TENSION HEADACHE RELIEF- acetaminophen and caffeine tablet
TENSION HEADACHE RELIEF by
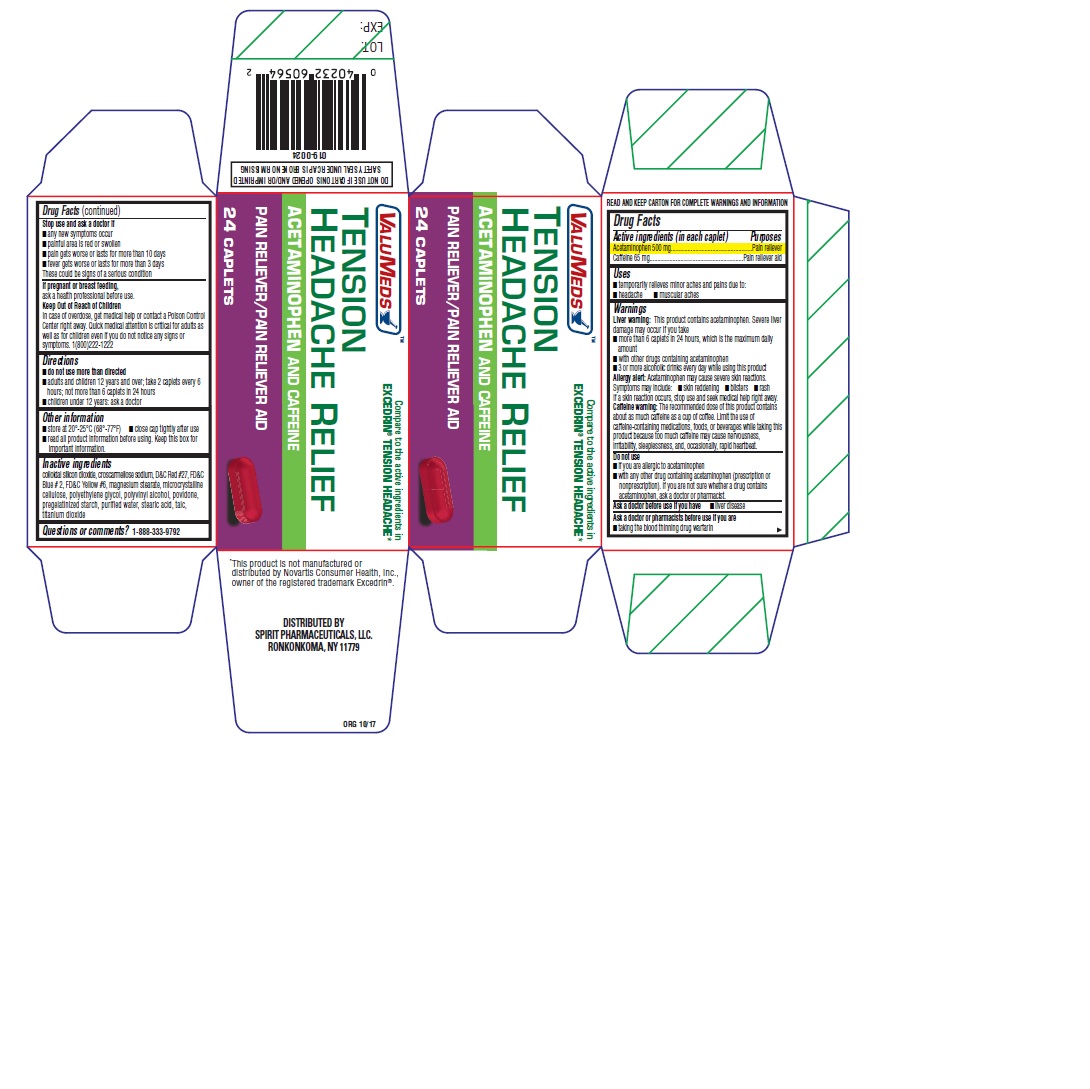
Drug Labeling and Warnings
TENSION HEADACHE RELIEF by is a Otc medication manufactured, distributed, or labeled by SPIRIT PHARMACEUTICALS LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- Uses
-
WARNINGS
Warnings
Liver Warning: This product contains acataminophen. Severe liver damage may occure if you take.
- more than 6 caplets in 24 hours, whihc is the maximum daily amount.
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions.
Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Caffeine warning: The recommended dose of this product contains about as much caffeine as a cup of coffee. Limit the use of caffeine-containing medications, foods, beverages while taking this product because too much caffeine may cause nervousness, irritability, sleeplessness, and, occasionally, rapid heartbeat.
Do not use
- if you are allergic to acetaminophen
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TENSION HEADACHE RELIEF
acetaminophen and caffeine tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 68210-0190 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg CAFFEINE (UNII: 3G6A5W338E) (CAFFEINE - UNII:3G6A5W338E) CAFFEINE 65 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C RED NO. 27 (UNII: 2LRS185U6K) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POVIDONE K30 (UNII: U725QWY32X) STARCH, PREGELATINIZED CORN (UNII: O8232NY3SJ) WATER (UNII: 059QF0KO0R) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color red Score no score Shape OVAL (CAPSULE SHAPED TABLET) Size 18mm Flavor Imprint Code S431 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68210-0190-2 1 in 1 CARTON 04/10/2018 1 24 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC: 68210-0190-1 100 in 1 BOTTLE; Type 0: Not a Combination Product 10/04/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part343 04/10/2018 Labeler - SPIRIT PHARMACEUTICALS LLC (179621011) Establishment Name Address ID/FEI Business Operations ELYSIUM PHARMACEUTICALS LIMITED 915664486 manufacture(68210-0190)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.