Clearasil ®Clear Skin Everyday Essentials
Clearasil Clear Skin Everyday Essentials by
Drug Labeling and Warnings
Clearasil Clear Skin Everyday Essentials by is a Otc medication manufactured, distributed, or labeled by RB Health (US) LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
CLEARASIL CLEAR SKIN EVERYDAY ESSENTIALS- salicylic acid and benzoyl peroxide
RB Health (US) LLC
----------
Clearasil ®Clear Skin Everyday Essentials
Warnings
For external use only
When using this product
- avoid contact with the eyes. If product gets into the eyes, rinse thoroughly with water.
- skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
- limit use to the face and neck
Directions
- wet face
- dispense product into hands and massage gently onto face and neck, avoiding the delicate eye area
- rinse thoroughly with warm water and pat dry
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor
- if bothersome dryness or peeling occurs, reduce application to once a day or every other day
Inactive ingredients
water, PPG-15 stearyl ether, hydrated silica, glycerin, stearyl alcohol, cetyl betaine, distearyldimonium chloride, sodium lauryl sulfate, cetyl alcohol, alcohol denat, steareth-21, sodium chloride, behenyl alcohol, synthetic wax, steareth-2, xanthan gum, fragrance, MEK, disodium EDTA, isopropyl alcohol, BHT, lavandula stoechas extract, methylchloroisothiazolinone, helichrysum italicum extract, methylisothiazolinone, cistus monspeliensis extract, mica, ferric ferrocyanide, titanium dioxide, FD&C blue no. 1
Warnings
For external use only
When using this product
- avoid contact with the eyes. If product gets into the eyes, rinse thoroughly with water.
- skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
- limit use to face and neck
Directions
- clean the skin thoroughly before applying this product
- wipe a pad over face and neck to cover the entire affected area with a thin layer one to three times daily
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor
- if bothersome dryness or peeling occurs, reduce application to once a day or every other day
Other information
- do not flush in toilet
- store in a cool, dry place but do not freeze
- keep jar tightly closed
Inactive ingredients
water, alcohol denat, isoceteth-20, aloe barbadensis leaf juice, sodium hydroxide, triethanolamine, allantoin, fragrance, menthol, disodium EDTA, potassium sorbate, sodium benzoate, citric acid
Warnings
For external use only
When using this product
- avoid unnecessary sun exposure and use a sunscreen
- avoid contact with the eyes, lips and mouth
- avoid contact with hair and dyed fabrics, which may be bleached by this product
- skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
- skin irritation may occur, characterized by redness, burning, itching, peeling or possibly swelling. Irritation may be reduced by using the product less frequently or in a lower concentration.
Directions
- clean the skin thoroughly before applying this product
- cover the entire affected area with a thin layer one to three times daily
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor
- if bothersome dryness or peeling occurs, reduce application to once a day or every other day
- if going outside, apply sunscreen after using this product. If irritation or sensitivity develops, stop use of both products and ask a doctor.
Other information
- keep outer box for full product information
- keep tightly closed
- store at 20-25°C (68-77°F)
Inactive ingredients
water, propylene glycol, aluminum hydroxide, bentonite, glyceryl stearate SE, PEG-12, isopropyl myristate, methylparaben, carbomer, potassium hydroxide, propylparaben
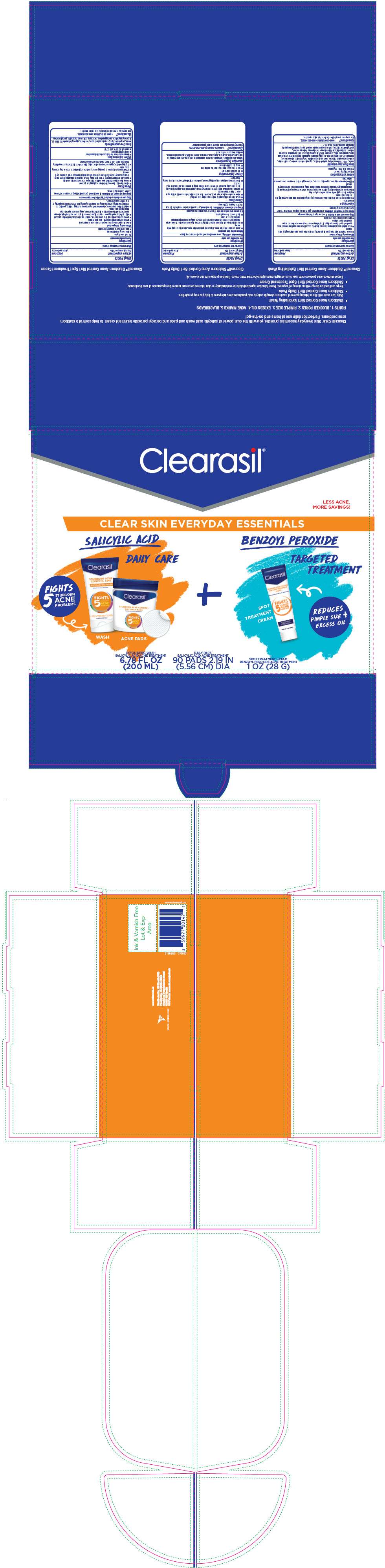
PRINCIPAL DISPLAY PANEL - Kit Carton
Clearasil ®
LESS ACNE.
MORE SAVINGS!
CLEAR SKIN EVERYDAY ESSENTIALS
SALICYLIC ACID
DAILY CARE
FIGHTS
5
STUBBORN
ACNE
PROBLEMS
WASH
ACNE PADS
+
BENZOYL PEROXIDE
TARGETED
TREATMENT
REDUCES
PIMPLE SIZE +
EXCESS OIL
SPOT
TREATMENT
CREAM
EXFOLIATING WASH
SALICYLIC ACID ACNE TREATMENT
6.78 FL OZ
(200 ML)
DAILY PADS
SALICYLIC ACID ACNE TREATMENT
90 PADS 2.19 IN
(5.56 CM) DIA
SPOT TREATMENT CREAM
BENZOYL PEROXIDE ACNE TREATMENT
1 OZ (28 G)
| CLEARASIL CLEAR SKIN EVERYDAY ESSENTIALS
salicylic acid and benzoyl peroxide kit |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - RB Health (US) LLC (081049410) |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.