OLOPATADINE HYDROCHLORIDE solution
OLOPATADINE HYDROCHLORIDE by
Drug Labeling and Warnings
OLOPATADINE HYDROCHLORIDE by is a Otc medication manufactured, distributed, or labeled by WALGREEN CO., Aurobindo Pharma Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Drug Facts
- Purpose
- Use
- Warnings
- Do not use
- When using this product
- Stop use and ask a doctor if you experience:
- Keep out of reach of children.
- Directions
- Other information
- Inactive ingredients
- Questions?
-
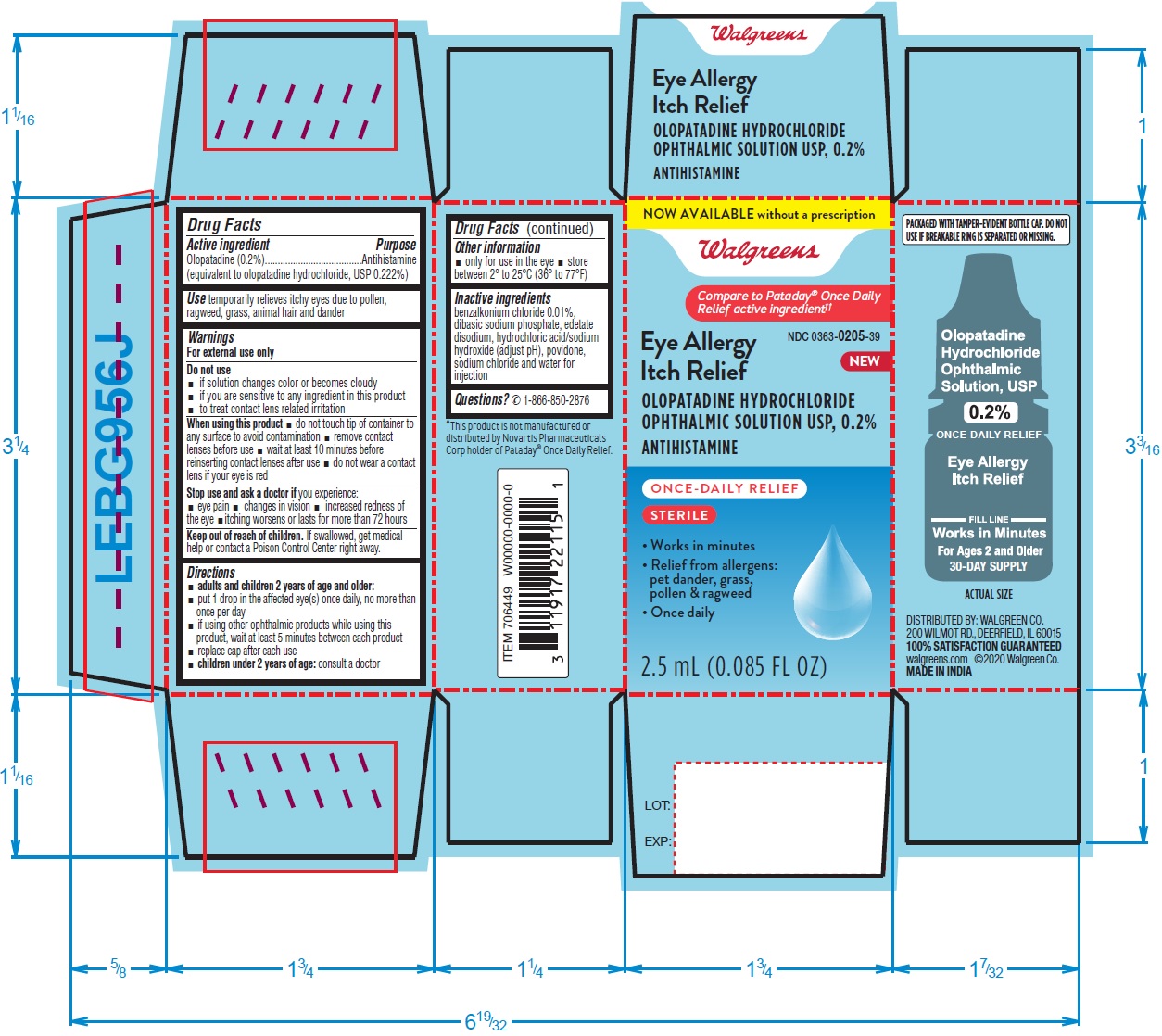
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL-0.2% (2.5 mL Container)
Walgreens NDC: 0363-8022-39
Eye Allergy Itch Relief
OLOPATADINE HYDROCHLORIDE OPHTHALMIC
SOLUTION USP, 0.2%
ANTIHISTAMINE
Once Daily 2.5 mL (0.085 FL OZ)

-
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL-0.2% (2.5 mL Container Carton)
ORIGINAL PRESCRIPTION STRENGTH
NDC: 0363-8022-39
Walgreens
Compare to the active ingredient in
Pataday® Once Daily Relief‡‡
Eye Allergy
Itch Relief
OLOPATADINE HYDROCHLORIDE
OPHTHALMIC SOLUTION USP, 0.2%
ANTIHISTAMINE
Once Daily
Works in minutes
Relief from allergens:
pet dander, grass,
pollen & ragweed
Sterile
2.5 mL (0.085 FL OZ)
-
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL-0.2% (2.5 mL Container Carton) Twin Pack
TWIN PACK
ORINIGAL PRESCRIPTION STRENGTH
NDC: 0363-8022-41
Walgreens
Compare to the active ingredient in
Pataday® Once Daily Relief‡‡Eye Allergy
Itch Relief
OLOPATADINE HYDROCHLORIDE
OPHTHALMIC SOLUTION USP, 0.2%
ANTIHISTAMINEOnce Daily
Works in minutes
Relief from allergens:
pet dander, grass,
pollen & ragweed
SterileTWO 2.5 mL BOTTLES
(0.085 FL OZ each)

-
INGREDIENTS AND APPEARANCE
OLOPATADINE HYDROCHLORIDE
olopatadine hydrochloride solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 0363-8022 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OLOPATADINE HYDROCHLORIDE (UNII: 2XG66W44KF) (OLOPATADINE - UNII:D27V6190PM) OLOPATADINE HYDROCHLORIDE 2 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) SODIUM PHOSPHATE, DIBASIC, UNSPECIFIED FORM (UNII: GR686LBA74) EDETATE DISODIUM (UNII: 7FLD91C86K) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM CHLORIDE (UNII: 451W47IQ8X) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0363-8022-39 1 in 1 CARTON 07/15/2020 1 2.5 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC: 0363-8022-41 2 in 1 CARTON 07/15/2020 2 2.5 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209995 07/15/2020 Labeler - WALGREEN CO. (008965063) Registrant - EUGIA Pharma Specialities Limited (650804896) Establishment Name Address ID/FEI Business Operations Eugia Pharma Specialities Limited 650498244 ANALYSIS(0363-8022) , MANUFACTURE(0363-8022)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.