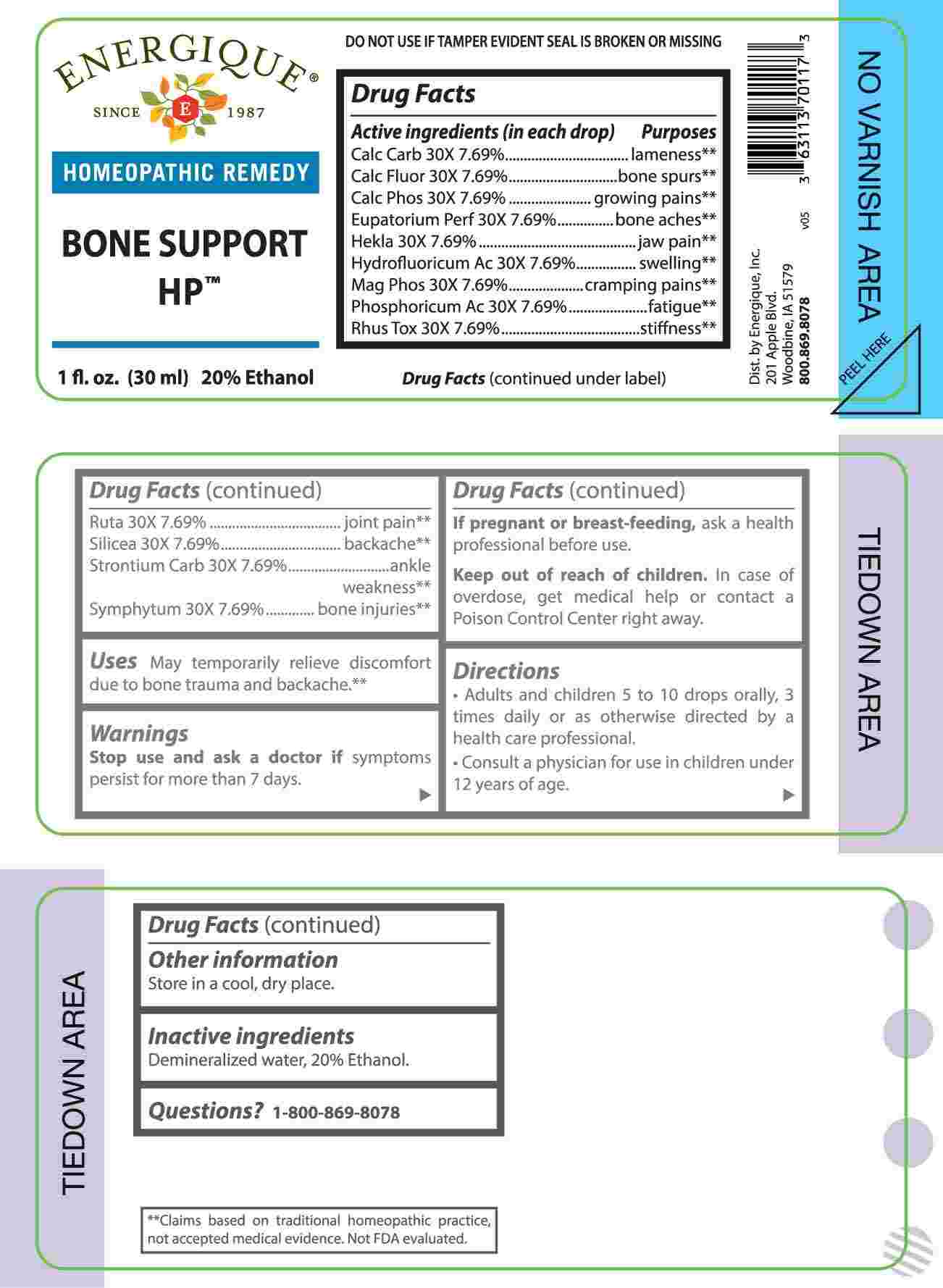
BONE SUPPORT HP- calcarea carbonica, calcarea fluorica, calcarea phosphorica, eupatorium perfoliatum, hekla lava, hydrofluoricum acidum, magnesia phosphorica, phosphoricum acidum, rhus tox, ruta graveolens, silicea, strontium carbonicum, symphytum officinale liquid
Bone Support by
Drug Labeling and Warnings
Bone Support by is a Homeopathic medication manufactured, distributed, or labeled by Energique, Inc., Apotheca Company. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
ACTIVE INGREDIENTS:
(in each drop): Calcarea Carbonica 30X 7.69%, Calcarea Fluorica 30X 7.69%, Calcarea Phosphorica 30X 7.69%, Eupatorium Perfoliatum 30X 7.69%, Hekla Lava 30X 7.69%, Hydrofluoricum Acidum 30X 7.69%, Magnesia Phosphorica 30X 7.69%, Phosphoricum Acidum 30X 7.69%, Rhus Tox 30X 7.69%, Ruta Graveolens 30X 7.69%, Silicea 30X 7.69%, Strontium Carbonicum 30X 7.69%, Symphytum Officinale 30X 7.69%.
-
PURPOSE:
Calcarea Carbonica - lameness,** Calcarea Fluorica – bone spurs,** Calcarea Phosphorica – growing pains,** Eupatorium Perfoliatum – bone aches,** Hekla Lava – jaw pain,** Hydrofluoricum Acidum - swelling,** Magnesia Phosphorica – cramping pains,** Phosphoricum Acidum - fatigue,** Rhus Tox - stiffness,** Ruta Graveolens – joint pain,** Silicea - backache,** Strontium Carbonicum – ankle weakness,** Symphytum Officinale – bone injuries.**
**Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
- USES:
-
WARNINGS:
Stop use and ask a doctor if symptons persist for more than 7 days.
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
DO NOT USE IF TAMPER EVIDENT SEAL IS BROKEN OR MISSING
Store in a cool, dry place.
- KEEP OUT OF REACH OF CHILDREN:
- DIRECTIONS:
- INACTIVE INGREDIENTS:
- QUESTIONS:
- PACKAGE LABEL DISPLAY:
-
INGREDIENTS AND APPEARANCE
BONE SUPPORT HP
calcarea carbonica, calcarea fluorica, calcarea phosphorica, eupatorium perfoliatum, hekla lava, hydrofluoricum acidum, magnesia phosphorica, phosphoricum acidum, rhus tox, ruta graveolens, silicea, strontium carbonicum, symphytum officinale liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 44911-0663 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OYSTER SHELL CALCIUM CARBONATE, CRUDE (UNII: 2E32821G6I) (OYSTER SHELL CALCIUM CARBONATE, CRUDE - UNII:2E32821G6I) OYSTER SHELL CALCIUM CARBONATE, CRUDE 30 [hp_X] in 1 mL CALCIUM FLUORIDE (UNII: O3B55K4YKI) (FLUORIDE ION - UNII:Q80VPU408O) CALCIUM FLUORIDE 30 [hp_X] in 1 mL TRIBASIC CALCIUM PHOSPHATE (UNII: 91D9GV0Z28) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM CATION 30 [hp_X] in 1 mL EUPATORIUM PERFOLIATUM FLOWERING TOP (UNII: 1W0775VX6E) (EUPATORIUM PERFOLIATUM FLOWERING TOP - UNII:1W0775VX6E) EUPATORIUM PERFOLIATUM FLOWERING TOP 30 [hp_X] in 1 mL HEKLA LAVA (UNII: C21158IIRK) (HEKLA LAVA - UNII:C21158IIRK) HEKLA LAVA 30 [hp_X] in 1 mL HYDROFLUORIC ACID (UNII: RGL5YE86CZ) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 30 [hp_X] in 1 mL MAGNESIUM PHOSPHATE, DIBASIC TRIHYDRATE (UNII: HF539G9L3Q) (MAGNESIUM CATION - UNII:T6V3LHY838) MAGNESIUM PHOSPHATE, DIBASIC TRIHYDRATE 30 [hp_X] in 1 mL PHOSPHORIC ACID (UNII: E4GA8884NN) (PHOSPHORIC ACID - UNII:E4GA8884NN) PHOSPHORIC ACID 30 [hp_X] in 1 mL TOXICODENDRON PUBESCENS LEAF (UNII: 6IO182RP7A) (TOXICODENDRON PUBESCENS LEAF - UNII:6IO182RP7A) TOXICODENDRON PUBESCENS LEAF 30 [hp_X] in 1 mL RUTA GRAVEOLENS FLOWERING TOP (UNII: N94C2U587S) (RUTA GRAVEOLENS FLOWERING TOP - UNII:N94C2U587S) RUTA GRAVEOLENS FLOWERING TOP 30 [hp_X] in 1 mL SILICON DIOXIDE (UNII: ETJ7Z6XBU4) (SILICON DIOXIDE - UNII:ETJ7Z6XBU4) SILICON DIOXIDE 30 [hp_X] in 1 mL STRONTIUM CARBONATE (UNII: 41YPU4MMCA) (STRONTIUM CATION - UNII:37077S2C93) STRONTIUM CARBONATE 30 [hp_X] in 1 mL COMFREY ROOT (UNII: M9VVZ08EKQ) (COMFREY ROOT - UNII:M9VVZ08EKQ) COMFREY ROOT 30 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 44911-0663-1 30 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 11/09/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 11/09/2022 Labeler - Energique, Inc. (789886132) Registrant - Apotheca Company (844330915) Establishment Name Address ID/FEI Business Operations Apotheca Company 844330915 manufacture(44911-0663) , api manufacture(44911-0663) , label(44911-0663) , pack(44911-0663)
Trademark Results [Bone Support]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 BONE SUPPORT 77215344 not registered Dead/Abandoned |
ISI Brands Inc. 2007-06-26 |
 BONE SUPPORT 76511611 not registered Dead/Abandoned |
Bone Support AB 2003-04-17 |
 BONE SUPPORT 75570378 not registered Dead/Abandoned |
TWIN LABORATORIES INC 1998-10-15 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.