BLOCKAID- lidocaine, tetracaine cream
Blockaid by
Drug Labeling and Warnings
Blockaid by is a Otc medication manufactured, distributed, or labeled by SofTap Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- DRUG FACTS
- PURPOSE
-
OTHER INGREDIENTS
Deionized water, caprylc/capric triglyceride,stearic acid, glycerin, glyceryl stearate, petrolatum, safflower oil, shea butter, polysorbate 60, stearyl alcohol, sweet almond oil, tocopheryl acetate, bht, propylene glycol, sodium carbonate, phenoxyethanol, methylparaben, polyparaben. May contain: FD&C Green #3, FD&C Yellow #5, FD&C Blue #1
- USES
- DO NOT USE
-
KEEP OUT OF
The eye and mouth. If accidental contact occurs, you may feel burning or stinging. Wash with water or eyewash immediately. If your eyes have pain, blurry vision, extreme sensitivity to light, or a feeling of sand in the eye, contact an eye care physician immediately.
WHEN USING THIS PRODUCT
You may not feel pain. Avoid sources of heat or injury.
You may have temporary rash, redness, swelling, or itching
Contact your physician promptly if you notice any unusual effects such as dizziness or drowsiness, difficulty breathing, trembling, chest pain, or irregular heartbeat.Made in the USA for SofTap
550 N Canyons Pkwy
Livermore, CA 94551
www.softaps.com

-
DIRECTIONS
For external use only. Prior to application, a sensitivity test is advised. Before applying, cleanse the skin with an alcohol-free or acetone-free cleanser. Apply 1-2 cc to the desired area for up to 6 times per day. Discontinue use if allergic reaction occurs. Store in a cool dark place (up to 80 degrees F) or refrigerate.
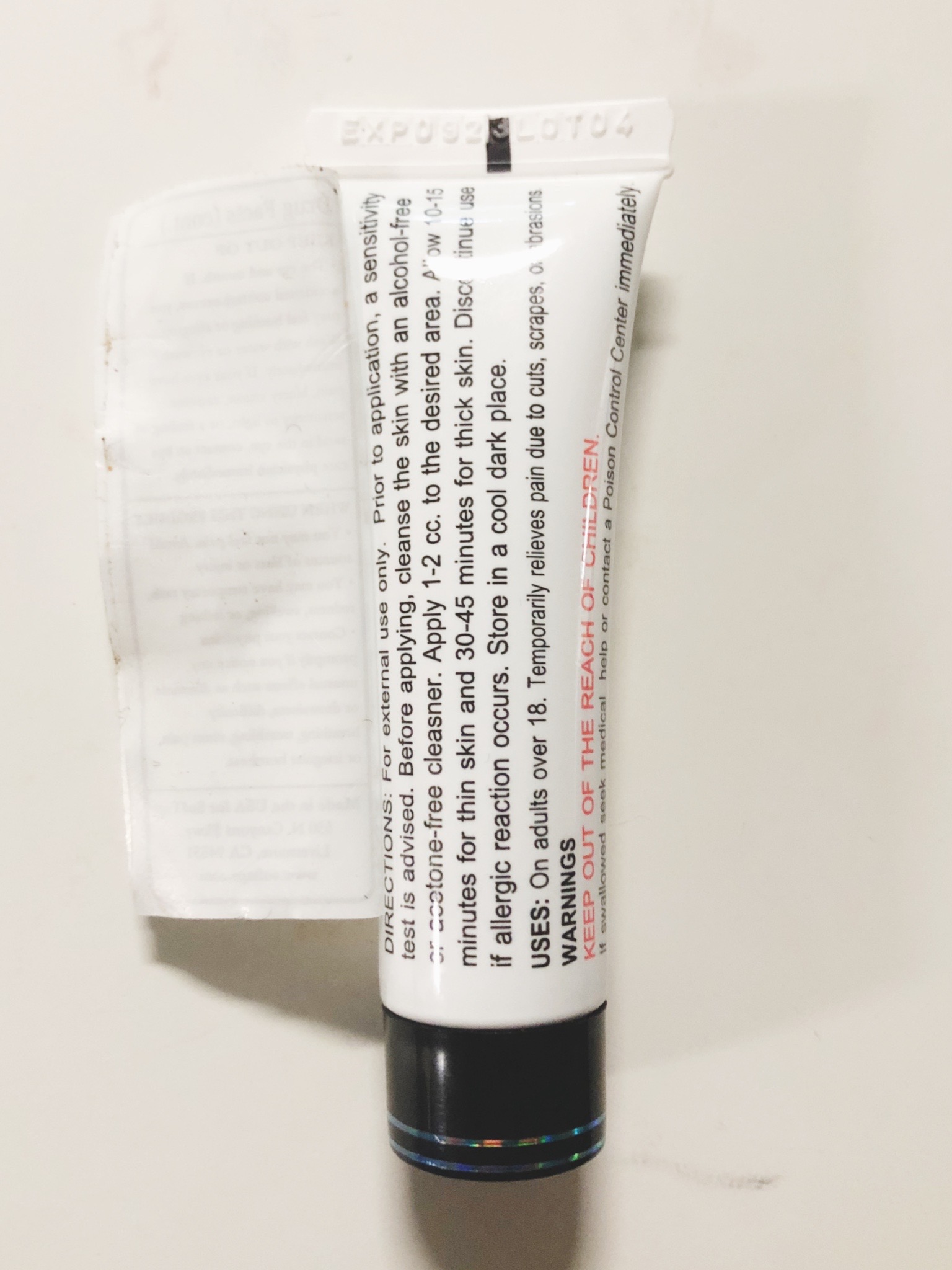
- WARNINGS
- WARNINGS
- BLOCKAID
-
INGREDIENTS AND APPEARANCE
BLOCKAID
lidocaine, tetracaine creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 82718-1110 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TETRACAINE (UNII: 0619F35CGV) (TETRACAINE - UNII:0619F35CGV) TETRACAINE 0.02 g in 10 g LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 0.03 g in 10 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PHENOXYETHANOL (UNII: HIE492ZZ3T) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) METHYLPARABEN (UNII: A2I8C7HI9T) FD&C GREEN NO. 3 (UNII: 3P3ONR6O1S) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) SODIUM CARBONATE (UNII: 45P3261C7T) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) GLYCERIN (UNII: PDC6A3C0OX) PETROLATUM (UNII: 4T6H12BN9U) STEARIC ACID (UNII: 4ELV7Z65AP) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) ALMOND OIL (UNII: 66YXD4DKO9) PROPYLPARABEN (UNII: Z8IX2SC1OH) SAFFLOWER OIL (UNII: 65UEH262IS) SHEA BUTTER (UNII: K49155WL9Y) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) POLYSORBATE 60 (UNII: CAL22UVI4M) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) Product Characteristics Color blue (light blue) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 82718-1110-1 10 g in 1 TUBE; Type 0: Not a Combination Product 05/06/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 05/06/2022 Labeler - SofTap Inc (780568353)
Trademark Results [Blockaid]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 BLOCKAID 98120245 not registered Live/Pending |
Blockaid Ltd. 2023-08-07 |
 BLOCKAID 88900141 not registered Live/Pending |
Zenwise LLC 2020-05-04 |
 BLOCKAID 85498545 4973352 Live/Registered |
Blockaid Pty Ltd 2011-12-19 |
 BLOCKAID 78798998 not registered Dead/Abandoned |
Great Southern Wood Preserving, Inc. 2006-01-25 |
 BLOCKAID 78699743 3353596 Live/Registered |
Acoustics First Corporation 2005-08-24 |
 BLOCKAID 78432237 3034674 Live/Registered |
Alexis A. Lawson and Michael R. LawsonPartnership, The 2004-06-09 |
 BLOCKAID 76581996 not registered Dead/Abandoned |
Great Southern Wood Preserving, Inc. 2004-03-17 |
 BLOCKAID 75936284 2592436 Dead/Cancelled |
INVERNESS MEDICAL - BIOSTAR INC. INVERNESS MEDICAL PROFESSIONAL DIAGNOSTICS 2000-03-06 |
 BLOCKAID 74556501 1908310 Live/Registered |
Biomet, Inc. 1994-08-02 |
 BLOCKAID 73664526 1472503 Dead/Cancelled |
RIDDELL, INC. 1987-06-04 |
 BLOCKAID 72231113 0827088 Dead/Expired |
GLIDDEN COMPANY, THE 1965-10-22 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.

