VESICARE LS- solifenacin succinate suspension
VESIcare by
Drug Labeling and Warnings
VESIcare by is a Prescription medication manufactured, distributed, or labeled by Astellas Pharma US, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use VESICARE LS safely and effectively. See full prescribing information for VESICARE LS.
VESICARE LS® (solifenacin succinate) oral suspension
Initial U.S. Approval: 2004INDICATIONS AND USAGE
VESIcare LS is a muscarinic antagonist indicated for the treatment of neurogenic detrusor overactivity in pediatric patients aged 2 years and older. (1)
DOSAGE AND ADMINISTRATION
- The recommended once daily dose of VESIcare LS is based on patient weight, refer to Table 1. Dosing should be initiated at the recommended starting dose. Dosage may be titrated to the lowest effective dose but should not exceed the maximum recommended dose. (2.1)
- Instruct patients or their caregivers that patients should take VESIcare LS orally followed by liquid (e.g., water or milk).
- Do not exceed the recommended starting dose of VESIcare LS in patients with:
DOSAGE FORMS AND STRENGTHS
Oral suspension: 5 mg/5 mL (1 mg/mL) of solifenacin succinate. (3)CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Angioedema and Anaphylactic Reactions: Promptly discontinue VESIcare LS and provide appropriate therapy. (5.1)
- Urinary Retention: VESIcare LS is not recommended for use in patients with clinically significant bladder outlet obstruction in the absence of clean intermittent catherization. (5.2)
- Gastrointestinal Disorders: VESIcare LS is not recommended for use in patients with decreased gastrointestinal motility. (5.3)
- Central Nervous System Effects: Somnolence has been reported with solifenacin succinate. Advise patients not to drive or operate heavy machinery until they know how VESIcare LS affects them. (5.4)
- Controlled Narrow-Angle Glaucoma: Use VESIcare LS with caution in patients being treated for narrow-angle glaucoma. (5.5)
-
QT Prolongation in Patients at High Risk of QT Prolongation:
VESIcare LS is not recommended for use in patients at high risk of QT prolongation, including patients with a known history of QT prolongation and patients taking medications known to prolong the QT interval. (5.6)
ADVERSE REACTIONS
The most common adverse reactions (> 2%) were constipation, dry mouth and urinary tract infection. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Astellas Pharma US, Inc. at 1-800-727-7003 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
CYP3A4 Inhibitors: Do not exceed the recommended starting dose of VESIcare LS with concomitant use of strong CYP3A4 inhibitors. (7.1)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 10/2022
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Information
2.2 Dosing Recommendations in Patients with Renal Impairment
2.3 Dosing Recommendations in Patients with Hepatic Impairment
2.4 Dosing Recommendations in Patients Taking CYP3A4 Inhibitors
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Angioedema and Anaphylactic Reactions
5.2 Urinary Retention
5.3 Gastrointestinal Disorders
5.4 Central Nervous System Effects
5.5 Controlled Narrow-Angle Glaucoma
5.6 QT Prolongation in Patients at High Risk of QT Prolongation
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Strong CYP3A4 Inhibitors
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Information
Dosing Information in Pediatric Patients Aged 2 Years and Older
The recommended starting and maximum VESIcare LS oral suspension doses are shown in mL in Table 1. VESIcare LS oral suspension has a concentration of 1 mg/1 mL. The recommended doses are weight-based and are administered once daily. After administration of the recommended starting dose, the dose may be increased to the lowest effective dose but should not exceed the maximum recommended dose.
Table 1: Once Daily Recommended Dosage According to Patient Body Weight Weight range
Starting dose
Maximum dose
9 kg to 15 kg
2 mL
4 mL
greater than 15 kg to 30 kg
3 mL
5 mL
greater than 30 kg to 45 kg
3 mL
6 mL
greater than 45 kg to 60 kg
4 mL
8 mL
greater than 60 kg
5 mL
10 mL
Evaluate patients periodically for potential dose adjustment.
VESIcare LS oral suspension should be taken once daily. Instruct patients or their caregivers that patients should take VESIcare LS orally followed by liquid (e.g., water or milk). Instruct patients to take any missed doses as soon as they remember, unless more than 12 hours have passed since the missed dose. If more than 12 hours have passed, the missed dose can be skipped, and the next dose should be taken at the usual time.
2.2 Dosing Recommendations in Patients with Renal Impairment
Do not exceed the recommended VESIcare LS oral suspension starting dose in patients with severe renal impairment (CLcr < 30 mL/min/1.73 m2) [see Use in Specific Populations (8.6)].
2.3 Dosing Recommendations in Patients with Hepatic Impairment
Do not exceed the recommended VESIcare LS oral suspension starting dose in patients with moderate hepatic impairment (Child-Pugh B). Do not use VESIcare LS in patients with severe hepatic impairment (Child-Pugh C) [see Use in Specific Populations (8.7)].
2.4 Dosing Recommendations in Patients Taking CYP3A4 Inhibitors
Do not exceed the recommended VESIcare LS oral suspension starting dose when VESIcare LS is administered with strong CYP3A4 inhibitors such as ketoconazole [see Drug Interactions (7.1)].
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
VESIcare LS is contraindicated in patients:
- With gastric retention [see Warnings and Precautions (5.3)],
- With uncontrolled narrow-angle glaucoma [see Warnings and Precautions (5.5)], and
- Who have demonstrated hypersensitivity to solifenacin succinate or the inactive ingredients in VESIcare LS oral suspension. Reported adverse reactions have included anaphylaxis and angioedema [see Adverse Reactions (6.2)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Angioedema and Anaphylactic Reactions
Angioedema of the face, lips, tongue, and/or larynx have been reported with solifenacin succinate. In some cases, angioedema occurred after the first dose, however, cases have been reported to occur hours after the first dose or after multiple doses. Anaphylactic reactions have also been reported in patients treated with solifenacin succinate. Angioedema associated with upper airway swelling and anaphylactic reactions may be life-threatening.
VESIcare LS oral suspension is contraindicated in patients with a known or suspected hypersensitivity to solifenacin succinate [see Contraindications (4)]. If involvement of the tongue, hypopharynx, or larynx occurs, promptly discontinue VESIcare LS oral suspension and provide appropriate therapy and/or measures necessary to ensure a patent airway.
5.2 Urinary Retention
The use of VESIcare LS oral suspension, like other antimuscarinic drugs, in patients with clinically significant bladder outlet obstruction may result in urinary retention. The use of VESIcare LS oral suspension is not recommended in patients with clinically significant bladder outflow obstruction in the absence of clean intermittent catheterization because of the risk of urinary retention.
5.3 Gastrointestinal Disorders
The use of VESIcare LS oral suspension, like other antimuscarinic drugs, in patients with conditions associated with decreased gastrointestinal motility may result in further decreased gastrointestinal motility. VESIcare LS oral suspension is contraindicated in patients with gastric retention [see Contraindications (4)]. The use of VESIcare LS oral suspension is not recommended in patients with conditions associated with decreased gastrointestinal motility.
5.4 Central Nervous System Effects
Solifenacin succinate is associated with antimuscarinic central nervous system (CNS) adverse reactions [see Adverse Reactions (6.2)]. A variety of CNS antimuscarinic adverse reactions have been reported, including headache, confusion, hallucinations, and somnolence. Monitor patients for signs of antimuscarinic CNS adverse reactions, particularly after beginning treatment or increasing the dose. Advise patients not to drive or operate heavy machinery until they know how VESIcare LS affects them. If a patient experiences antimuscarinic CNS adverse reactions, consider dose reduction or drug discontinuation.
5.5 Controlled Narrow-Angle Glaucoma
VESIcare LS should be used with caution in patients being treated for narrow-angle glaucoma [see Contraindications (4)].
5.6 QT Prolongation in Patients at High Risk of QT Prolongation
In a study of the effect of solifenacin succinate on the QT interval conducted in 76 healthy adult women [see Clinical Pharmacology (12.2)], solifenacin succinate 30 mg (three times the largest maximum recommended dose in pediatric patients) was associated with a mean increase in the Fridericia-corrected QT interval of 8 msec (90% CI, 4, 13). The QT prolonging effect appeared less with solifenacin succinate 10 mg than with solifenacin succinate 30 mg, and the effect of solifenacin succinate 30 mg did not appear as large as that of the positive control moxifloxacin at its therapeutic dose.
The use of VESIcare LS is not recommended in patients at high risk of QT prolongation, including patients with a known history of QT prolongation and patients who are taking medications known to prolong the QT interval.
-
6 ADVERSE REACTIONS
- Angioedema and Anaphylactic Reactions [see Warnings and Precautions (5.1)]
- Urinary Retention [see Warnings and Precautions (5.2)]
- Gastrointestinal Disorders [see Warnings and Precautions (5.3)]
- Central Nervous System Effects [see Warnings and Precautions (5.4)]
- QT Prolongation in Patients at High Risk of QT Prolongation [see Warnings and Precautions (5.6)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of VESIcare LS oral suspension was evaluated in two open-label trials (Studies 1 and 2) [see Clinical Studies (14)]. The two studies included 95 pediatric patients aged 2 to 17 years with neurogenic detrusor overactivity (NDO) who were 53% female, 58% White, 34% Asian and 2% Black. Treatment was initiated at the weight-based starting recommended dose and was titrated up or down in 2.5 mg increments over 12 weeks to the lowest effective dose (not to exceed the maximum recommended dose). Subsequent to the dose titration period, patients continued their optimized dose for a 40-week maintenance period (mean exposure duration 301 days, range 1 to 413 days).
The most commonly reported adverse reactions were constipation, dry mouth, urinary tract infection, abdominal pain, urinalysis bacterial test positive, and somnolence. The incidence of adverse reactions was similar between patients taking the starting recommended dose and patients taking the maximum recommended dose with the exception of constipation, which was reported, in 8.5% of patients taking the maximum recommended dose compared to 0% of patients taking the starting recommended dose.
Table 2 lists the adverse reactions reported in Studies 1 and 2 at an incidence equal to or greater than 1%.
Table 2: Adverse Reactions Reported in ≥ 1% of Neurogenic Detrusor Overactivity (NDO) Patients Aged 2 to 17 Years Taking VESIcare LS Oral Suspension in Studies 1 and 2 Adverse Reaction
Percentage (%) of Patients Reporting
Adverse Reactions
N=95
Constipation
7.4
Dry mouth
3.2
Urinary tract infection
2.1
Abdominal pain
1.1
Urinalysis bacterial test positive
1.1
Somnolence
1.1
Adverse reactions reported in ≥ 1% of solifenacin succinate-treated adult patients and at an incidence greater than in placebo-treated adult patients in clinical adult trials were:
Gastrointestinal disorders: dry mouth, constipation, nausea, dyspepsia, upper abdominal pain, vomiting
Infections and infestations: urinary tract infection, influenza, pharyngitis
Nervous system disorders: dizziness
Eye disorders: blurred vision, dry eyes
Renal and urinary disorders: urinary retention
General disorders and administration site conditions: lower limb edema, fatigue
Psychiatric disorders: depression
Respiratory, thoracic and mediastinal disorders: cough
Vascular disorders: hypertension
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of solifenacin succinate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
General disorders and administration site conditions: peripheral edema, hypersensitivity reactions (including angioedema with airway obstruction, rash, pruritus, urticaria, anaphylactic reaction);
Nervous system disorders: dizziness, headache, confusion, hallucinations, delirium, somnolence;
Cardiac disorders: QT prolongation, Torsade de Pointes, atrial fibrillation, tachycardia, palpitations;
Hepatobiliary disorders: liver disorders mostly characterized by abnormal liver function tests, AST (aspartate aminotransferase), ALT (alanine aminotransferase), GGT (gamma-glutamyl transferase);
Renal and urinary disorders: renal impairment, urinary retention;
Metabolism and nutrition disorders: decreased appetite, hyperkalemia;
Skin and subcutaneous tissue disorders: exfoliative dermatitis, erythema multiforme, dry skin;
Eye disorders: glaucoma;
Gastrointestinal disorders: gastroesophageal reflux disease, ileus, vomiting, abdominal pain, dysgeusia, sialadenitis;
Respiratory, thoracic and mediastinal disorders: dysphonia, nasal dryness;
Musculoskeletal and connective tissue disorders: muscular weakness.
-
7 DRUG INTERACTIONS
7.1 Strong CYP3A4 Inhibitors
Solifenacin is a substrate of CYP3A4. Concomitant use of ketoconazole, a strong CYP3A4 inhibitor, significantly increased the exposure of solifenacin [see Clinical Pharmacology (12.3)]. The dosage of VESIcare LS greater than the starting dose is not recommended when concomitantly used with strong CYP3A4 inhibitors [see Dosage and Administration (2.4)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no studies with the use of solifenacin succinate in pregnant women or adolescents to inform a drug-associated risk of major birth defects, miscarriages, or adverse maternal or fetal outcomes. No adverse developmental outcomes were observed in animal reproduction studies with oral administration of solifenacin succinate to pregnant mice during the period of organogenesis at a dose resulting in 1.2 times the systemic exposure at the maximum recommended human dose (MRHD) of 10 mg/day. However, administration of doses 3.6 times and greater than the MRHD during organogenesis produced maternal toxicity in the pregnant mice and resulted in developmental toxicity and reduced fetal body weights in offspring [see Data].
In the U.S. general population, the estimated background risk of major birth defects or miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
Oral administration of 14C-solifenacin succinate to pregnant mice resulted in the recovery of radiolabel in the fetus indicating that solifenacin-related product can cross the placental barrier. In pregnant mice, administration of solifenacin succinate at a dose of 250 mg/kg/day (7.9 times the systemic exposure at the MRHD of 10 mg), resulted in an increased incidence of cleft palate and increased maternal lethality. Administration of solifenacin succinate to pregnant mice during organogenesis at greater than or equal to 3.6 times (100 mg/kg/day and greater) the systemic exposure at the MRHD, resulted in reduced fetal body weights and reduced maternal body weight gain. No embryo-fetal toxicity or teratogenicity was observed in fetuses from pregnant mice treated with solifenacin succinate at a dose of 30 mg/kg/day (1.2 times the systemic exposure at the MRHD). Administration of solifenacin succinate to pregnant rats and rabbits at a dose of 50 mg/kg/day (< 1 times and 1.8 times the systemic exposure at the MRHD, respectively), resulted in no findings of embryo-fetal toxicity. Oral pre- and post-natal administration of solifenacin succinate at 100 mg/kg/day (3.6 times the systemic exposure at the MRHD) during the period of organogenesis through weaning, resulted in reduced peripartum and postnatal survival, reduced body weight gain by the pups, and delayed physical development (eye opening and vaginal patency). An increase in the percentage of male offspring was also observed in litters from offspring (F2 generation) exposed to maternal doses of 250 mg/kg/day. There were no effects on natural delivery in mice treated with 1.2 times (30 mg/kg/day) the expected systemic exposure at the MRHD.
8.2 Lactation
Risk Summary
There is no information on the presence of solifenacin in human milk, the effects on the breastfed child, or the effects on milk production. Solifenacin is present in mouse milk [see Data]. When a drug is present in animal milk, it is likely that the drug will be present in human milk. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for VESIcare LS and any potential adverse effects on the breastfed child from VESIcare LS or from the underlying maternal condition.
Data
Animal Data
Oral administration of 14C-solifenacin succinate to lactating mice resulted in the recovery of radioactivity in maternal milk. Lactating female mice orally administered solifenacin succinate at a maternally toxic dose of 100 mg/kg/day (3.6 times the systemic exposure at the MRHD) had increased postpartum pup mortality, pups with reduced body weights, or delays in the onset of reflex and physical development. Pups from lactating dams orally administered solifenacin succinate at a dose of 30 mg/kg/day (1.2 times the systemic exposure at the MRHD) had no discernible adverse findings. The concentrations of solifenacin in animal milk does not necessarily predict the concentration of drug in human milk.
8.4 Pediatric Use
The safety and effectiveness of VESIcare LS have been established in pediatric patients aged 2 years and older for the treatment of neurogenic detrusor overactivity (NDO) and the information on this use is discussed throughout the labeling. The safety and effectiveness of VESIcare LS have not been established in pediatric patients less than 2 years of age.
8.6 Renal Impairment
Solifenacin plasma concentrations are greater in adults with severe renal impairment compared to adults with normal renal function [see Clinical Pharmacology (12.3)]. Because increased solifenacin plasma concentrations increase the risk of antimuscarinic adverse reactions, the maximum recommended dose of VESIcare LS oral suspension in pediatric patients 2 years of age and older with severe renal impairment (CLcr < 30 mL/min/1.73 m2) is the starting dose [see Dosage and Administration (2.2)]. The recommended dose in patients with mild or moderate renal impairment is the same as in patients with normal renal function.
8.7 Hepatic Impairment
Solifenacin plasma concentrations are greater in adults with moderate hepatic impairment compared to adults with normal hepatic function [see Clinical Pharmacology (12.3)]. Because increased solifenacin plasma concentrations increase the risk of antimuscarinic adverse reactions, the maximum recommended dose of VESIcare LS oral suspension in pediatric patients 2 years of age and older with moderate hepatic impairment (Child-Pugh B) is the starting dose [see Dosage and Administration (2.3)] and VESIcare LS oral suspension is not recommended for use in patients with severe hepatic impairment (Child-Pugh C).
-
10 OVERDOSAGE
Overdosage with VESIcare LS oral suspension can potentially result in severe antimuscarinic effects and should be treated accordingly. The highest dose ingested in an accidental overdose of solifenacin succinate was 280 mg (28 times the maximum dosage) in a 5-hour period in adults. This case was associated with mental status changes. Some cases reported a decrease in the level of consciousness.
Intolerable antimuscarinic adverse reactions (fixed and dilated pupils, blurred vision, failure of heel-to-toe exam, tremors, and dry skin) occurred on day 3 in normal adult volunteers taking 50 mg daily (5 times the maximum recommended dosage) and resolved within 7 days following discontinuation of drug.
The highest overdose reported in a pediatric patient was the accidental ingestion of 95 mg of solifenacin succinate (9.5 times the maximum recommended dosage). The child experienced severe antimuscarinic adverse reactions and was treated with gastric lavage and supportive measures until all antimuscarinic signs and symptoms reversed.
In the event of overdose with VESIcare LS oral suspension, treat with gastric lavage and appropriate supportive measures. ECG monitoring is also recommended.
-
11 DESCRIPTION
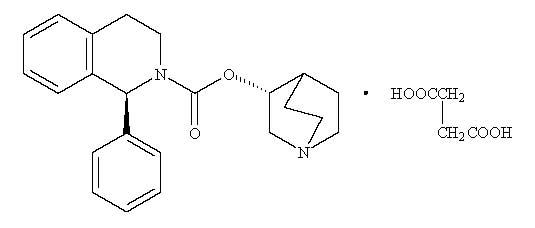
VESIcare LS (solifenacin succinate) oral suspension is a muscarinic receptor antagonist. Chemically, solifenacin succinate is a butanedioic acid compound with (1S)-(3R)-1-azabicyclo[2.2.2]oct-3-yl 3,4-dihydro-1-phenyl-2(1H)-iso-quinolinecarboxylate (1:1) having an empirical formula of C23H26N2O2C4H6O4, and a molecular weight of 480.55. The structural formula of solifenacin succinate is:
Solifenacin succinate is a white to pale-yellowish-white crystal or crystalline powder. It is freely soluble at room temperature in water, glacial acetic acid, dimethyl sulfoxide, and methanol.
VESIcare LS contains 1 mg/mL of solifenacin succinate as an oral suspension (1 mg solifenacin succinate is equivalent to 0.75 mg solifenacin). The inactive ingredients are polacrilin potassium, methylparaben, propylparaben, propylene glycol, simethicone emulsion 30%, carbomer homopolymer Type B, xylitol, acesulfame potassium, natural orange flavor, sodium hydroxide, and purified water. VESIcare LS is a white to off-white color aqueous homogenous oral suspension with an orange flavor.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Solifenacin is a competitive muscarinic receptor antagonist. Muscarinic receptors play an important role in several major cholinergically mediated functions, including contractions of urinary bladder smooth muscle.
12.2 Pharmacodynamics
Cardiac Electrophysiology
The effect of 10 mg and 30 mg solifenacin succinate (three times the maximum recommended dose) on the QT interval was evaluated in adults at the time of peak plasma concentration of solifenacin in a multi-dose, randomized, double-blind, placebo and positive-controlled (moxifloxacin 400 mg) trial [see Warnings and Precautions (5.6)]. After receiving placebo and moxifloxacin sequentially, adult subjects were randomized to one of two treatment groups. One group (n=51) completed 3 additional sequential periods of dosing with solifenacin succinate 10, 20, and 30 mg while the second group (n=25) in parallel completed a sequence of placebo and moxifloxacin. Study subjects were female volunteers aged 19 to 79 years. The 30 mg dose of solifenacin succinate (three times the highest recommended dose) was chosen for use in this study because this dose results in a solifenacin exposure that covers those observed upon coadministration of 10 mg solifenacin succinate with strong CYP3A4 inhibitors (e.g., ketoconazole, 400 mg). Due to the sequential dose escalating nature of the study, baseline ECG measurements were separated from the final QT assessment (of the 30 mg dose level) by 33 days.
The median difference from baseline in heart rate associated with the 10 and 30 mg doses of solifenacin succinate compared to placebo was -2 and 0 beats/minute, respectively. Because a significant period effect on QTc was observed, the QTc effects were analyzed utilizing the parallel placebo control arm rather than the pre-specified intra-patient analysis. Representative results are shown in Table 3.
Table 3: QTc changes in msec (90% CI) from baseline at Tmax (relative to placebo) in adults* Drug/Dose Fridericia Method
(using mean difference)- * Results displayed are those derived from the parallel design portion of the study and represent the comparison of Group 1 to
time-matched placebo effects in Group 2.Solifenacin succinate 10 mg
2 (-3, 6)
Solifenacin succinate 30 mg
8 (4, 13)
Moxifloxacin was included as a positive control in this study and, given the length of the study, its effect on the QT interval was evaluated in 3 different sessions. The placebo-subtracted mean changes (90% CI) in QTcF for moxifloxacin in the three sessions were 11 (7, 14), 12 (8, 17), and 16 (12, 21), respectively.
The QT interval prolonging effect of the highest solifenacin succinate dose (three times the maximum therapeutic dose) studied was not as large as that of the positive control moxifloxacin at its recommended dose. However, the confidence intervals overlapped, and this study was not designed to draw direct statistical conclusions between the drugs or the dose levels.
12.3 Pharmacokinetics
Absorption
After oral administration of VESIcare LS in pediatric patients with neurogenic detrusor overactivity (NDO) aged 2 to 17 years, peak plasma concentrations (Cmax) of solifenacin were reached within 2 to 6 hours after administration (tmax) and, at steady-state, the dose-normalized Cmax ranged from 2.5 - 29 ng/mL/mg. The absolute bioavailability of solifenacin in adults is approximately 90%, with plasma concentrations of solifenacin proportional to the dose administered.
- Effect of Food
- Food intake did not significantly affect the Cmax and AUC of solifenacin following oral administration of VESIcare LS.
Distribution
Solifenacin is approximately 98% (in vivo) bound to human plasma proteins, principally to α1-acid glycoprotein. Solifenacin is highly distributed to non-CNS tissues, having a median steady-state volume of distribution of 211 L in pediatric neurogenic detrusor overactivity (NDO) patients from 2 to 17 years old.
Elimination
The median elimination half-life of solifenacin is approximately 26 hours in pediatric neurogenic detrusor overactivity (NDO) patients 2 years of age and older.
- Metabolism
-
Solifenacin is extensively metabolized in the liver. The primary pathway for elimination is by way of CYP3A4; however, alternate metabolic pathways exist. The primary metabolic routes of solifenacin are through N-oxidation of the quinuclidin ring and 4R-hydroxylation of the tetrahydroisoquinoline ring. One pharmacologically active metabolite (4R-hydroxy solifenacin), occurring at low concentrations and unlikely to contribute significantly to clinical activity, and three pharmacologically inactive metabolites (N-glucuronide and the N-oxide and 4R-hydroxy-N-oxide of solifenacin) have been found in human plasma after oral dosing.
- Excretion
- Following the administration of 10 mg of 14C-solifenacin succinate to healthy adult volunteers, 69% of the radioactivity was recovered in the urine and 23% in the feces over 26 days. Less than 15% (as mean value) of the dose was recovered in the urine as intact solifenacin. The major metabolites identified in urine were N-oxide of solifenacin, 4R-hydroxy solifenacin, and 4R-hydroxy-N-oxide of solifenacin and, in feces, 4R-hydroxy solifenacin.
Specific Populations
- Patients with Renal Impairment
- In studies with solifenacin succinate 10 mg in adults, there was a 2.1-fold increase in AUC and a 1.6-fold increase in t1/2 of solifenacin in patients with severe renal impairment compared to subjects with normal renal function [see Use in Specific Populations (8.6)].
- Patients with Hepatic Impairment
- In studies with solifenacin succinate 10 mg in adults, there was a 2-fold increase in the t1/2 and a 35% increase in AUC of solifenacin in patients with moderate hepatic impairment compared to subjects with normal hepatic function [see Use in Specific Populations (8.7)]. Solifenacin succinate has not been studied in patients with severe hepatic impairment.
Drug Interaction Studies
Interaction studies have only been performed in adults. In pediatric patients 2 years of age and older, metabolism via CYP3A4 is the major route of elimination and the metabolic capacity of the liver is similar.
- Strong CYP3A4 Inhibitors
- In a crossover study in adults, following blockade of CYP3A4 by coadministration of the strong CYP3A4 inhibitor, ketoconazole 400 mg once daily for 21 days, the mean Cmax and AUC of solifenacin increased by 1.5 and 2.7-fold, respectively [see Dosage and Administration (2.4) and Drug Interactions (7.1)].
- CYP3A4 Inducers
- Because solifenacin is a substrate of CYP3A4, inducers of CYP3A4 may decrease the concentration of solifenacin.
- Warfarin
- In a crossover study in adults, subjects received a single oral dose of warfarin 25 mg on the 10th day of dosing with either solifenacin succinate 10 mg or matching placebo once daily for 16 days. For R-warfarin, when it was coadministered with solifenacin succinate, the mean Cmax increased by 3% and AUC decreased by 2%. For S-warfarin, when it was coadministered with solifenacin succinate, the mean Cmax and AUC increased by 5% and 1%, respectively.
- Oral Contraceptives
- In a crossover study in adults, subjects received 2 cycles of 21 days of oral contraceptives containing 30 ug ethinyl estradiol and 150 ug levonorgestrel. During the second cycle, subjects received additional solifenacin succinate 10 mg or matching placebo once daily for 10 days starting from the 12th day of receipt of oral contraceptives. For ethinyl estradiol, when it was administered with solifenacin succinate, the mean Cmax and AUC increased by 2% and 3%, respectively. For levonorgestrel, when it was administered with solifenacin succinate, the mean Cmax and AUC decreased by 1%.
- Digoxin
- In a crossover study in adults, subjects received digoxin (loading dose of 0.25 mg on day 1, followed by 0.125 mg from days 2 to 8) for 8 days. Consecutively, they received solifenacin succinate 10 mg or matching placebo with digoxin 0.125 mg for an additional 10 days. When digoxin was coadministered with solifenacin succinate, the mean Cmax and AUC increased by 13% and 4%, respectively.
-
Drugs Metabolized by Cytochrome P450 Enzymes
In vitro studies demonstrated that, at therapeutic concentrations, solifenacin does not inhibit CYP1A1/2, 2C9, 2C19, 2D6, or 3A4 derived from human liver microsomes.
- * Results displayed are those derived from the parallel design portion of the study and represent the comparison of Group 1 to
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No increase in tumors was found following the administration of solifenacin succinate to male and female mice for 104 weeks at doses up to 200 mg/kg/day (5 and 9 times, respectively, of the exposure at the maximum recommended human dose [MRHD] of 10 mg), and male and female rats for 104 weeks at doses up to 20 and 15 mg/kg/day, respectively (< 1 times the exposure at the MRHD).
Solifenacin succinate was not mutagenic in the in vitro Salmonella typhimurium or Escherichia coli microbial mutagenicity test or chromosomal aberration test in human peripheral blood lymphocytes with or without metabolic activation or in the in vivo micronucleus test in rats.
Solifenacin succinate had no effect on reproductive function, fertility, or early embryonic development of the fetus in male and female mice treated with 250 mg/kg/day (13 times the exposure at the MRHD) of solifenacin succinate, and in male rats treated with 50 mg/kg/day (< 1 times the exposure at the MRHD) and female rats treated with 100 mg/kg/day (1.7 times the exposure at the MRHD) of solifenacin succinate.
13.2 Animal Toxicology and/or Pharmacology
Juvenile Animal Toxicology Data
Dose-related increased mortality without preceding clinical signs occurred in juvenile mice treated before weaning for a duration of 12 weeks, from day 10 after birth, with doses that achieved a pharmacological effect. Animals dosed from postnatal day 10 onwards had higher mortality compared to the mortality in adult mice. No increased frequency in mortality was observed in juvenile mice that were treated after weaning for a duration of 4 weeks, from day 21 after birth onwards. Plasma exposure at postnatal day 10 was higher than in adult mice; the systemic exposure at postnatal day 21 was comparable to the systemic exposure in adult mice.
-
14 CLINICAL STUDIES
The efficacy of VESIcare LS oral suspension was evaluated in two 52-week, open-label, baseline-controlled, sequential dose titration studies in a total of 95 pediatric patients 2 years of age and older with neurogenic detrusor overactivity (NDO). Study 1 (NCT01981954) included patients 2 to less than 5 years of age and Study 2 (NCT01565694) included patients 5 to 17 years of age. Entry criteria required that patients had a diagnosis of neurogenic detrusor overactivity (NDO) confirmed by urodynamics demonstrating the presence of involuntary detrusor contractions with detrusor pressure increase greater than 15 cm H2O and that patients or their caregivers practiced clean intermittent catheterization (CIC). VESIcare LS oral suspension was administered once daily. All patients initially received a weight-based starting dose, followed by dose titration to their lowest effective dose, up to a maximum dose. The duration of the dose titration period was up to 12 weeks and this period was followed by a dose maintenance period of at least 40 weeks.
In Study 1, a total of 19 patients aged 2 to less than 5 years of age received VESIcare LS oral suspension. Of these, 17 patients completed treatment through week 24 and had adequate urodynamic measurements for evaluation of efficacy. The optimized maintenance dose within this study population included 63% of patients at the maximum dose, 26% of patients at an intermediate dose, and 5% of patients at the starting dose. The study population included 7 males (41%) and 10 females (59%).
In Study 2, a total of 76 patients aged 5 to 17 years of age received VESIcare LS oral suspension. Of these, 49 patients (24 patients aged 5 to < 12 years and 25 patients aged 12 to 17 years) completed treatment through week 24 and had adequate urodynamic measurements for evaluation of efficacy. The optimized maintenance dose within this study population included 58% of patients at the maximum dose, 15% of patients at an intermediate dose, and 7% of patients at the starting dose. This study population included 24 males (49%) and 25 females (51%).
The primary efficacy endpoint was change from baseline in the patients’ maximum cystometric (bladder) capacity (MCC) after 24 weeks of treatment with VESIcare LS oral suspension. As shown in Table 4, an improvement in MCC was observed in patients aged 2 to less than 5 years of age and in patients aged 5 to 17 years of age. The magnitude of the observed changes from baseline in the primary and secondary efficacy endpoints were comparable between patients 5 to less than 12 years of age and patients 12 to 17 years of age.
The results for the primary efficacy endpoint in the 2 clinical studies of VESIcare LS oral suspension in pediatric patients with neurogenic detrusor overactivity (NDO) are shown in Table 4. Treatment effects were maintained over 52 weeks.
Table 4: Change from Baseline in Maximum Cystometric Capacity (MCC) at 24 Weeks in Pediatric Patients with Neurogenic Detrusor Overactivity (NDO) Treated with VESIcare LS Oral Suspension in Studies 1 and 2 Aged 2 to Less than 5 Years
(N=17)
Mean (SD)Aged 5 to 17 Years
(N=49)
Mean (SD)N is the number of patients who took at least one dose and provided valid values for MCC at baseline and Week 24. - * Baseline values for patients aged 5 to 17 years are based on patients who took at least one dose and provided valid baseline
and at least one post-baseline values for MCC.Maximum Cystometric Capacity (mL)
Baseline
Week 24
Change from baseline
98 (40)
137 (37)
39 (36)
95% CI: 21, 57
224 (133)*
279 (127)
57 (108)
95% CI: 26, 88
Results for other urodynamic parameters and from patient urinary diaries, which were secondary efficacy endpoints in Studies 1 and 2, are shown in Tables 5 and 6, respectively.
Table 5: Changes from Baseline in Other Urodynamic Parameters at Week 24 in Pediatric Patients with Neurogenic Detrusor Overactivity (NDO) Treated with VESIcare LS Oral Suspension in Studies 1 and 2 N is the number of patients who took at least one dose and provided valid values for MCC at baseline and Week 24. - * Baseline values for patients aged 5 to 17 years are based on patients who took at least one dose and provided valid baseline
and at least one post-baseline values for MCC.- † For patients who showed a detrusor contraction during the urodynamic assessment at Week 24.
Aged 2 to Less than 5 Years
(N=17)
Mean (SD)
Aged 5 to 17 Years
(N=49)
Mean (SD)
Bladder Compliance (mL/cm H2O)
Baseline
Change from baseline
5.7 (4.9)
5.8 (7.3)
95% CI: 2.2, 9.6
14.6 (36.4)*
9.1 (28.6)
95% CI: 1.0, 17.2
Number of Overactive Detrusor Contractions
(> 15 cm H2O)
Baseline
Change from baseline
9.9 (11.6)
-7.0 (9.3)
95% CI: -11.8, -2.2
3.9 (4.7)*
-2.3 (5.1)
95% CI: -3.7, -0.8
Percentage of Expected Bladder Volume Prior To First Detrusor Contraction > 15 cm H2O†
Baseline (median)
Change from baseline (median)
15.8%
31.1%
27.7%*
13.3%
Table 6: Changes from Baseline in Maximum Catheterized Urine Volume and Number of Incontinence Episodes at Week 24 in Pediatric Patients with Neurogenic Detrusor Overactivity (NDO) Treated with VESIcare LS Oral Suspension in Studies 1 and 2 - * For patients aged 2 to less than 5 years, the number of incontinence episodes per 24 hours was evaluated as the number of periods
between clean intermittent catheterizations with incontinence per 24 hours.Aged 2 to Less than 5 Years
(N=17)
Aged 5 to 17 Years
(N=49)
Maximum Catheterized Urine Volume/Day (mL)
Baseline
Change from baseline
77 (43.0)
45 (54.7)
95% CI: 15, 76
204 (92.7)
68 (88.1)
95% CI: 43, 92
Number of Incontinence Episodes/24 hours*
Baseline
Change from baseline
3.9 (0.8)
-1.6 (1.2)
95% CI: -2.3, -0.9
3.4 (2.9)
-1.6 (2.0)
95% CI: -2.2, -1.0
- * Baseline values for patients aged 5 to 17 years are based on patients who took at least one dose and provided valid baseline
-
16 HOW SUPPLIED/STORAGE AND HANDLING
VESIcare LS is supplied as a white to off-white colored 5 mg/5 mL (1 mg/mL) aqueous oral suspension of solifenacin succinate, packaged in 150 mL amber polyethylene terephthalate (PET) bottles, which are capped with child-resistant high-density polyethylene-polypropylene caps with a pulp and vinyl seal liner.
Carton contains one bottle (NDC: 51248-250-99)
Store at 20°C to 25°C (68°F to 77°F) with excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Store in original bottle to protect from degradation. Dispense in a tight, light-resistant container. Discard any unused product 28 days after opening the original bottle.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Patient Information).
Angioedema and Anaphylactic Reactions
Inform patients and/or their caregivers that angioedema and anaphylactic reactions have been reported in patients treated with solifenacin succinate. Angioedema and anaphylactic reactions may be life-threatening. Advise patients to promptly discontinue VESIcare LS therapy and seek immediate attention if they experience edema of the tongue or laryngopharynx, or difficulty breathing [see Contraindications (4) and Warnings and Precautions (5.1)].
Urinary Retention
Inform patients and/or their caregivers that VESIcare LS may cause urinary retention in patients with conditions associated with bladder outlet obstruction [see Warnings and Precautions (5.2)].
Gastrointestinal Disorders
Inform patients and/or their caregivers that VESIcare LS oral suspension may cause further decrease in gastrointestinal motility in patients with conditions associated with decreased gastrointestinal motility. VESIcare LS oral suspension has been associated with constipation and dry mouth. Advise patients and/or their caregivers to contact their health care providers if they experience severe abdominal pain or become constipated for 3 or more days [see Warnings and Precautions (5.3)].
Central Nervous System Effects
Because VESIcare LS oral suspension, like other antimuscarinic agents, may cause central nervous system effects or blurred vision, advise patients and/or caregivers to exercise caution in decisions to engage in potentially dangerous activities until the drug’s effect on the patient has been determined [see Warnings and Precautions (5.4)].
Narrow-Angle Glaucoma
Inform patients and/or caregivers that VESIcare LS, like other antimuscarinics, may cause worsening of the glaucoma condition in patients with narrow-angle glaucoma [see Warnings and Precautions (5.5)].
Dry Skin
Inform patients and/or caregivers that VESIcare LS, like other antimuscarinics, may cause dry skin due to decreased sweating. Heat prostration due to decreased sweating can occur when antimuscarinic drugs are used in a hot environment [see Adverse Reactions (6.2)].
Administration Instructions
Ensure that the patient has an appropriate measuring device and instructions for measuring the correct dose. Instruct patients or their caregivers to use an oral dosing syringe to correctly measure the prescribed amount of VESIcare LS oral suspension. Inform patients that oral dosing syringes may be obtained from their pharmacy. Instruct patients or their caregivers to shake the bottle well prior to administration of each dose and that patients should take VESIcare LS orally followed by liquid (e.g., water or milk) 1 time a day. Inform patients or their caregivers that simultaneous use of VESIcare LS oral suspension with food and/or other liquids may result in a bitter taste.
- Clean the measuring device and allow to dry after each use.
- Discard VESIcare LS 28 days (4 weeks) after first opening the bottle.
Instruct patients or their caregivers that patients should take VESIcare LS orally followed by liquid (e.g., water or milk). Instruct patients to take any missed doses as soon as they remember, unless more than 12 hours have passed since the missed dose. If more than 12 hours have passed, the missed dose can be skipped and the next dose should be taken at the usual time.
Marketed and Distributed by:
Astellas Pharma US, Inc.
Northbrook, IL 60062VESIcare LS is a registered trademark of Astellas US LLC.
© 2004 – 2022 Astellas Pharma US, Inc.
366572-VES -
PATIENT PACKAGE INSERT
Patient Information
VESIcare LS® (VES-ih-care LS)
(solifenacin succinate)
oral suspension
What is VESIcare LS?
- VESIcare LS is a prescription medicine for children aged 2 years of age and older with a condition called neurogenic detrusor overactivity. It is used to increase the amount of urine your bladder can hold and reduce urine leakage.
Who should not take VESIcare LS?
Do not take VESIcare LS if you:
- have delayed or slow emptying of your stomach (gastric retention).
- have an eye problem called “uncontrolled narrow-angle glaucoma”.
- are allergic to solifenacin succinate or any of the ingredients in VESIcare LS. See the end of this leaflet for a complete list of ingredients.
Before you take VESIcare LS, tell your doctor if you have any medical conditions, including if you:
- have any stomach or intestinal problems or problems with constipation.
- have trouble emptying your bladder or you have a weak urine stream.
- have liver problems.
- have kidney problems.
- have a heart problem called “QT prolongation”.
- are pregnant or plan to become pregnant. It is not known if VESIcare LS will harm your unborn baby. Talk to your doctor if you are pregnant or plan to become pregnant.
- are breastfeeding or plan to breastfeed. It is not known if VESIcare LS passes into your breast milk. Talk to your doctor about the best way to feed your baby if you take VESIcare LS.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. VESIcare LS may affect the way other medicines work, and other medicines may affect how VESIcare LS works.
How should I take VESIcare LS?
- Take VESIcare LS exactly as your doctor tells you to take it.
- If you have questions about dosing, talk to your pharmacist for instructions on how to prepare and administer VESIcare LS.
- Shake the bottle well before taking each dose.
- You should take VESIcare LS by mouth 1 time a day.
- Use an oral dosing device, such as an oral syringe, to correctly measure your dose. Ask your pharmacist for an appropriate oral dosing device.
- You should take VESIcare LS followed by water or milk. Avoid taking VESIcare LS at the same time with food or other drinks, as this may result in a bitter taste.
- Do not take 2 doses of VESIcare LS the same day. If you miss a dose of VESIcare LS, take it as soon as possible. If it has been 12 hours since taking the last dose of VESIcare LS, skip that dose and take the next dose at the usual time.
- If you take too much VESIcare LS, call your doctor or go to the nearest hospital emergency room right away.
What should I avoid while taking VESIcare LS?
VESIcare LS can cause blurred vision or drowsiness. Do not drive or operate heavy machinery until you know how VESIcare LS affects you.
What are the possible side effects of VESIcare LS?
VESIcare LS may cause serious side effects including:
-
Serious allergic reaction. Stop taking VESIcare LS and get medical help right away if you have:
- hives, skin rash, or swelling
- severe itching
- swelling of your face, mouth, or tongue
- trouble breathing
-
Central Nervous System (CNS) effects. Talk to your doctor right away if you get any of these side effects:
- headache
- confusion
- hear voices, see things, or sense things that are not there (hallucinations)
- sleepiness
The most common side effects of VESIcare LS include:
- constipation. Call your doctor if you get severe stomach area (abdominal) pain or become constipated for 3 or more days.
- dry mouth
- urinary tract infection
Other side effects have been observed with anticholinergic medicines such as VESIcare LS and may include:
- blurred vision
- dry eyes
-
dry skin due to decreased sweating. Heat exhaustion or heat stroke due to decreased sweating can happen when anticholinergic medications such as VESIcare LS are used in hot environments. Symptoms may include:
- decreased sweating
- dizziness
- tiredness
- nausea
- increase in body temperature
Tell your doctor if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of VESIcare LS. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store VESIcare LS?
- Store VESIcare LS at room temperature, between 68°F to 77°F (20°C to 25°C) with the cap closed.
- Store VESIcare LS in the original bottle.
- Throw away VESIcare LS oral suspension 28 days (4 weeks) after first opening.
- Safely throw away VESIcare LS that has expired or is unused.
Keep VESIcare LS and all medicines out of the reach of children.
General information about the safe and effective use of VESIcare LS.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use VESIcare LS for a condition for which it was not prescribed. Do not give VESIcare LS to other people, even if they have the same symptoms you have. It may harm them.
This leaflet summarizes the most important information about VESIcare LS. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about VESIcare LS that is written for health professionals.
What are the ingredients in VESIcare LS?
Active ingredient: solifenacin succinate
Inactive ingredients: polacrilin potassium, methylparaben, propylparaben, propylene glycol, simethicone emulsion 30%, carbomer homopolymer Type B, xylitol, acesulfame potassium, natural orange flavor, sodium hydroxide, and purified water.
Marketed and Distributed by:
Astellas Pharma US, Inc.
Northbrook, IL 60062
VESIcare LS is a registered trademark of Astellas US LLC.
© 2004 – 2022 Astellas Pharma US, Inc.
For more information, call 1-800-727-7003.
366572-VESThis Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 10/2022
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
NDC: 51248-250-99
VESIcare LS®
(solifenacin succinate)
Oral Suspension
5 mg/5 mL
(1 mg/mL)
Shake the bottle well before use.
Discard remaining VESIcare LS® 28 days after first opening.
150 mL
-
Package/Label Display Panel
NDC: 51248-250-99
VESIcare LS®
(solifenacin succinate)
Oral Suspension
5 mg/5 mL
(1 mg/mL)
Rx only
Shake well before use.
150 mL
-
INGREDIENTS AND APPEARANCE
VESICARE LS
solifenacin succinate suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 51248-250 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SOLIFENACIN SUCCINATE (UNII: KKA5DLD701) (SOLIFENACIN - UNII:A8910SQJ1U) SOLIFENACIN SUCCINATE 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength POLACRILIN POTASSIUM (UNII: 0BZ5A00FQU) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) DIMETHICONE (UNII: 92RU3N3Y1O) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) XYLITOL (UNII: VCQ006KQ1E) ACESULFAME POTASSIUM (UNII: 23OV73Q5G9) SODIUM HYDROXIDE (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) Product Characteristics Color Score Shape Size Flavor ORANGE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 51248-250-99 1 in 1 CARTON 05/26/2020 07/31/2026 1 150 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA209529 05/26/2020 07/31/2026 Labeler - Astellas Pharma US, Inc. (605764828)
Trademark Results [VESIcare]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 VESICARE 88833757 not registered Live/Pending |
Astellas Pharma Inc. 2020-03-13 |
 VESICARE 75932952 2535639 Live/Registered |
ASTELLAS PHARMA INC. 2000-03-02 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.