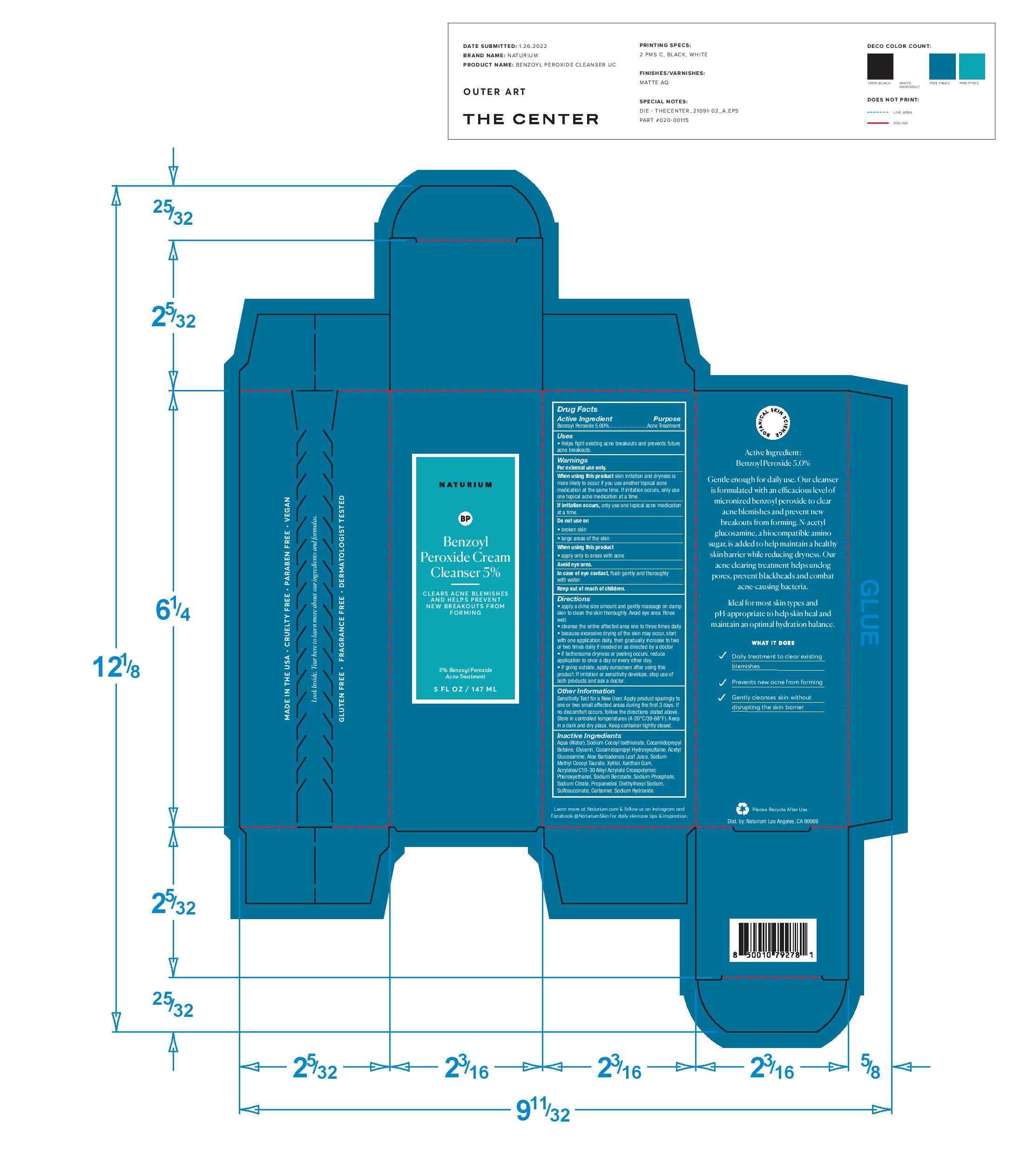
Naturium Benzoyl Peroxide Cream Cleanser 5%
Benzoyl Peroxide by
Drug Labeling and Warnings
Benzoyl Peroxide by is a Otc medication manufactured, distributed, or labeled by The Center Brands, LLC, DIMENSIONAL MERCHANDISING INC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
BENZOYL PEROXIDE- naturium benzoyl peroxide cream cleanser 5% cream
The Center Brands, LLC
----------
Naturium Benzoyl Peroxide Cream Cleanser 5%
Warnings
For external use only.
When using this product skin irritation and dryness is
more likely to occur if you use another topical acne
medication at the same time. If irritation occurs, only use
one topical acne medication at a time.
If irritation occurs, only use one topical acne medication
at a time.
Do not use on
broken skin
large areas of the skin
When using this product
apply only to areas with acne
Avoid eye area.
In case of eye contact, flush gently and thoroughly
with water.
Keep out of reach of children.
Directions
apply a dime size amount and gently massage on damp
skin to clean the skin thoroughly. Avoid eye area. Rinse
well.
cleanse the entire affected area one to three times daily
because excessive drying of the skin may occur, start
with one application daily, then gradually increase to two
or two times daily if needed or as directed by a doctor
if bothersome dryness or peeling occurs, reduce
application to once a day or every other day.
if going outside, apply sunscreen after using this
product. If irritation or sensitivity develops, stop use of
both products and ask a doctor.
Directions
apply a dime size amount and gently massage on damp
skin to clean the skin thoroughly. Avoid eye area. Rinse
well.
cleanse the entire affected area one to three times daily
because excessive drying of the skin may occur, start
with one application daily, then gradually increase to two
or two times daily if needed or as directed by a doctor
if bothersome dryness or peeling occurs, reduce
application to once a day or every other day.
if going outside, apply sunscreen after using this
product. If irritation or sensitivity develops, stop use of
both products and ask a doctor.
Other Information
Sensitivity Test for a New User. Apply product sparingly to
one or two small affected areas during the first 3 days. If
no discomfort occurs, follow the directions stated above.
Store in controlled temperatures (4-20°C/39-68°F). Keep
in a dark and dry place. Keep container tightly closed.
Inactive Ingredients
Aqua (Water), Sodium Cocoyl Isethionate, Cocamidopropyl
Betaine, Glycerin, Cocamidopropyl Hydroxysultaine, Acetyl
Glucosamine, Aloe Barbadensis Leaf Juice, Sodium
Methyl Cocoyl Taurate, Xylitol, Xanthan Gum,
Acrylates/C10-30 Alkyl Acrylate Crosspolymer,
Phenoxyethanol, Sodium Benzoate, Sodium Phosphate,
Sodium Citrate, Propanediol, Diethylhexyl Sodium,
Sulfosuccinate, Carbomer, Sodium Hydroxide.
| BENZOYL PEROXIDE
naturium benzoyl peroxide cream cleanser 5% cream |
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||
| Labeler - The Center Brands, LLC (076228814) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Paklab | 177711082 | manufacture(82800-001) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.