INTELLISHADE CLEAR SPF 50- avobenzone, homosalate, octinoxate, octisalate cream
Intellishade Clear SPF 50 by
Drug Labeling and Warnings
Intellishade Clear SPF 50 by is a Otc medication manufactured, distributed, or labeled by Revision, LLC, Goodier Cosmetics LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
Revision Siincare Intellishade Clear Spectrum SPF 50 1.7 oz
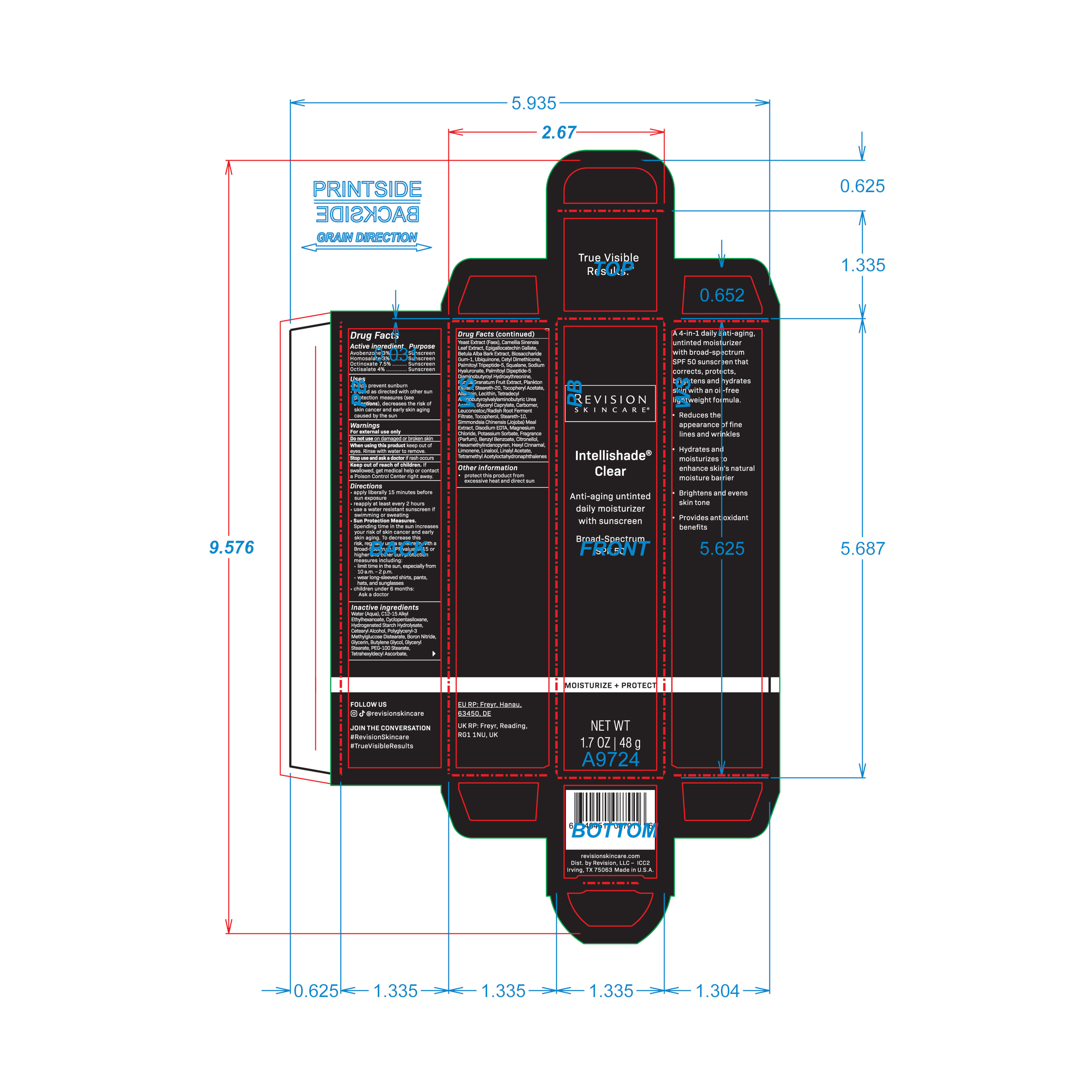
ITC_carton_retail_ICC2_V5_OL
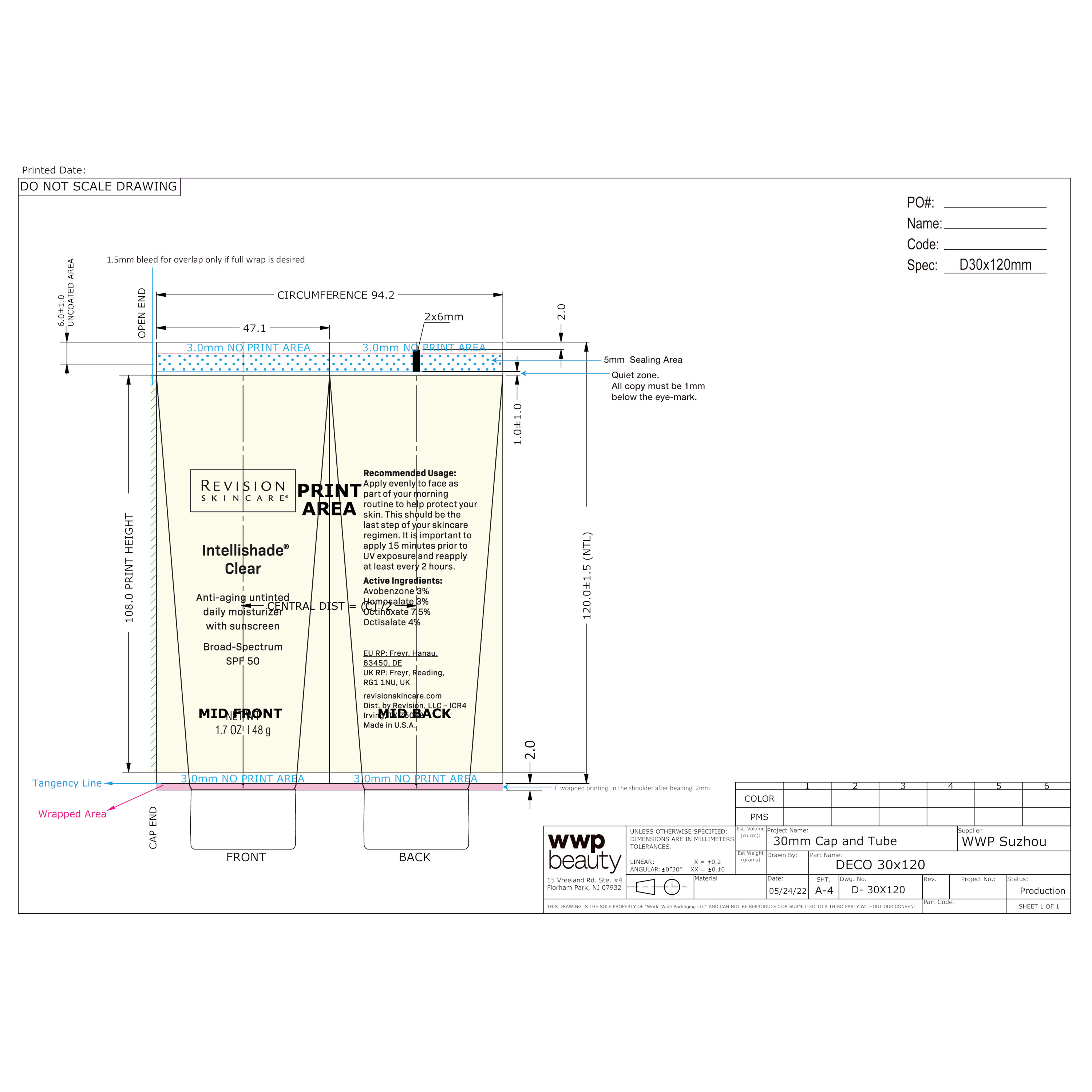
ITC_retail_tube_ICR4_V1_OL
Anti-aging untinted
daily moisturizer
with sunscreen
Broad-Spectrum
SPF 50Moisturize + Protect
NET WT 1.7 OZ | 48 gActive ingredient Purpose
Avobenzone 3% ........... Sunscreen
Homosalate 3% ............ Sunscreen
Octinoxate 7.5% .......... Sunscreen
Octisalate 4% ............... SunscreenUses
helps prevent sunburn
if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sunDirections
apply liberally 15 minutes before sun exposure
reapply at least every 2 hours
use a water resistant sunscreen if swimming or sweating
Sun Protection Measures.
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen
with a Broad-Spectrum SPF value of 15 or higher and other sun protection measures including:
limit time in the sun, especially from 10 a.m. – 2 p.m.
wear long-sleeved shirts, pants, hats, and sunglasses
children under 6 months: Ask a doctorWater (Aqua), C12-15 Alkyl Ethylhexanoate, Cyclopentasiloxane, Hydrogenated Starch Hydrolysate, Cetearyl Alcohol, Polyglyceryl-3 Methylglucose Distearate, Boron Nitride, Glycerin, Butylene Glycol, Glyceryl Stearate, PEG-100 Stearate, Tetrahexyldecyl Ascorbate, Yeast Extract (Faex), Camellia Sinensis Leaf Extract, Epigallocatechin Gallate, Betula Alba Bark Extract, Biosaccharide Gum-1, Ubiquinone, Cetyl Dimethicone, Palmitoyl Tripeptide-5, Squalane, Sodium Hyaluronate, Palmitoyl Dipeptide-5 Diaminobutyroyl Hydroxythreonine, Punica Granatum Fruit Extract, Plankton Extract, Steareth-20, Tocopheryl Acetate, Allantoin, Lecithin, Tetradecyl Aminobutyroylvalylaminobutyric Urea Acetate, Glyceryl Caprylate, Carbomer, Leuconostoc/Radish Root Ferment Filtrate, Tocopherol, Steareth-10, Simmondsia Chinensis (Jojoba) Meal Extract, Disodium EDTA, Magnesium Chloride, Potassium Sorbate, Fragrance (Parfum), Benzyl Benzoate, Citronellol, Hexamethylindanopyran, Hexyl Cinnamal, Limonene, Linalool, Linalyl Acetate, Tetramethyl Acetyloctahydronaphthalenes
-
Revision Siincare Intellishade Clear Spectrum SPF 50 8 oz
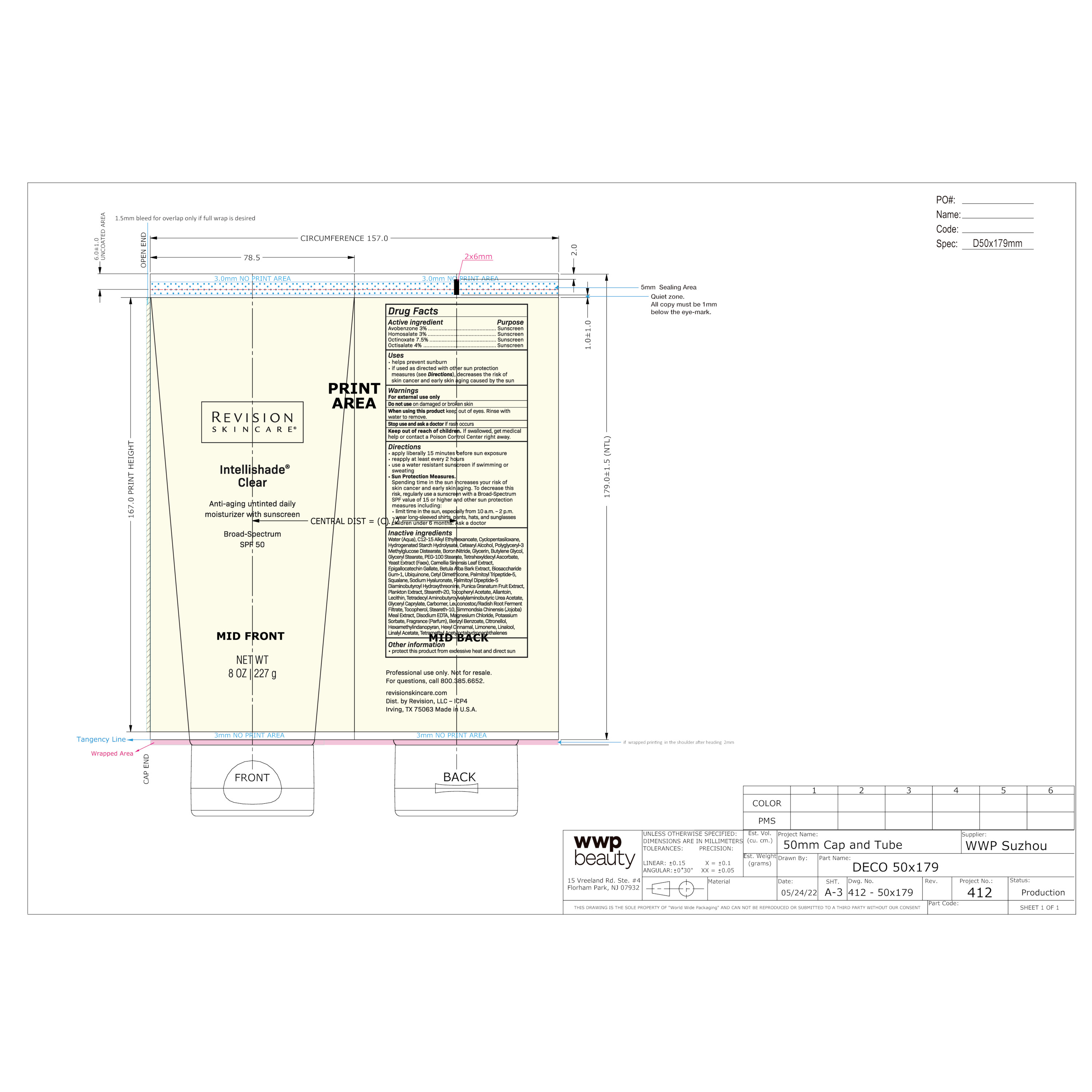
ITC_pro_tube_ICP4
Anti-aging untinted
daily moisturizer
with sunscreen
Broad-Spectrum
SPF 50Moisturize + Protect
NET WT 1.7 OZ | 48 gActive ingredient Purpose
Avobenzone 3% ........... Sunscreen
Homosalate 3% ............ Sunscreen
Octinoxate 7.5% .......... Sunscreen
Octisalate 4% ............... SunscreenUses
helps prevent sunburn
if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sunDirections
apply liberally 15 minutes before sun exposure
reapply at least every 2 hours
use a water resistant sunscreen if swimming or sweating
Sun Protection Measures.
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen
with a Broad-Spectrum SPF value of 15 or higher and other sun protection measures including:
limit time in the sun, especially from 10 a.m. – 2 p.m.
wear long-sleeved shirts, pants, hats, and sunglasses
children under 6 months: Ask a doctorWater (Aqua), C12-15 Alkyl Ethylhexanoate, Cyclopentasiloxane, Hydrogenated Starch Hydrolysate, Cetearyl Alcohol, Polyglyceryl-3 Methylglucose Distearate, Boron Nitride, Glycerin, Butylene Glycol, Glyceryl Stearate, PEG-100 Stearate, Tetrahexyldecyl Ascorbate, Yeast Extract (Faex), Camellia Sinensis Leaf Extract, Epigallocatechin Gallate, Betula Alba Bark Extract, Biosaccharide Gum-1, Ubiquinone, Cetyl Dimethicone, Palmitoyl Tripeptide-5, Squalane, Sodium Hyaluronate, Palmitoyl Dipeptide-5 Diaminobutyroyl Hydroxythreonine, Punica Granatum Fruit Extract, Plankton Extract, Steareth-20, Tocopheryl Acetate, Allantoin, Lecithin, Tetradecyl Aminobutyroylvalylaminobutyric Urea Acetate, Glyceryl Caprylate, Carbomer, Leuconostoc/Radish Root Ferment Filtrate, Tocopherol, Steareth-10, Simmondsia Chinensis (Jojoba) Meal Extract, Disodium EDTA, Magnesium Chloride, Potassium Sorbate, Fragrance (Parfum), Benzyl Benzoate, Citronellol, Hexamethylindanopyran, Hexyl Cinnamal, Limonene, Linalool, Linalyl Acetate, Tetramethyl Acetyloctahydronaphthalenes
- DOSAGE & ADMINISTRATION
- PURPOSE
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
INTELLISHADE CLEAR SPF 50
avobenzone, homosalate, octinoxate, octisalate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 64651-7720 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 7.5 g in 100 g OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 4 g in 100 g AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 g in 100 g HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 3 g in 100 g Inactive Ingredients Ingredient Name Strength CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) BORON NITRIDE (UNII: 2U4T60A6YD) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) PEG-100 STEARATE (UNII: YD01N1999R) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) TETRAHEXYLDECYL ASCORBATE (UNII: 9LBV3F07AZ) YEAST (UNII: 3NY3SM6B8U) GREEN TEA LEAF (UNII: W2ZU1RY8B0) EPIGALLOCATECHIN GALLATE (UNII: BQM438CTEL) BETULA PUBESCENS BARK (UNII: 3R504894L9) BIOSACCHARIDE GUM-1 (UNII: BB4PU4V09H) UBIDECARENONE (UNII: EJ27X76M46) CETYL DIMETHICONE 25 (UNII: U4AS1BW4ZB) PALMITOYL TRIPEPTIDE-5 (UNII: 2A3916MQHO) SQUALANE (UNII: GW89575KF9) HYALURONATE SODIUM (UNII: YSE9PPT4TH) PALMITOYLLYSYLVALYLDIAMINOBUTYROYLTHREONINE (UNII: 1615WE9073) POMEGRANATE (UNII: 56687D1Z4D) STEARETH-20 (UNII: L0Q8IK9E08) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) ALLANTOIN (UNII: 344S277G0Z) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) CARBOMER INTERPOLYMER TYPE A (ALLYL SUCROSE CROSSLINKED) (UNII: 59TL3WG5CO) TOCOPHEROL (UNII: R0ZB2556P8) STEARETH-10 (UNII: FD0913P475) SIMMONDSIA CHINENSIS SEED (UNII: D24K2Q1F6H) EDETATE DISODIUM (UNII: 7FLD91C86K) MAGNESIUM CHLORIDE (UNII: 02F3473H9O) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 64651-7720-1 1 in 1 CARTON 06/26/2013 1 48 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC: 64651-7720-2 227 g in 1 TUBE; Type 0: Not a Combination Product 06/26/2013 3 NDC: 64651-7720-3 3 g in 1 TUBE; Type 0: Not a Combination Product 06/26/2013 
Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 06/26/2013 Labeler - Revision, LLC (805995131) Registrant - Revision, LLC (805995131) Establishment Name Address ID/FEI Business Operations Goodier Cosmetics LLC 007317209 manufacture(64651-7720)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.