 9. Pour the full Pulmozyme dose into the nebulizer. 
Step 10. Connect the plastic T. - Connect the plastic T to the nebulizer cap (See Figure M).
- If you are using the Sidestream nebulizer, attach the mouthpiece to the top of the nebulizer (See Figure N).
- If you are using the PARI BABY nebulizer, connect the elbow piece and mask to the nebulizer outlet (See Figure O).
Step 11. Attach the tube to the cup. - Connect the open end of the long tube to the port on the bottom of the nebulizer cup by pushing up firmly (See Figure P).
Step 12. Turn on the compressor. - Turn on the compressor and check to see that mist is coming out of the nebulizer (See Figure Q).
- If you are using the MOBILAIRE™ compressor, turn the compressor control knob fully to the right, then turn on the compressor. The pressure gauge should move between 35 psi and 45 psi, the highest pressure output (See Figure R).
Taking your dose of Pulmozyme with a nebulizer: Step 13. Breathe through the mouthpiece. - Skip to Step 14 if you are using the PARI BABY to give Pulmozyme to your child.
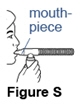
- Place the mouthpiece between your teeth and on top of your tongue (See Figure S).
- Breathe slowly in and out through your mouth. Do not block the airflow with your tongue.
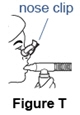
- Do not breathe through your nose. If you have problems breathing only through your mouth, use a nose clip (See Figure T).
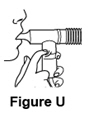
- Do not be concerned if liquid droplets form in the long connecting tube during treatment. When the nebulizer begins spitting, gently tap the nebulizer cup and continue breathing until the nebulizer cup is empty or stops making mist (See Figure U).
- If you need to stop treatment before you are finished, or you begin coughing, turn off the compressor and do not spill any of the medicine.
- To start treatment again, turn on the compressor and continue breathing slowly in and out through your mouth.
- The complete treatment usually takes from 10 to 15 minutes for most nebulizers and compressors.
- If you are using the Sidestream nebulizer with the MOBILAIRE™ or Porta-Neb compressors, treatment usually takes from 2 to 6 minutes.
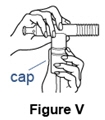
- It is important to inhale the full dose of Pulmozyme. If you find a leak or feel any moisture coming from the nebulizer during treatment, turn off the compressor and check to make sure the nebulizer cap is sealed correctly before continuing (See Figure V).
If you are using the PARI BABY nebulizer to give Pulmozyme to your child, follow the instructions below in Step 14. If not, go to Step 15
Step 14. Breathing through the facemask- During the treatment your child may sit, lie down or stand.
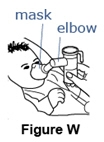
- Place the facemask gently but firmly over your child's nose and mouth (See Figure W).
- Make sure there are no air gaps between the mask and your child's face. This will help make sure that your child will get the full dose of Pulmozyme.
- It is important that you try to keep the body of the nebulizer upright during the entire treatment (See Figure W). The elbow piece will allow you to move the mask for a good fit while keeping the nebulizer body upright.
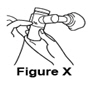
- When the nebulizer begins "spitting," gently tap the nebulizer cup and continue treatment until the nebulizer is empty or stops making a mist (See Figure X).
- If you need to stop the treatment or your child begins to cough during treatment, turn the compressor off. Do not spill any Pulmozyme.
- If you have not removed the mask and you want to begin the treatment again, turn on the compressor.
- If you have removed the mask, repeat the steps above to replace the mask on your child's face and restart the compressor.
- The complete treatment usually takes from 10 to 15 minutes.
It is important that your child inhale the full dose of Pulmozyme. If you find a leak or feel moisture coming from the nebulizer during the treatment, turn off the compressor and make sure the nebulizer cap is sealed correctly before starting the compressor again (See Figure Y). 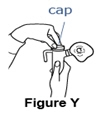
After your treatment with Pulmozyme:
Step 15. Prepare the nebulizer for cleaning and storage.- Turn off the compressor and take apart the nebulizer system. Set aside the flex tube and the long connecting tube.
Note: The Sidestream nebulizer does not use a flex tube. - Throw away the Pulmozyme ampule in your household trash.
- Follow the manufacturer's recommendations for care of your nebulizer and compressor.
How should I store Pulmozyme? - Store Pulmozyme in its foil pouch in the refrigerator between 36°F to 46°F (2°C to 8°C) until you are ready to use it.
- When traveling, Pulmozyme should be kept cold in its foil pouch.
- Do not freeze Pulmozyme.
- Protect Pulmozyme from excessive heat and strong light.
- Do not use Pulmozyme if it has been left at room temperature for a total time of 24 hours or if it becomes cloudy or discolored.
- Do not use Pulmozyme past the expiration date printed on the ampule.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Genentech, Inc.
A Member of the Roche Group
1 DNA Way
South San Francisco, CA 94080-4990
US License No. 1048Approved: December 2014
-
Instructions for Use
PULMOZYME® (PULL-muh-zyme)
(dornase alfa)
Inhalation Solution
Instructions for Use with the eRapid™ Nebulizer System
See the other side of this Instructions for Use for information on use with Jet Nebulizers and Compressors
Read this Instructions for Use before you start taking Pulmozyme and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
This information does not take the place of the Manufacturer's eRapid Nebulizer System Instruction Booklet. This information is needed to show you the right way to use the eRapid Nebulizer System.
The eRapid Nebulizer System changes the Pulmozyme liquid medicine into a fine mist you inhale by breathing through a mouthpiece.
Do not use any other inhaled medicines in the nebulizer at the same time. Keep all other inhaled medication systems completely separate from Pulmozyme.
The eRapid Nebulizer System should only be used by adults and children who can use a mouthpiece, and not by younger children who need a mask to take Pulmozyme.
Follow the instructions on this side of the sheet to give Pulmozyme using the eRapid Nebulizer System.
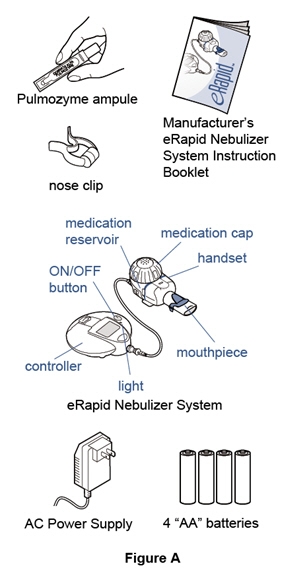
Supplies you will need to give a dose of Pulmozyme (See Figure A):
- 1 ampule of Pulmozyme
- eRapid Nebulizer System, including the:
- eRapid Nebulizer Handset (handset)
- eBase Controller (controller)
- Power source for the controller, using either:
- 4 "AA" batteries (disposable or rechargeable)
- or an AC Power Supply plugged into a typical wall outlet (110 volt power outlet)
- Nose clip (optional)
- Manufacturer's eRapid Nebulizer System Instruction Booklet
Preparing the eRapid nebulizer system: Step 1. Clean a flat table surface and wash your hands. - Clean a flat table surface.
- Wash your hands well with soap and water before using the Pulmozyme ampule and nebulizer. This helps prevent infection (See Figure B).
Step 2. Gather the nebulizer and test it. - Place the eRapid system parts on a clean, flat table surface within reach.
- Make sure the controller batteries are charged or that the unit is plugged into a power outlet (See Figure C).
- Press and hold the ON/OFF button on the controller for a few seconds to test if the controller will turn on (See Figure D).
- When controller is turned on, press and hold the ON/OFF button to turn the controller off.
Step 3. Gather the Pulmozyme ampule and check the expiration date. - Remove 1 foil pouch of Pulmozyme from the refrigerator. Open the foil pouch and remove 1 ampule of Pulmozyme. Put the remaining ampules back in the foil pouch and return them to the refrigerator.
- Check the expiration date printed on the ampule (See Figure E).
Do not use Pulmozyme ampule if expiration date has passed.
Step 4. Check the Pulmozyme ampule. - Check the ampule for leaks by turning it upside down and gently squeezing (See Figure F).
Do not use the ampule if it is leaking. Throw it away and get a new one. - Check the Pulmozyme liquid in the ampule and make sure it is clear and free of particles.
Do not use Pulmozyme if the liquid is cloudy or discolored. Take the Pulmozyme back to the pharmacy, hospital, or clinic that gave you the medicine.
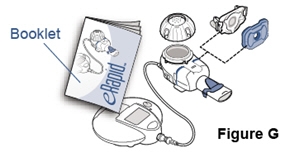
Step 5. Put together the eRapid nebulizer system. - The eRapid Nebulizer System has several small parts that must be put together the right way to give your dose of Pulmozyme (Figure G).
- The parts must be cleaned and disinfected at least 1 time before first use.
- See the Manufacturer's eRapid Nebulizer System Instruction Booklet for cleaning instructions and step-by-step instructions for how to put together your eRapid Nebulizer System (See Figure G).
Step 6. Open the Pulmozyme ampule. - Hold the tab at the bottom of the Pulmozyme ampule firmly. Twist off the top. Do not squeeze the body of the ampule (See Figure H).
Step 7. Pour the full Pulmozyme dose into the nebulizer. - Turn the ampule upside-down and squeeze gently to empty the medicine into the medication reservoir. Keep squeezing until the ampule is empty. It is very important that you squeeze out all the medicine in the ampule (See Figure I).
Step 8. Place the cap on the Medication Reservoir. 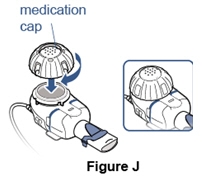
Step 9. Turn on the nebulizer. - Press and hold ON/OFF button on the controller for a few seconds (See Figure K).
- The controller will beep and the light will turn green. The nebulizer will start making mist (See Figure L).
Taking your dose of Pulmozyme:
Step 10. Breathe through the mouthpiece.
- If you need to stop treatment before you are finished, or you begin coughing, press and hold the ON/OFF button on the controller for 1 second (See Figure O).
- To restart your treatment, press and hold the ON/OFF button for 1 second.
- Continue your treatment until the controller beeps twice.
- The nebulizer will shut off by itself when your dose is complete. The complete treatment usually takes from 1 minute to 5 minutes.
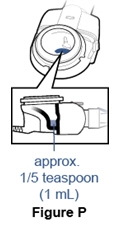
Step 11. Check that you received your full dose. - After your treatment, about 1/5 teaspoon (1mL) of medicine should be left in the medication reservoir (See Figure P).
- Open the medication cap and check the medication reservoir. If more than 1/5 teaspoon (1 mL) is left in the medication reservoir, put the medication cap back on and continue treatment.
- When treatment is complete, throw away the 1/5 teaspoon (1 mL) of medicine that is left in the medication reservoir.
After Your Treatment with Pulmozyme: Step 12. Cleaning the nebulizer - If the controller is on, turn off the controller by pressing and holding the ON/OFF button.
- Take apart the nebulizer system.
- Throw away the empty Pulmozyme ampule in your household trash.
- See the Manufacturer's eRapid Nebulizer System Instruction Booklet for cleaning instructions (See Figure Q).
- The handset, including the medication reservoir, medication cap, aerosol head, and mouthpiece, must be cleaned after each use and disinfected after each day of use.
- Use of the EasyCare cleaning aid is recommended for cleaning the aerosol head located inside the handset 1 time each week.
- Replace the handset after you use your eRapid Nebulizer System 90 times. Replace it even if you use the EasyCare cleaning aid.
- Your handset has been tested for only 90 doses of Pulmozyme. Delivery of the right dose of PULMOZYME cannot be certain after 90 doses.

How should I store Pulmozyme? - Store Pulmozyme in its foil pouch in the refrigerator between 36°F to 46°F (2°C to 8°C) until you are ready to use it.
- When traveling, Pulmozyme should be kept cold.
- Do not freeze Pulmozyme.
- Protect Pulmozyme from excessive heat and strong light.
- Do not use Pulmozyme if it has been left at room temperature for a total time of 24 hours or if it becomes cloudy or discolored.
- Do not use Pulmozyme past the expiration date printed on the ampule.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Genentech, Inc.
A Member of the Roche Group
1 DNA Way
South San Francisco, CA 94080-4990
US License No. 1048Approved: December 2014
Representative sample of labeling (see the HOW SUPPLIED section for complete listing):
-
PRINCIPAL DISPLAY PANEL - 2.5 mL Ampule Pouch Carton
NDC: 50242-100-40
DORNASE ALFA
PULMOZYME®
INHALATION SOLUTION
KEEP REFRIGERATED
4824504
Genentech, Inc.
A Member of the Roche Group
1 DNA Way, South San Francisco, CA 94080-4990
US License No.: 1048
-
INGREDIENTS AND APPEARANCE
PULMOZYME
dornase alfa solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50242-100 Route of Administration RESPIRATORY (INHALATION) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DORNASE ALFA (UNII: 953A26OA1Y) (DORNASE ALFA - UNII:953A26OA1Y) DORNASE ALFA 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength CALCIUM CHLORIDE (UNII: M4I0D6VV5M) 0.15 mg in 1 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 8.77 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50242-100-40 5 in 1 CARTON 12/30/1993 1 6 in 1 POUCH 1 2.5 mL in 1 AMPULE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA103532 12/30/1993 Labeler - Genentech, Inc. (080129000) Registrant - Genentech, Inc. (080129000) Establishment Name Address ID/FEI Business Operations Genentech, Inc. (SSF) 080129000 API MANUFACTURE(50242-100) , ANALYSIS(50242-100) , MANUFACTURE(50242-100) Establishment Name Address ID/FEI Business Operations Roche Singapore Technical Operations Pte. Ltd. (RSTO) 937189173 ANALYSIS(50242-100) Establishment Name Address ID/FEI Business Operations Genentech, Inc. (OCN) 146373191 ANALYSIS(50242-100) Establishment Name Address ID/FEI Business Operations Roche Diagnostics GmbH 323105205 ANALYSIS(50242-100)
Trademark Results [Pulmozyme]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 PULMOZYME 74163515 1769081 Live/Registered |
GENENTECH, INC. 1991-05-03 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.