YUVIWEL- navepegritide kit
Yuviwel by
Drug Labeling and Warnings
Yuviwel by is a Prescription medication manufactured, distributed, or labeled by Ascendis Pharma Endocrinology, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use YUVIWEL safely and effectively. See full prescribing information for YUVIWEL.
YUVIWEL® (navepegritide) for injection, for subcutaneous use
Initial U.S. Approval: 2026INDICATIONS AND USAGE
YUVIWEL is a C-type natriuretic peptide (CNP) analog indicated to increase linear growth in pediatric patients 2 years of age and older with achondroplasia with open epiphyses. (1)
This indication is approved under accelerated approval based on an improvement in annualized growth velocity. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s). (1)
DOSAGE AND ADMINISTRATION
- Administer once-weekly by subcutaneous injection. Dosage is based on body weight. (2.1)
- Periodically monitor growth and adjust dose according to body weight. Discontinue when no further growth potential, as indicated by epiphyseal closure. (2.2)
- See Full Prescribing Information for instructions on preparation and administration. (2.3, 2.4)
DOSAGE FORMS AND STRENGTHS
For injection: 1.3 mg, 2.8 mg, and 5.5 mg as a lyophilized powder in single-dose vial for reconstitution. (3)
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
Risk of Low Blood Pressure: Transient decreases in blood pressure have been reported with a once daily CNP analog. Advise patients to contact their healthcare provider if they experience symptoms of decreased blood pressure while being treated with YUVIWEL. (5.1)
ADVERSE REACTIONS
Most common adverse reactions (≥ 5%): vomiting, injection-site reaction, pain in extremity, and nausea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Ascendis Pharma at 1-844-442-7236 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
Renal Impairment: Not recommended for patients with moderate or severe renal impairment (eGFR < 60 mL/min/1.73 m2). (8.6)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 2/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage and Administration
2.2 Monitor Growth
2.3 Preparation of YUVIWEL for Administration
2.4 Administration Instructions
2.5 Missed Dose
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Low Blood Pressure
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.6 Renal Impairment
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.6 Immunogenicity
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
YUVIWEL® is indicated to increase linear growth in pediatric patients 2 years of age and older with achondroplasia with open epiphyses. This indication is approved under accelerated approval based on an improvement in annualized growth velocity [see Clinical Studies (14)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage and Administration
The recommended once-weekly dosage of YUVIWEL is based on the patient´s body weight (see Table 1).
YUVIWEL is administered by subcutaneous injection. YUVIWEL must be reconstituted prior to use [see Dosage and Administration (2.3)].
Table 1: Recommended YUVIWEL Weekly Dosage and Injection Volume Patient Body weight Weekly Dose Injection Volume Vial Strength for Reconstitution* - * The concentration of navepegritide is 2.2 mg/mL in a reconstituted 1.3 mg vial; 4.6 mg/mL in a reconstituted 2.8 mg vial; and 5.5 mg/mL in a reconstituted 5.5 mg vial.
8 to 9.9 kg 0.88 mg 0.4 mL 1.3 mg 10 to 13.4 kg 1.2 mg 0.55 mL 13.5 to 17.5 kg 1.6 mg 0.35 mL 2.8 mg 17.6 to 23 kg 2.1 mg 0.45 mL 23.1 to 30.5 kg 2.8 mg 0.6 mL 30.6 to 41.2 kg 3.6 mg 0.65 mL 5.5 mg 41.3 to 55.9 kg 5 mg 0.9 mL 56 to 73.5 kg 6.6 mg 1.2 mL (use 2 Kits)
Administer 0.6 mL from each Kit73.6 to 90 kg 8.8 mg 1.6 mL (use 2 Kits)
Administer 0.8 mL from each KitSwitching from Daily C-type Natriuretic Peptide (CNP) Analog
Start once-weekly YUVIWEL on the day after completing the last dose of daily CNP therapy.
2.2 Monitor Growth
Periodically monitor the patient's growth and adjust the dosage according to the actual body weight [see Dosage and Administration (2.1)]. Discontinue YUVIWEL upon confirmation of no further growth potential, indicated by closure of the epiphyses.
2.3 Preparation of YUVIWEL for Administration
Patients and caregivers who will administer YUVIWEL should receive appropriate training by a healthcare provider prior to use. Refer to the Instructions for Use for complete preparation and administration instructions with illustrations.
Before administration, reconstitute YUVIWEL using the provided prefilled diluent syringe containing Sterile Water for Injection as described below.
- Needles and syringes supplied with YUVIWEL are for single use only.
- If the product was refrigerated, allow the YUVIWEL vial and the prefilled diluent syringe to reach room temperature (about 30 minutes) before reconstitution.
- Screw a preparation needle onto the prefilled diluent syringe and inject the entire diluent volume into the vial. Shake the vial up and down for 15 seconds. Do not swirl or roll. The reconstituted YUVIWEL vial should stand at room temperature for 5 minutes after shaking. Dispose the diluent syringe with attached preparation needle immediately after injecting the diluent into the vial.
- YUVIWEL should be visually inspected for particles or discoloration prior to administration, whenever solution and container permit. Once reconstituted, YUVIWEL is a clear and colorless solution. Do not use the solution if it is discolored, cloudy or contains visible particles. Air bubbles may be seen, and this is normal.
- Screw a new preparation needle onto the injection syringe and withdraw the prescribed injection volume from the reconstituted vial. Remove air from the withdrawn dose volume before continuing and ensure the withdrawn dose volume is correct after removing any air.
- Remove and dispose the preparation needle from the injection syringe.
- Screw the injection needle onto the injection syringe before administration.
- Two Kits are needed to achieve a complete dose for patients with body weight 56 kg or greater, where the prescribed injection volume is greater than 1 mL.
- Reconstituted YUVIWEL can be stored at room temperature up to 30°C (86°F) for up to 4 hours.
2.4 Administration Instructions
Refer to the Instructions for Use for complete administration instructions with illustrations.
- Using the prepared syringe, administer the prescribed injection volume [see Dosage and Administration (2.1, 2.3)].
- Administer YUVIWEL subcutaneously in the abdominal region (2 inches from the belly button) or thighs. If a caregiver is administering YUVIWEL, subcutaneous injection in the buttocks or back of the upper arm is also acceptable.
- Rotate injection sites. Do not give an injection into sites other than described above. Avoid injecting where the skin is red, swollen, or scarred.
- Administer YUVIWEL once weekly on the dosing day, at any time of day.
- All YUVIWEL components are for single use only. Do not draw up more than one injection volume, as specified in Table 1, from a vial. For injection volumes greater than 1 mL, use two Kits to achieve a complete dose. The two injections must be given one after the other, using different injection sites.
- Discard the unused reconstituted solution.
2.5 Missed Dose
- If a dose of YUVIWEL is missed, administer the missed dose as soon as possible but not more than 2 days after the missed dosing day.
- To avoid missed doses, YUVIWEL can be taken up to 2 days before or 2 days after the scheduled dosing day. Resume once-weekly dosing for the next dose at the previously scheduled dosing day.
- If more than 2 days have passed from the dosing day, skip the missed dose and administer the dose on the next regularly scheduled day.
- At least 5 days should elapse between doses.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Low Blood Pressure
Transient decreases in blood pressure have been reported with a once daily CNP analog. Subjects with hemodynamically significant cardiovascular disease were excluded from participation in navepegritide clinical trials. Advise patients to contact their healthcare provider if they experience symptoms of decreased blood pressure (e.g., dizziness, fatigue and/or nausea) while being treated with YUVIWEL.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of YUVIWEL was evaluated in pediatric patients with achondroplasia in two randomized, placebo-controlled trials of navepegritide. Trial 1 included a 52-week, randomized, double-blind, placebo-controlled period, followed by a 52-week, single-arm, open-label extension (OLE) period. In Trial 1, 84 pediatric participants with achondroplasia (mean age 5.7 years; range: 2 to 12 years) were randomized to subcutaneous navepegritide 0.1 mg/kg/week (n = 57) or placebo (n = 27) [see Clinical Studies (14)]. Trial 2 included a randomized, double-blind, placebo-controlled dose-finding period. In Trial 2, 57 pediatric participants with achondroplasia (mean age 5.9 years; range: 2 to 10 years) were randomized 3:1 to subcutaneous navepegritide 0.006, 0.02, 0.05, or 0.1 mg/kg or placebo for 52 weeks. At Week 52, all participants transitioned to an OLE period during which they received navepegritide 0.1 mg/kg/week for 104 weeks.
The adverse reaction rates for navepegritide were derived from pediatric participants with achondroplasia who received navepegritide 0.1 mg/kg/week or placebo during the double-blind period of Trials 1 and 2. Of the dosages evaluated in Trials 1 and 2, 0.1 mg/kg/week is most similar to the approved weight-based dosage listed in Table 1.
Adverse reactions reported in the placebo-controlled pooled periods of Trials 1 and 2 in ≥ 5% of navepegritide-treated patients and at an incidence at least 2% greater than with placebo are presented in Table 2.
Table 2: Adverse Reactions Reported in ≥ 5% of Participants Treated with Navepegritide 0.1 mg/kg/week and ≥ 2% Higher Than Placebo During the Placebo-Controlled Period of Trials 1 and 2 Adverse Reaction NAVEPEGRITIDE 0.1 mg /kg/week
N = 68
n (%)Placebo
N = 42
n (%)- * Includes injection-site swelling, injection-site erythema, injection-site bruising, injection-site reaction, injection-site pruritus, injection-site discoloration, injection-site hemorrhage, injection-site pain, injection-site vesicles, and injection-site edema.
Vomiting 14 (21) 6 (14) Injection-site reaction* 13 (19) 6 (14) Pain in extremity 8 (12) 3 (7) Nausea 4 (6) 0 Injection-Site Reactions
During the 52-week double-blind period of Trials 1 and 2, 13 of 68 (19%) participants receiving navepegritide 0.1 mg/kg/week experienced a total of 25 events of injection-site reactions, while 6 of 42 (14%) participants receiving placebo experienced a total of 6 events of injection site reactions, corresponding to 0.4 events per person year exposure and 0.2 events per person year exposure, respectively.
Other Adverse Reactions from Trials 1 and 2 (Pooled)
Hypertrichosis
Hypertrichosis was reported in 2 of 68 patients (3%) receiving navepegritide 0.1 mg/kg/week compared to none receiving placebo in the double-blind periods of Trials 1 and 2. Cases presented as localized hair growth at injection sites or generalized increased body hair growth affecting limbs, back, or shoulders. To reduce the risk of local skin changes, rotate the site of injection with each dose.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data on the use of YUVIWEL in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. In animal reproduction studies, subcutaneous administration of navepegritide during the period of organogenesis in pregnant rats and rabbits resulted in no impact on embryo-fetal survival or congenital malformations at doses up to 10- and 7-fold, respectively, the exposure at the maximum recommended human dose (MRHD) (see Data).
Achondroplasia is an autosomal dominant genetic disorder with 100% penetrance. Therefore, there is a 50% risk for a parent with achondroplasia to have a child with achondroplasia. The estimated background risk of birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Animal Data
In an embryo-fetal developmental toxicity study in pregnant rats, navepegritide was administered subcutaneously during the period of organogenesis (gestation day 6 to 20) at doses from 0.45 to 1.3 mg/kg/day. There was no effect on embryo-fetal survival, fetal toxicity, or embryo-fetal development up to the highest dose tested, corresponding to 10-fold the exposure at the MRHD [based on area under the curve (AUC)].
In an embryo-fetal developmental toxicity study in pregnant rabbits, navepegritide was administered subcutaneously during the period of organogenesis (gestation day 7 to 27) at doses from 0.3 to 0.9 mg/kg/every fourth day. There was no effect on embryo-fetal survival, fetal toxicity, or congenital malformations up to the highest dose tested, corresponding to 7-fold the exposure at the MRHD (based on AUC).
8.2 Lactation
Risk Summary
There is no information available regarding the presence of navepegritide in human milk or regarding the potential effects on milk production or on the breastfed newborn/infant. High molecular weight therapeutic compounds, including navepegritide, are expected to have low passage into human milk. Further, no or low anticipated oral absorption of navepegritide will limit any systemic bioavailability in the breastfed newborn/infant. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for YUVIWEL and any potential adverse effects on the breastfed infant from YUVIWEL or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of YUVIWEL to increase linear growth have been established in pediatric patients aged 2 years and older with achondroplasia with open epiphyses.
Use of YUVIWEL for this indication is supported by evidence from a 52-week, randomized, placebo-controlled trial in 84 pediatric patients with achondroplasia [see Adverse Reactions (6.1), Clinical Studies (14)].
The safety and effectiveness of YUVIWEL in pediatric patients less than 2 years of age have not been established.
8.6 Renal Impairment
YUVIWEL is not recommended for patients with moderate or severe renal impairment (eGFR < 60 mL/min/1.73 m2). The recommended dosage for patients with mild renal impairment (eGFR ≥ 60 mL/min/1.73 m2) is the same as the recommended dosage for patients with normal renal function [see Clinical Pharmacology (12.3)].
-
11 DESCRIPTION
YUVIWEL for injection contains navepegritide, a C-type natriuretic peptide (CNP) analog. Navepegritide is a prodrug of active CNP consisting of a CNP moiety transiently conjugated to two branched 20 kDa methoxy polyethylene glycol (mPEG) moieties via a proprietary TransCon® Linker. The amino acid sequence of the CNP moiety is identical to the 38 amino acid sequence of residues 89-126 of human CNP. The structural formula of navepegritide is as follows:
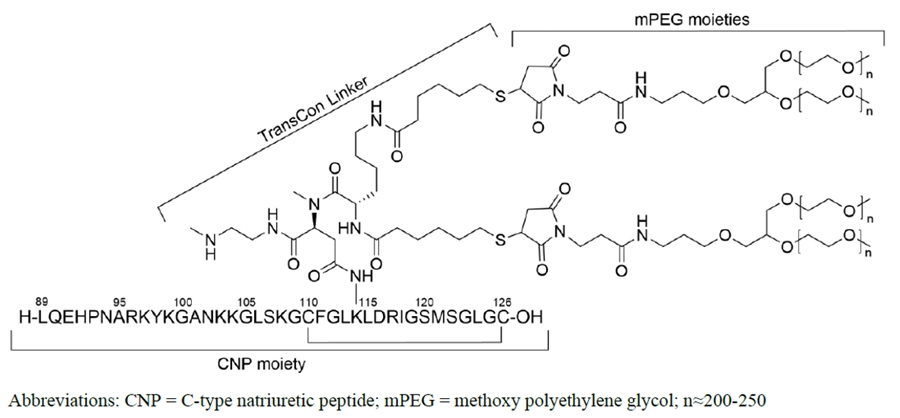
The molecular formula is C231H386N64O67S5 + 4 × (C2H4O)n, where n is between 200 and 250. The average molecular weight is approximately 45 kDa.
YUVIWEL is provided as a sterile, lyophilized white to off-white powder for reconstitution to a colorless solution with Sterile Water for Injection, USP (diluent). The diluent for reconstitution of YUVIWEL is provided in a prefilled syringe.
The compositions of YUVIWEL are shown in Table 3.
Table 3: Content of YUVIWEL Strength Composition of YUVIWEL (gross content per vial)* CNP(89-126)
Concentration After Reconstitution- * An overfill is included in the vial to compensate for loss in the vial and during transfer.
1.3 mg/vial Navepegritide equivalent to 1.9 mg CNP(89-126), succinic acid (1.0 mg), trehalose dihydrate (72.2 mg), tromethamine and hydrochloric acid (q.s. for adjustment to pH 5.0) 2.2 mg/mL 2.8 mg/vial Navepegritide equivalent to 4.0 mg CNP(89-126), succinic acid (1.0 mg), trehalose dihydrate (66.7 mg), tromethamine and hydrochloric acid (q.s. for adjustment to pH 5.0) 4.6 mg/mL 5.5 mg/vial Navepegritide equivalent to 6.9 mg CNP(89-126), succinic acid (1.5 mg), trehalose dihydrate (91.9 mg), tromethamine and hydrochloric acid (q.s. for adjustment to pH 5.0) 5.5 mg/mL -
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
CNP released from navepegritide has the same receptor binding affinity and activity as endogenous CNP. CNP binds to natriuretic peptide receptor-B (NPR-B), which inhibits the mitogen activated protein kinase signaling (MAPK) pathway.
Achondroplasia is caused by a gain-of-function variant in the fibroblast growth factor receptor 3 (FGFR3) leading to overactive downstream signaling. The overly active FGFR3 inhibits endochondral ossification leading to short stature and skeletal dysplasia. Like endogenous CNP, CNP released from navepegritide binds to NPR-B, stimulating an increase in cyclic guanosine monophosphate (cGMP) and signaling through protein kinase G, resulting in an inhibition of the MAPK signaling pathway and thereby antagonizing the overactive FGFR3 signaling in achondroplasia. CNP promotes chondrocyte differentiation and proliferation, thereby stimulating skeletal bone growth in patients with achondroplasia.
12.2 Pharmacodynamics
Natriuretic Peptide Receptor B (NPR-B) Activity Marker
There was a dose-dependent (0.006 mg/kg/week to 0.1 mg/kg/week) increase in plasma cGMP levels in pediatric patients with achondroplasia following 52 weeks of once-weekly administration of navepegritide.
Cardiac Electrophysiology
At the approved recommended dose, YUVIWEL does not prolong the QT interval to any clinically relevant extent.
12.3 Pharmacokinetics
The pharmacokinetics of navepegritide, a prodrug releasing CNP following subcutaneous administration, have been investigated at single doses of 0.003 to 0.15 mg/kg in healthy adults, and at weekly doses of 0.006 to 0.1 mg/kg in patients with achondroplasia. Plasma concentrations of navepegritide and CNP released from navepegritide increased proportionally with dose within the range of 0.01 to 0.15 mg/kg.
In patients with achondroplasia administered navepegritide 0.1 mg/kg once-weekly, the predicted steady state geometric mean (CV) maximum plasma concentration (Cmax) of navepegritide was 1,360 ng/mL (8.3%) and the predicted geometric mean (CV) exposure over the weekly dosing interval AUC was 193,000 h*ng/mL (6.1%). For CNP released from navepegritide, the predicted geometric mean (CV) Cmax was 36.0 pmol/L (23%) and the predicted geometric mean (CV) AUC over the weekly dosing interval was 4,410 h*pmol/L (23%).
In patients with achondroplasia, steady state levels of navepegritide and CNP released from navepegritide were achieved after approximately 3 once-weekly doses. Median accumulation ratios of navepegritide and CNP released from navepegritide following once-weekly dose administration were 1.9 and 1.7, respectively.
Absorption
In patients with achondroplasia administered navepegritide 0.1 mg/kg once-weekly, the geometric mean (CV) time to reach maximum concentration (Tmax) of navepegritide was 43.4 hours (15%). For CNP released from navepegritide, the geometric mean (CV) Tmax was 24.4 hours (13%).
The absolute bioavailability of navepegritide following subcutaneous dose administration has not been investigated.
Distribution
The model-derived geometric mean (CV) apparent volume of distribution of navepegritide and CNP released from navepegritide was 1.8 (33%) L and 5.1 (30%) L, respectively.
Elimination
The model-derived geometric mean (CV) steady state apparent clearance of navepegritide and CNP released from navepegritide was 0.052 (33%) and 1,950 (39%) L/day, respectively.
In patients with achondroplasia administered navepegritide 0.1 mg/kg once weekly, the predicted mean apparent elimination half-life of navepegritide was 6.7 days, and the mean apparent elimination half-life of CNP released from navepegritide was 5.3 days.
Metabolism
Following subcutaneous dose administration, navepegritide releases CNP via auto-cleavage of the TransCon Linker that follows first-order kinetics, resulting in continuous systemic exposure of CNP over the weekly dosing interval.
CNP metabolism follows natural degradation pathways for peptides, resulting in small peptide fragments and amino acids.
Specific Populations
Based on a population pharmacokinetic analysis, no clinically meaningful effects on the pharmacokinetics of navepegritide were observed for age (2 to 59.6 years), sex (66% male, 34% female), race (21% Asian, 1.8% Black or African American, 75% White, and 1.8% Other), ethnicity (11% Hispanic or Latino, 87% not Hispanic or Latino, 1% not reported or unknown) and mild (eGFR: 60 to < 89 mL/min/1.73 m2) renal impairment. The effect of hepatic impairment on the pharmacokinetics of navepegritide was not studied.
Drug Interaction Studies
No in vitro assessments or clinical studies evaluating the drug-drug interaction potential of navepegritide have been conducted.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assays used. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in studies of other products.
Of 73 patients who received navepegritide 0.1 mg/kg once-weekly for up to 3 years, 30% (22/73) developed antibodies against navepegritide or CNP. These antibodies were transiently detected at a low level with no identified clinically significant effect on the pharmacokinetics, efficacy, or safety of navepegritide. The neutralizing ability of anti-drug antibodies is unknown due to the limitations of the neutralizing antibody assay.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis and Mutagenesis
Long-term animal studies to address the carcinogenic potential of navepegritide have not been conducted.
Navepegritide was not genotoxic in an in vitro bacterial reverse-mutation assay (Ames test), an in vitro human lymphocyte chromosomal aberration assay or an in vivo rat bone marrow micronucleus assay.
Impairment of Fertility
In fertility studies, navepegritide was administered by subcutaneous injection at 0.5, 1.0, and 2.0 mg/kg/week to male and female rats. Navepegritide had no effect on mating performance, fertility, litter characteristics or early embryo-fetal development up to the highest dose tested corresponding to 3-fold the MRHD in male rats (based on body surface area) and 4-fold exposure in female rats (based on AUC).
-
14 CLINICAL STUDIES
The effectiveness of YUVIWEL has been established in a clinical trial of navepegritide consisting of a randomized, double-blind, placebo-controlled 52-week period, followed by a single-arm 52-week OLE period (Trial 1; NCT05598320).
Trial 1 enrolled 84 treatment-naïve pediatric patients with genetically confirmed achondroplasia: 57 patients received navepegritide 0.1 mg/kg administered subcutaneously once weekly, and 27 received placebo.
The mean age at enrollment was 5.7 years (range: 2 to 12). Forty-five patients (54%) were male, and 39 patients (46%) were female. Overall, 74 patients (88%) were White, 8 patients (10%) were Asian, and 2 patients (2%) were mixed race. The patients had a mean baseline CDC-based height Z-score of -5.0 and mean baseline height of 89 cm (range: 64 to 120).
The primary efficacy endpoint was annualized growth velocity (AGV) at Week 52. Height Z-scores calculated using reference data from untreated children with achondroplasia (achondroplasia-specific height Z-score) and using reference data from the general population (CDC-based height Z-score) were also evaluated.
Treatment with once-weekly navepegritide for 52 weeks resulted in a least-squares mean treatment difference in AGV of 1.5 cm/year and a least-squares mean increase from baseline in achondroplasia-specific height Z-score of 0.3 (see Table 4).
Table 4: Growth Parameters in Pediatric Patients with Achondroplasia Treated with Navepegritide and Placebo at Week 52 in Trial 1 NAVEPEGRITIDE
(N = 57)Placebo
(N = 27)Treatment Difference
[95% CI]*p-value Note: Data presented are least-square (LS) means unless otherwise noted. Abbreviations: AGV, annualized growth velocity; CI, confidence interval; NA, not applicable. - * Treatment differences between YUVIWEL and placebo were estimated from an analysis of covariance (ANCOVA) model that included treatment, strata (defined by sex and age), baseline age, and baseline achondroplasia-specific height Z-score as covariates.
- † Calculated using the reference data from untreated patients with achondroplasia (CLARITY Natural History Study).
- ‡ Calculated using the Centers for Disease Control and Prevention (CDC) reference data from the general population.
- § The endpoint was analyzed outside of the pre-specified multiple testing strategy.
Annualized growth velocity (cm/year) 5.9 4.4 1.5 [1.0, 1.9] < 0.0001 Change from baseline Achondroplasia-specific height Z-score† 0.3 0.0 0.3 [0.2, 0.4] < 0.0001 CDC height Z-score‡ 0.1 -0.2 0.3 [0.1, 0.5] NA§ Improvements in AGV and height Z-scores were observed in navepegritide-treated patients across all predefined subgroups analyzed, including age group, sex, and region. In patients < 5 years of age, the least-squares mean treatment difference for AGV at Week 52 was 1.0 cm/year. In patients ≥ 5 years of age, the least-squares mean treatment difference was 1.8 cm/year.
Long-term Treatment Effect
All 57 patients in a 52-week randomized, double-blind, placebo-controlled Phase 2 dose-finding trial (Trial 2; NCT04085523) entered the single-arm OLE and continued treatment with navepegritide. AGV was maintained among those who had received 2 years of treatment with navepegritide 0.1 mg/kg once weekly.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
YUVIWEL (navepegritide) for injection is supplied as a lyophilized white to off-white powder in a vial, in three strengths: 1.3 mg, 2.8 mg, and 5.5 mg (see Table 5). Each YUVIWEL carton contains 4 Kits with 1 Prescribing Information, and 1 Instructions for Use.
Table 5: YUVIWEL Strengths and Contents of Each Kit Strength Contents of Each Kit (4 Kits in each carton) Carton NDC 1.3 mg vial - One 1.3 mg single-dose vial of YUVIWEL for injection with yellow cap
- One 0.8 mL Sterile Water for Injection, USP (clear, colorless prefilled diluent syringe)
- 2 single use preparation needles (21 gauge)
- 1 single use injection syringe
- 1 single use injection needle (30 gauge)
73362-201-01 2.8 mg vial - One 2.8 mg single-dose vial of YUVIWEL for injection with blue cap
- One 0.8 mL Sterile Water for Injection, USP (clear, colorless prefilled diluent syringe)
- 2 single use preparation needles (21 gauge)
- 1 single use injection syringe
- 1 single use injection needle (30 gauge)
73362-202-01 5.5 mg vial - One 5.5 mg single-dose vial of YUVIWEL for injection with violet cap
- One 1.1 mL Sterile Water for Injection, USP (clear, colorless prefilled diluent syringe)
- 2 single use preparation needles (21 gauge)
- 1 single use injection syringe
- 1 single use injection needle (30 gauge)
73362-203-01 Only use the vial, needles, and syringes that are in the Kits for handling of YUVIWEL [see Dosage and Administration (2.3)] and do not use the components for other medicinal products.
Storage and Handling
- Store YUVIWEL refrigerated between 2°C to 8°C (36°F to 46°F). Store in the original packaging until time of use to protect from light. Do not freeze.
- YUVIWEL can also be stored at room temperature up to 30°C (86°F) for up to 6 months and can be returned to refrigeration within the 6 months. Do not use YUVIWEL beyond the expiration date or 6 months after the date it was first removed from refrigeration (whichever is earlier).
- Reconstituted YUVIWEL can be stored at room temperature up to 30°C (86°F) for up to 4 hours.
- Discard unused reconstituted solution.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Administration Instructions
Provide appropriate instructions for injection to the patient/caregiver based on the YUVIWEL Instructions for Use (available at www.Yuviwel.com). Patients/caregivers and healthcare providers may also call the Ascendis Pharma Customer Support toll-free number at 1-844-442-7236 (1-844-44ASCENDIS) for assistance or additional training, if needed.
- Advise patients/caregivers to refer to the Instructions for Use that accompanies YUVIWEL for complete reconstitution and administration instructions with illustrations [see Dosage and Administration (2.3, 2.4)].
- Advise patients/caregivers to administer YUVIWEL once weekly on the dosing day, at any time of day.
- Advise patients and caregivers to rotate the site of injection to reduce the risk of injection site reactions [see Dosage and Administration (2.4)].
Missed Dose [see Dosage and Administration (2.5)]
- Advise patients/caregivers that to avoid missed doses, YUVIWEL can be taken up to 2 days before or 2 days after the scheduled dosing day.
- If a dose is missed and more than 2 days have passed from the scheduled day, advise patients/caregivers to skip the missed dose and administer the next dose on the regularly scheduled day.
- To change the regular dosing day to a different day of the week, advise patients/caregivers to ensure that at least 5 days will elapse between the last dose and the newly established regular dosing day.
Risk of Low Blood Pressure
Advise patients and/or caregivers to contact their healthcare provider if they experience symptoms of decreased blood pressure (e.g., dizziness, fatigue, and/or nausea) while being treated with YUVIWEL [see Warnings and Precautions (5.1)].
-
SPL UNCLASSIFIED SECTION
© 2026 Ascendis Pharma. All rights reserved. YUVIWEL®, Ascendis®, TransCon®, the Ascendis Pharma logo and the company logo are trademarks owned by the Ascendis Pharma Group.
Manufactured for:
Ascendis Pharma Growth Disorders A/S
Tuborg Boulevard 12
DK-2900 Hellerup, DenmarkFor information about YUVIWEL contact:
Ascendis Pharma Endocrinology, Inc.
Princeton, New Jersey 08540, USA
1-844-442-7236
www.YUVIWEL.com -
PATIENT PACKAGE INSERT
PATIENT INFORMATION
YUVIWEL® (YOU-vih-well)
(navepegritide)
For subcutaneous use onlyThis Patient Information has been approved by the U.S. Food and Drug Administration. Issued: 2/2026 What is YUVIWEL?
YUVIWEL is a prescription medicine used to increase linear growth in children 2 years of age and older with achondroplasia whose growth plates (epiphyses) are open.Before you give YUVIWEL to your child, tell the healthcare provider about all of your child´s medical conditions, including if your child: - has kidney problems.
- is pregnant or plans to become pregnant. It is not known if YUVIWEL will harm the unborn baby.
- is breastfeeding or plans to breastfeed. It is not known if YUVIWEL passes into the breast milk. Talk to your child's healthcare provider about the best way to feed your child´s baby, if your child receives YUVIWEL.
Know the medicines your child takes. Keep a list of them to show your child's healthcare provider and pharmacist when your child gets a new medicine.How should I give YUVIWEL?
Read the detailed Instructions for Use that comes with your child's YUVIWEL for information on how to store, prepare, and give YUVIWEL, as well as how to throw away (dispose of) YUVIWEL the right way.- Use YUVIWEL exactly as instructed by your child's healthcare provider.
- If YUVIWEL has been stored in the refrigerator, let the vial and Prefilled Diluent Syringe sit at room temperature for about 30 minutes before use.
- YUVIWEL is given as an injection under the skin (subcutaneous). YUVIWEL can be injected in either the stomach area (2 inches from the belly button) or thighs. If a caregiver gives the YUVIWEL injection, the buttocks or the back of the upper arms can be used as injection sites. Choose a different injection site each time. Avoid injecting where skin is red, swollen, or scarred.
- Your child´s healthcare provider will show you how to prepare and inject YUVIWEL the right way before you use it for the first time at home.
- Inject YUVIWEL 1 time each week on the scheduled dosing day. You can choose any time during the day to give your child the injection.
- If your child misses a dose of YUVIWEL, give the missed dose as soon as possible but not more than 2 days after the missed dosing day. To avoid missed doses, YUVIWEL can be given up to 2 days before or 2 days after the scheduled dosing day. If more than 2 days have passed, your child should skip the missed dose, and the next dose should be given on the regularly scheduled dosing day. In both cases, your child can go back to the regular 1 time each week dosing schedule. At least 5 days must pass between doses.
- In case you are not sure when to inject YUVIWEL, call your child´s healthcare provider or pharmacist. Do not give YUVIWEL more often than as directed by the healthcare provider.
- Your child's dose of YUVIWEL depends on body weight. The healthcare provider will tell you which strength of YUVIWEL to use and how much to give to your child.
- Your healthcare provider will monitor your child's growth and instruct you on when your child should stop using YUVIWEL if they determine that your child is no longer able to grow. Stop giving YUVIWEL to your child if instructed by your child's healthcare provider.
What are the possible side effects of YUVIWEL?
YUVIWEL may cause serious side effects, including:- risk of low blood pressure. If your child experiences a decrease in blood pressure or symptoms of low blood pressure (dizziness, feeling tired, and/or nausea) while being treated with YUVIWEL, call your child's healthcare provider.
Injection site reactions like redness, itching, skin discoloration, bleeding, swelling, bruising, pain and small fluid-filled blisters that form at the injection site.
These are not all the possible side effects of YUVIWEL. For more information, ask your child´s healthcare provider or pharmacist.
Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store YUVIWEL? - Store YUVIWEL in the refrigerator between 36°F to 46°F (2°C to 8°C) in the original carton to protect from light.
- Do not freeze YUVIWEL.
- YUVIWEL can also be stored at room temperature up to 86°F (30°C) for up to 6 months and can be returned to the refrigerator within the 6 months. Do not use YUVIWEL past the expiration date or 6 months after the date it was first removed from the refrigerator (whichever comes first).
- After mixing, YUVIWEL can be stored at room temperature up to 86°F (30°C) and must be used within 4 hours. Throw away unused medicine. Throw away medicine if not used within 4 hours after mixing.
General information about the safe and effective use of YUVIWEL.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use YUVIWEL for a condition for which it was not prescribed. Do not give YUVIWEL to other people, even if they have the same symptoms that your child has. It may harm them. You can ask a pharmacist or healthcare provider for information about YUVIWEL that is written for health professionals.What are the ingredients in YUVIWEL?
Active ingredients: navepegritide
Inactive ingredients: succinic acid, trehalose dihydrate, tromethamine and hydrochloric acid. The diluent contains Sterile Water for Injection, USP.Manufactured for:
Ascendis Pharma Growth Disorders A/S
Tuborg Boulevard 12,
DK-2900 Hellerup, Denmark
For more information, go to www.YUVIWEL.com, or call Ascendis Signature Access Program® (A·S·A·P) at 1-844-442-7236. Ascendis Pharma Endocrinology Inc., Princeton, New Jersey 08540, USA
© 2026 Ascendis Pharma. All rights reserved. YUVIWEL®, Ascendis®, TransCon®, the Ascendis Pharma logo and the company logo are trademarks owned by the Ascendis Pharma Group. -
INSTRUCTIONS FOR USE
Yuviwel® (YOU-vih-well)
(navepegritide) for injection, for subcutaneous useAscendis Pharma
For Use Under the Skin Only (subcutaneous)
Single-Dose Vial
Discard unused Portion1 time a week injection
This leaflet contains:
PRESCRIBING INFORMATION and
INSTRUCTIONS FOR USEAdditional information
If you need help, please ask your healthcare provider for advice. You may also call the Ascendis Pharma Customer Support toll-free number at 1-844-442-7236 (1-844-44ASCENDIS) for assistance or additional training, if needed.

Yuviwel.com
This Instructions for Use contains information on how to prepare and inject Yuviwel.
Parts Overview
Step 1
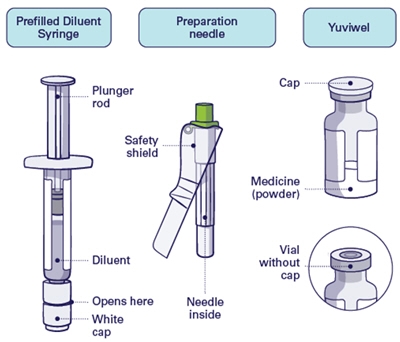
Important: Do not use preparation needle for injection
Step 2 and Step 3
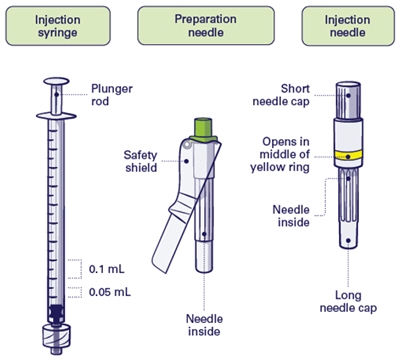
Important: Do not use preparation needle for injection
Supplies Not Included:
You will also need a sharps disposal container and alcohol wipes.
Get to know Yuviwel
Make sure you have received training from your healthcare provider before injecting.
Read and follow the Instructions for Use step-by-step to make sure you inject Yuviwel the right way.
Important: Prior to use, read the Instructions for Use carefully even if you have used a similar product. The steps may not be the same.
If you are blind or visually impaired, do not use Yuviwel without help from a person who is trained to use it.
How Yuviwel is supplied and injected
Yuviwel is injected under the skin (subcutaneous injection) 1 time a week.
Each Yuviwel carton includes 4 Kits, with 1 Prescribing Information and 1 Instructions for Use.
Yuviwel is available in the following strengths:
1.3 mg/vial 2.8 mg/vial 5.5 mg/vial Always check that you inject the dose strength prescribed by your healthcare provider.
If you are injecting yourself with Yuviwel, you should use the stomach area (2 inches from belly button) or thighs as injection sites.
If someone else gives you the Yuviwel injection, the buttocks or the back of the upper arms can be used as injection sites.
Only inject into injection sites instructed by your healthcare provider.
Important before starting Yuviwel
Check all parts and their packaging for signs of damage.
Do not use if any parts or packaging appear damaged. Use a new Kit.
Your healthcare provider may prescribe a dose that is more than 1 milliliter (mL) and requires the use of two Kits to deliver two injections for a complete dose.
If you have been prescribed a dose that is more than 1 mL:
- - Prepare and give your first injection, using one Kit (follow Steps 1 through 3.10)
- - Then, using a new Kit, immediately prepare and give a second injection by repeating steps 1 through 3.10.
Check the expiration date on the Kit. Do not use Yuviwel if expired. Use a new Kit.
Only use the vial, needles, and syringes that come in your Kit.
If you do not have an alcohol wipe, use soap and water to clean the injection site and the grey rubber at the top of the vial.
The vial, needles, and syringes in the Kit are for one time use only.
Do not reuse any of the parts. Do not recap needles.
Be careful when handling needles to reduce the risk of needlestick injury and infection.
Do not throw away opened or used needles in your household trash. Use a sharps disposal container.
After unpacking and to avoid contamination, do not touch the top of the vial, the tip of the Prefilled Diluent Syringe or the tip of the injection syringe.
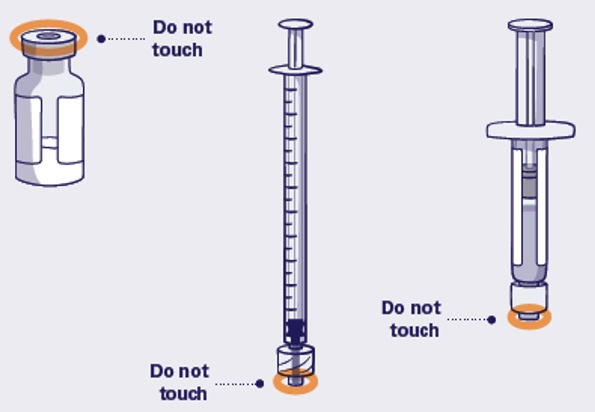
Do not change the injection angle after the needle has been inserted into the skin. Changing the angle can cause the needle to bend or break off under the skin. If a needle breaks in your skin, get medical help right away.
Throwing away Yuviwel parts
Throw away (dispose of) used needles, syringes and the vial in an FDA-cleared sharps disposal container right away after use.
- - Do not throw away (dispose of) used needles and syringes in the household trash.
- - Do not reuse needles. Always use a new needle for each injection.
If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- - Made of heavy-duty plastic,
- - Can be closed with a tight-fitting, puncture resistant lid, without sharps being able to come out,
- - Upright and stable during use,
- - Leak-resistant, and
- - Properly labeled to warn of hazardous waste inside the container
When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes.
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: www.fda.gov/safesharpsdisposal
Storing Yuviwel
Store Yuviwel refrigerated between 36°F to 46°F (2°C to 8°C) in the original carton to protect from light. Do not freeze. Yuviwel can also be stored at room temperature up to 86°F (30°C) for up to 6 months and can be returned to the refrigerator within the 6 months.
Do not use Yuviwel past the expiration date or 6 months after the date it was first removed from the refrigerator (whichever comes first).
Keep Yuviwel and all medicines out of the reach of children. Small parts in the Kit may cause a choking hazard for children and pets.
Yuviwel does not contain preservatives. After mixing, Yuviwel can be stored at room temperature up to 86°F (30°C) and must be used within 4 hours. Throw away unused medicine. Throw away medicine if not used within 4 hours after mixing.
Step 1
Prepare medicineGather supplies:
1 Kit, 2 alcohol wipes and a sharps disposal container.
Note: If the Kit has been stored in the refrigerator, let the vial and Prefilled Diluent Syringe sit at room temperature for about 30 minutes before mixing (reconstitution).

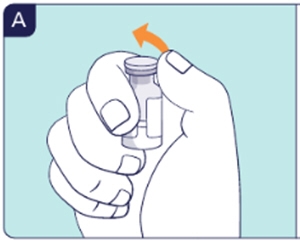
1.1 Wash hands with soap and water or use hand sanitizer. 1.2 Flip off the cap of the vial (See A).
You can use 1 or 2 hands to remove the cap.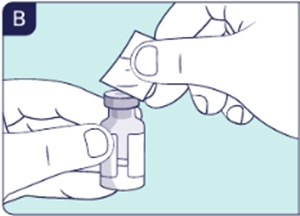
1.3 Wipe the grey rubber at the top of the vial with an alcohol wipe (See B). Do not touch the top after cleaning it. 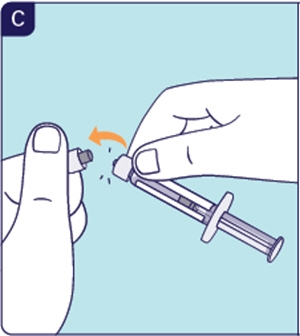
1.4 Break off the white cap of the Prefilled Diluent Syringe (See C). Do not touch the tip of the Prefilled Diluent Syringe after breaking off the cap.
Be careful not to push the plunger rod to avoid spilling diluent. If you spill diluent, use a new Kit.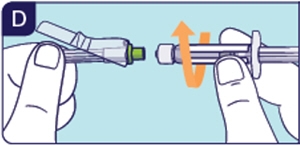
1.5 Unpack preparation needle and screw it on the Prefilled Diluent Syringe (See D). 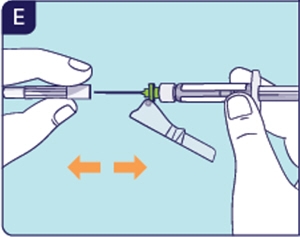
1.6 Pull back the safety shield and remove the needle cap (See E). Throw away (dispose of) the needle cap.
If the safety shield locks, use a new Kit.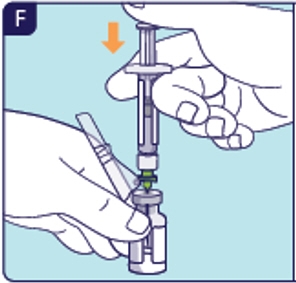
1.7 Insert preparation needle into the center of the circle on top of the vial (See F). 1.8 Push all the diluent into the vial (See F). 1.9 Pull preparation needle out of the vial. 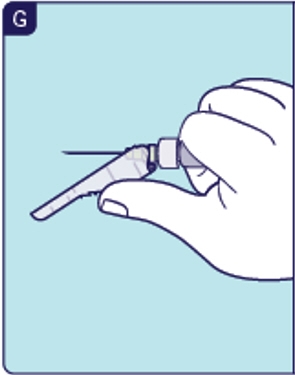
1.10 To avoid a needle stick injury, push the safety shield over preparation needle with 1 hand or press the shield against a hard surface directly after use until you hear it click and it locks (See G). 1.11 Place the Prefilled Diluent Syringe and preparation needle in the sharps disposal container. 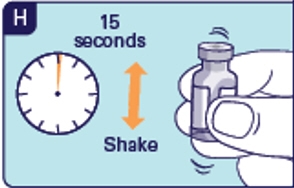
1.12 Hold the glass part of the vial and shake up and down for 15 seconds (See H). Do not swirl or roll as the medicine will not be fully mixed. 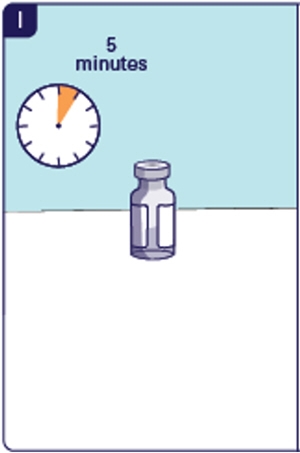
1.13 Put the vial down and leave it for 5 minutes to let the medicine settle (See I). 1.14 Check the medicine. The medicine should be clear and colorless. You may see air bubbles. This is normal. Do not use the medicine if you see particles.
Throw away medicine if not used within 4 hours after mixing.Step 2
Prepare injection
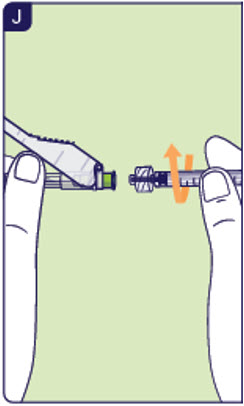
2.1 Unpack the injection syringe and a new preparation needle. 2.2 Screw a new preparation needle on the injection syringe (See J). 2.3 Pull back the safety shield and remove the needle cap.
Do not remove needle cap until ready to continue.
If the safety shield locks unexpectedly, use a new Kit.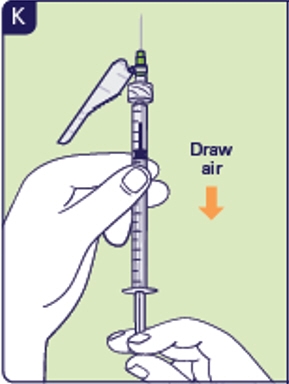
2.4 Pull the plunger rod out to draw air into the injection syringe equal to the prescribed Yuviwel dose in milliliters (mL) (See K). This is needed to withdraw the right dose. 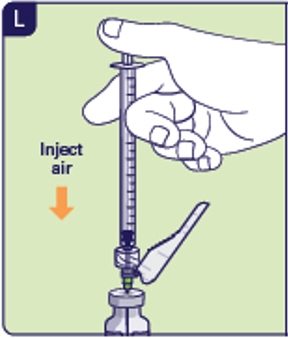
2.5 Insert preparation needle into the center of the circle on top of the vial and inject the air (See L). 2.6 Turn the vial and the injection syringe upside down. 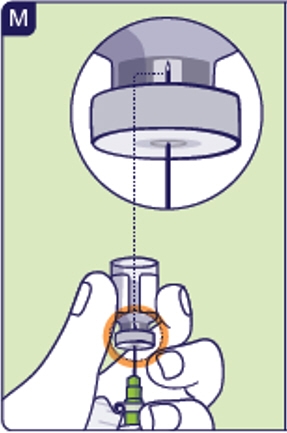
2.7 Position preparation needle within the liquid to allow the medicine to be withdrawn. If you insert the needle all the way in, you will only draw air and no medicine (See M). Turn the vial to the gap in the vial label to see the medicine better. 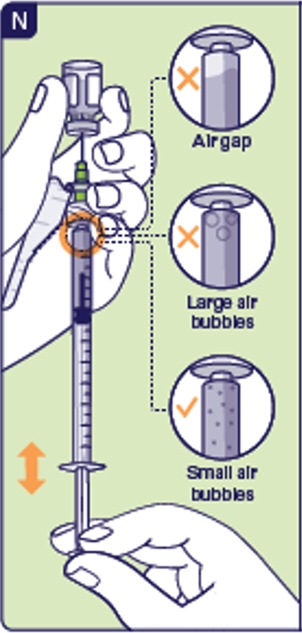
2.8 Withdraw the prescribed dose, by pulling and pushing the plunger to remove air gaps and large air bubbles (See N). Tapping the injection syringe may help air rise to the top. Before you continue:
Check for air gaps at the top of the syringe.
Check for large air bubbles.
If you see any air gaps or large air bubbles, pull and push the plunger to remove. Small air bubbles are okay (See N).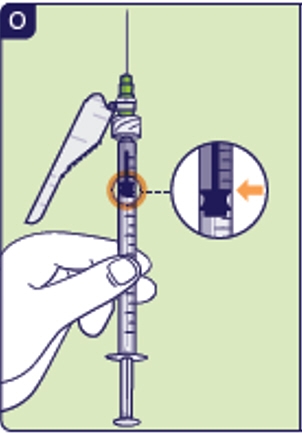
2.9 Check that you have the right dose (See O). The plunger has 2 rings. Your dose should be in line with the top ring of the black part of the plunger. If you did not withdraw the right dose, go back to step 2.7. 2.10 Pull preparation needle out of the vial. 
2.11 Lock the safety shield over the preparation needle (See G). 2.12 Rest the injection syringe carefully on the table while you unpack the injection needle (See P). Step 3
Inject and throw away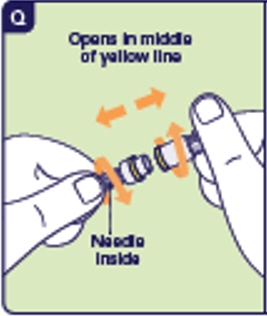
3.1 Gather the injection needle from the Kit. 3.2 Twist and pull off the short needle cap of the injection needle (See Q). Twisting both ways on the needle may help. You may have to twist hard. 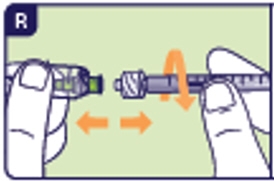
3.3 Unscrew the used preparation needle and place in the sharps disposal container (See R). 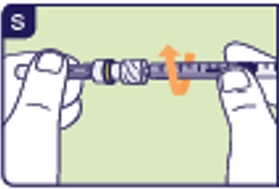
3.4 Screw the injection needle onto the injection syringe (See S). 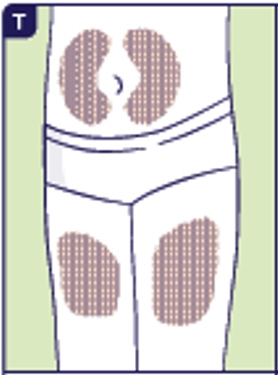
3.5 Choose a site for injection. You can inject in either the stomach area (2 inches from the belly button) or thighs (See T). Choose a different injection site each time. If someone else gives you the Yuviwel injection, the buttocks or the back of the upper arms can be used as injection sites (See T and U). 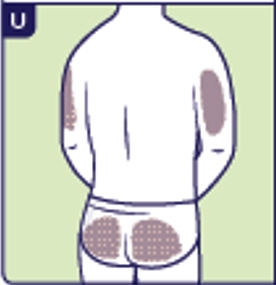
Avoid injecting where skin is red, swollen, or scarred. 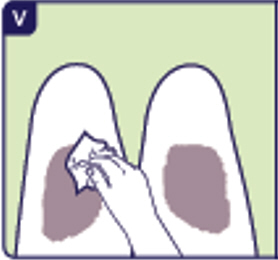
3.6 Clean the injection site with an alcohol wipe (See V). 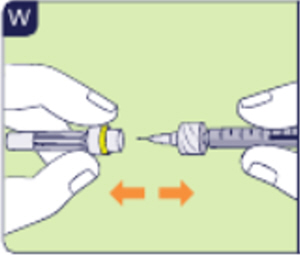
3.7 Pull off the long needle cap of the injection needle (See W). 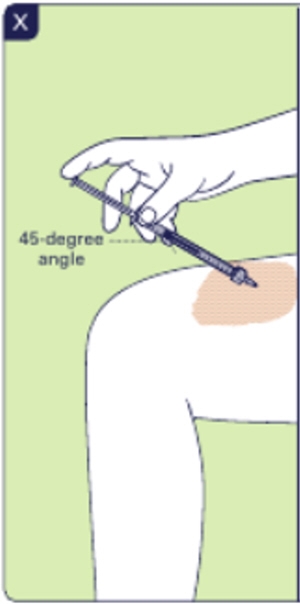
3.8 Insert the injection needle into the skin at a 45-degree angle. You can use 1 or 2 hands to inject. Inject the dose by slowly pushing down the plunger rod (See X). This may take some time, depending on the dose. 3.9 Pull out the injection needle from the skin. Do not recap the needle. 3.10 Place the injection needle and all other parts, including the vial and any remaining medicine, in the sharps disposal container. Important
If your Yuviwel dose is more than 1 mL each week:
If your prescribed dose is more than 1 mL, get a new Kit and repeat steps 1 through 3.10 to give a second injection for a complete dose. The second injection must be given right after the first injection, using a different injection site.
Remember to choose a different injection site for each Yuviwel injection.
Do not reuse any parts for the 2 injections.
This Instructions for Use has been approved by the U.S. Food and Drug Administration
Approved: 2/2026 -
PRINCIPAL DISPLAY PANEL - 1.3 mg Kit Carton
NDC: 73362-201-01
Rx Onlyyuviwel®
(navepegritide) for injection1.3 mg/vial
For Subcutaneous Use Only
Single-Dose Vial. Discard Unused Portion
Once-weekly injectionPrior to Use, read the Instructions for Use
Keep out of the reach of childrenThis carton contains 4 Kits and 1 Patient Information.
Each Kit contains:- 1 single-dose vial of Yuviwel 1.3 mg/vial
- 1 prefilled diluent syringe containing 0.8 mL Sterile Water
for Injection, USP - 2 single use preparation needles
- 1 single use injection needle
- 1 single use injection syringe
- 1 Instructions for Use
- 1 Prescribing Information

-
PRINCIPAL DISPLAY PANEL - 2.8 mg Kit Carton
NDC: 73362-202-01
Rx Onlyyuviwel®
(navepegritide) for injection2.8 mg/vial
For Subcutaneous Use Only
Single-Dose Vial. Discard Unused Portion
Once-weekly injectionPrior to Use, read the Instructions for Use
Keep out of the reach of childrenThis carton contains 4 Kits and 1 Patient Information.
Each Kit contains:- 1 single-dose vial of Yuviwel 2.8 mg/vial
- 1 prefilled diluent syringe containing 0.8 mL Sterile Water
for Injection, USP - 2 single use preparation needles
- 1 single use injection needle
- 1 single use injection syringe
- 1 Instructions for Use
- 1 Prescribing Information

-
PRINCIPAL DISPLAY PANEL - 5.5 mg Kit Carton
NDC: 73362-203-01
Rx Onlyyuviwel®
(navepegritide) for injection5.5 mg/vial
For Subcutaneous Use Only
Single-Dose Vial. Discard Unused Portion
Once-weekly injectionPrior to Use, read the Instructions for Use
Keep out of the reach of childrenThis carton contains 4 Kits and 1 Patient Information.
Each Kit contains:- 1 single-dose vial of Yuviwel 5.5 mg/vial
- 1 prefilled diluent syringe containing 1.1 mL Sterile Water
for Injection, USP - 2 single use preparation needles
- 1 single use injection needle
- 1 single use injection syringe
- 1 Instructions for Use
- 1 Prescribing Information

-
INGREDIENTS AND APPEARANCE
YUVIWEL
navepegritide kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 73362-201 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73362-201-01 4 in 1 CARTON 02/27/2026 1 NDC: 73362-201-02 1 in 1 BOX Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 VIAL, GLASS 1 Part 2 1 SYRINGE, GLASS 1 Part 1 of 2 YUVIWEL
navepegritide injection, powder, lyophilized, for solutionProduct Information Item Code (Source) NDC: 73362-205 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Navepegritide (UNII: Y3BH8M899D) (CNP-38 - UNII:U1KK9B22J6) Navepegritide 1.3 mg Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73362-205-01 1 in 1 VIAL, GLASS; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA219164 02/27/2026 Part 2 of 2 WATER
water solutionProduct Information Item Code (Source) NDC: 73362-801 Route of Administration SUBCUTANEOUS Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73362-801-01 1 in 1 SYRINGE, GLASS; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA219164 02/27/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA219164 02/27/2026 YUVIWEL
navepegritide kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 73362-202 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73362-202-01 4 in 1 CARTON 02/27/2026 1 NDC: 73362-202-02 1 in 1 BOX Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 VIAL, GLASS 1 Part 2 1 SYRINGE, GLASS 1 Part 1 of 2 YUVIWEL
navepegritide injection, powder, lyophilized, for solutionProduct Information Item Code (Source) NDC: 73362-206 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Navepegritide (UNII: Y3BH8M899D) (CNP-38 - UNII:U1KK9B22J6) Navepegritide 2.8 mg Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73362-206-01 1 in 1 VIAL, GLASS; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA219164 02/27/2026 Part 2 of 2 WATER
water solutionProduct Information Item Code (Source) NDC: 73362-801 Route of Administration SUBCUTANEOUS Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73362-801-01 1 in 1 SYRINGE, GLASS; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA219164 02/27/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA219164 02/27/2026 YUVIWEL
navepegritide kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 73362-203 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73362-203-01 4 in 1 CARTON 02/27/2026 1 NDC: 73362-203-02 1 in 1 BOX Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 VIAL, GLASS 1 Part 2 1 SYRINGE, GLASS 1 Part 1 of 2 YUVIWEL
navepegritide injection, powder, lyophilized, for solutionProduct Information Item Code (Source) NDC: 73362-207 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Navepegritide (UNII: Y3BH8M899D) (CNP-38 - UNII:U1KK9B22J6) Navepegritide 5.5 mg Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73362-207-01 1 in 1 VIAL, GLASS; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA219164 02/27/2026 Part 2 of 2 WATER
water solutionProduct Information Item Code (Source) NDC: 73362-801 Route of Administration SUBCUTANEOUS Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73362-801-01 1 in 1 SYRINGE, GLASS; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA219164 02/27/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA219164 02/27/2026 Labeler - Ascendis Pharma Endocrinology, Inc. (118185491)
Trademark Results [Yuviwel]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 YUVIWEL 98615005 not registered Live/Pending |
Ascendis Pharma Growth Disorders A/S 2024-06-24 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.