MOMETASONE FUROATE spray, metered
MOMETASONE FUROATE by
Drug Labeling and Warnings
MOMETASONE FUROATE by is a Prescription medication manufactured, distributed, or labeled by Golden State Medical Supply, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
MOMETASONE FUROATE NASAL SPRAY
These highlights do not include all the information needed to use MOMETASONE FUROATE NASAL SPRAY safely and effectively. See full prescribing information for MOMETASONE FUROATE NASAL SPRAY.
MOMETASONE FUROATE Nasal Spray, 50 mcg
Initial U.S. Approval: 1997RECENT MAJOR CHANGES
Warnings and Precautions Glaucoma and Cataracts ( 5.2) 06/2018
INDICATIONS AND USAGE
Mometasone furoate nasal spray is a corticosteroid indicated for:
- Treatment of Nasal Symptoms of Allergic Rhinitis in patients ≥2 years of age ( 1.1)
- Treatment of Nasal Congestion Associated with Seasonal Allergic Rhinitis in patients ≥2 years of age ( 1.2)
- Prophylaxis of Seasonal Allergic Rhinitis in patients ≥12 years of age ( 1.3)
- Treatment of Nasal Polyps in patients ≥18 years of age ( 1.4)
DOSAGE AND ADMINISTRATION
For Intranasal Use Only
- Treatment of Nasal Symptoms of Allergic Rhinitis (
2.1)
Adults & Adolescents (12 yrs. and older): 2 sprays in each nostril once daily
Children (2–11 yrs.): 1 spray in each nostril once daily - Treatment of Nasal Congestion Associated with Seasonal Allergic Rhinitis (
2.2)
Adults & Adolescents (12 yrs. and older): 2 sprays in each nostril once daily
Children (2–11 yrs.): 1 spray in each nostril once daily - Prophylaxis of Seasonal Allergic Rhinitis (
2.3)
Adults & Adolescents (12 yrs. and older): 2 sprays in each nostril once daily - Treatment of Nasal Polyps (
2.4)
Adults (18 yrs. and older): 2 sprays in each nostril twice daily. 2 sprays in each nostril once daily may also be effective in some patients.
DOSAGE FORMS AND STRENGTHS
Nasal Spray: 50 mcg of mometasone furoate in each 100-microliter spray ( 3)
CONTRAINDICATIONS
Patients with known hypersensitivity to mometasone furoate or any of the ingredients of mometasone furoate nasal spray. ( 4)
WARNINGS AND PRECAUTIONS
- Epistaxis, nasal ulceration, Candida albicans infection, nasal septal perforation, impaired wound healing. Monitor patients periodically for signs of adverse effects on the nasal mucosa. Avoid use in patients with recent nasal ulcers, nasal surgery, or nasal trauma. ( 5.1)
- Glaucoma and cataracts. Consider referral to an ophthalmologist in patients who develop ocular symptoms or use mometasone furoate nasal spray long term. ( 5.2)
- Potential worsening of existing tuberculosis; fungal, bacterial, viral, or parasitic infections; or ocular herpes simplex. More serious or even fatal course of chickenpox or measles in susceptible patients. Use caution in patients with the above because of the potential for worsening of these infections. ( 5.4)
- Hypercorticism and adrenal suppression with higher than recommended dosages or at the regular dosage in susceptible individuals. If such changes occur, discontinue mometasone furoate nasal spray slowly. ( 5.5)
- Potential reduction in growth velocity in children. Monitor growth routinely in pediatric patients receiving mometasone furoate nasal spray. ( 5.6, 8.4)
ADVERSE REACTIONS
The most common adverse reactions (≥5%) included headache, viral infection, pharyngitis, epistaxis and cough. ( 6)
To report SUSPECTED ADVERSE REACTIONS, contact Apotex Corp. at 1-800-706-5575 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 7/2018
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1. INDICATIONS AND USAGE
1.1 Treatment of Allergic Rhinitis
1.2 Treatment of Nasal Congestion Associated with Seasonal Allergic Rhinitis
1.3 Prophylaxis of Seasonal Allergic Rhinitis
1.4 Treatment of Nasal Polyps
2. DOSAGE AND ADMINISTRATION
2.1 Treatment of Allergic Rhinitis
2.2 Treatment of Nasal Congestion Associated with Seasonal Allergic Rhinitis
2.3 Prophylaxis of Seasonal Allergic Rhinitis
2.4 Treatment of Nasal Polyps
3. DOSAGE FORMS AND STRENGTHS
4. CONTRAINDICATIONS
5. WARNINGS AND PRECAUTIONS
5.1 Local Nasal Effects
5.2 Glaucoma and Cataracts
5.3 Hypersensitivity Reactions
5.4 Immunosuppression
5.5 Hypothalamic-Pituitary-Adrenal Axis Effect
5.6 Effect on Growth
6. ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Post-Marketing Experience
7. DRUG INTERACTIONS
8. USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
10. OVERDOSAGE
11. DESCRIPTION
12. CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13. NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14. CLINICAL STUDIES
14.1 Allergic Rhinitis in Adults and Adolescents
14.2 Allergic Rhinitis in Pediatrics
14.3 Nasal Polyps in Adults 18 Years of Age and Older
14.4 Nasal Congestion Associated with Seasonal Allergic Rhinitis
16. HOW SUPPLIED/STORAGE AND HANDLING
17. PATIENT COUNSELING INFORMATION
17.1 Local Nasal Effect
17.2 Glaucoma and Cataracts
17.3 Immunosuppression
17.4 Use Regularly for Best Effect
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1. INDICATIONS AND USAGE
1.1 Treatment of Allergic Rhinitis
Mometasone furoate nasal spray 50 mcg is indicated for the treatment of the nasal symptoms of seasonal allergic and perennial allergic rhinitis, in adults and pediatric patients 2 years of age and older.
1.2 Treatment of Nasal Congestion Associated with Seasonal Allergic Rhinitis
Mometasone furoate nasal spray 50 mcg is indicated for the relief of nasal congestion associated with seasonal allergic rhinitis, in adults and pediatric patients 2 years of age and older.
-
2. DOSAGE AND ADMINISTRATION
Administer mometasone furoate nasal spray 50 mcg by the intranasal route only. Prior to initial use of mometasone furoate nasal spray, 50 mcg, the pump must be primed by actuating ten times or until a fine spray appears. The pump may be stored unused for up to 1 week without repriming. If unused for more than 1 week, reprime by actuating two times, or until a fine spray appears.
2.1 Treatment of Allergic Rhinitis
2.2 Treatment of Nasal Congestion Associated with Seasonal Allergic Rhinitis
2.3 Prophylaxis of Seasonal Allergic Rhinitis
Adults and Adolescents 12 Years of Age and Older:
The recommended dose for prophylaxis treatment of nasal symptoms of seasonal allergic rhinitis is 2 sprays (50 mcg of mometasone furoate in each spray) in each nostril once daily (total daily dose of 200 mcg).
In patients with a known seasonal allergen that precipitates nasal symptoms of seasonal allergic rhinitis, prophylaxis with mometasone furoate nasal spray 50 mcg (200 mcg/day) is recommended 2 to 4 weeks prior to the anticipated start of the pollen season.
2.4 Treatment of Nasal Polyps
Adults 18 Years of Age and Older:
The recommended dose for the treatment of nasal polyps is 2 sprays (50 mcg of mometasone furoate in each spray) in each nostril twice daily (total daily dose of 400 mcg). A dose of 2 sprays (50 mcg of mometasone furoate in each spray) in each nostril once daily (total daily dose of 200 mcg) is also effective in some patients.
-
3. DOSAGE FORMS AND STRENGTHS
Mometasone furoate nasal spray 50 mcg is a metered-dose, manual pump spray unit containing an aqueous suspension of 0.05% w/w mometasone furoate.
After initial priming (10 actuations), each actuation of the pump delivers a metered spray containing 100 mg or 100 microliter of suspension containing 50 mcg of mometasone furoate. Each bottle of mometasone furoate nasal spray 50 mcg provides 120 sprays.
- 4. CONTRAINDICATIONS
-
5. WARNINGS AND PRECAUTIONS
5.1 Local Nasal Effects
Epistaxis
In clinical studies, epistaxis was observed more frequently in patients with allergic rhinitis with mometasone furoate nasal spray than those who received placebo [ see Adverse Reactions (6)] .
Candida Infection
In clinical studies with mometasone furoate nasal spray 50 mcg, the development of localized infections of the nose and pharynx with Candida albicans has occurred. When such an infection develops, use of mometasone furoate nasal spray 50 mcg should be discontinued and appropriate local or systemic therapy instituted, if needed.
Nasal Septum Perforation
Instances of nasal septum perforation have been reported following the intranasal application of corticosteroids. As with any long-term topical treatment of the nasal cavity, patients using mometasone furoate nasal spray 50 mcg over several months or longer should be examined periodically for possible changes in the nasal mucosa.
5.2 Glaucoma and Cataracts
Glaucoma and cataracts may be reported with systemic and topical (including intranasal, inhaled and intraocular) corticosteroid use. Consider referral to an ophthalmologist in patients who develop ocular symptoms or use mometasone furoate nasal spray long term [see Adverse Reactions ( 6)].
Glaucoma and cataract formation was evaluated in one controlled study of 12 weeks' duration and one uncontrolled study of 12 months' duration in patients treated with mometasone furoate nasal spray, 50 mcg at 200 mcg/day, using intraocular pressure measurements and slit lamp examination. No significant change from baseline was noted in the mean intraocular pressure measurements for the 141 mometasone furoate nasal spray-treated patients in the 12-week study, as compared with 141 placebo-treated patients. No individual mometasone furoate nasal spray-treated patient was noted to have developed a significant elevation in intraocular pressure or cataracts in this 12-week study. Likewise, no significant change from baseline was noted in the mean intraocular pressure measurements for the 139 mometasone furoate nasal spray-treated patients in the 12-month study and again, no cataracts were detected in these patients. Nonetheless, intranasal and inhaled corticosteroids have been associated with the development of glaucoma and/or cataracts.
5.3 Hypersensitivity Reactions
Hypersensitivity reactions including instances of wheezing may occur after the intranasal administration of mometasone furoate monohydrate. Discontinue mometasone furoate nasal spray if such reactions occur [ see Contraindications (4)] .
5.4 Immunosuppression
Persons who are on drugs which suppress the immune system are more susceptible to infections than healthy individuals. Chickenpox and measles, for example, can have a more serious or even fatal course in nonimmune children or adults on corticosteroids. In such children or adults who have not had these diseases, particular care should be taken to avoid exposure. How the dose, route, and duration of corticosteroid administration affect the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) may be indicated. If exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. (See the respective package inserts for complete VZIG and IG prescribing information.) If chickenpox develops, treatment with antiviral agents may be considered.
Corticosteroids should be used with caution, if at all, in patients with active or quiescent tuberculous infection of the respiratory tract, or in untreated fungal, bacterial, systemic viral infections, or ocular herpes simplex because of the potential for worsening of these infections.
5.5 Hypothalamic-Pituitary-Adrenal Axis Effect
Hypercorticism and Adrenal Suppression
When intranasal steroids are used at higher than recommended dosages or in susceptible individuals at recommended dosages, systemic corticosteroid effects such as hypercorticism and adrenal suppression may appear. If such changes occur, the dosage of mometasone furoate nasal spray should be discontinued slowly, consistent with accepted procedures for discontinuing oral corticosteroid therapy.
5.6 Effect on Growth
Corticosteroids may cause a reduction in growth velocity when administered to pediatric patients. Monitor the growth routinely of pediatric patients receiving mometasone furoate nasal spray. To minimize the systemic effects of intranasal corticosteroids, including mometasone furoate nasal spray, titrate each patient's dose to the lowest dosage that effectively controls his/her symptoms [ see Use in Specific Populations (8.4)].
-
6. ADVERSE REACTIONS
Systemic and local corticosteroid use may result in the following:
- Epistaxis, ulcerations, Candida albicans infection, impaired wound healing [ see Warnings and Precautions (5.1)]
- Glaucoma and cataracts [ see Warnings and Precautions (5.2)]
- Immunosuppression [ see Warnings and Precautions (5.4)]
- Hypothalamic-pituitary-adrenal (HPA) axis effects, including growth reduction [ see Warnings and Precautions (5.5, 5.6), Use in Specific Populations (8.4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Allergic Rhinitis
Adults and adolescents 12 years of age and older
In controlled US and international clinical studies, a total of 3210 adult and adolescent patients 12 years and older with allergic rhinitis received treatment with mometasone furoate nasal spray 50 mcg at doses of 50 to 800 mcg/day. The majority of patients (n = 2103) were treated with 200 mcg/day. A total of 350 adult and adolescent patients have been treated for one year or longer. Adverse events did not differ significantly based on age, sex, or race. Four percent or less of patients in clinical trials discontinued treatment because of adverse events and the discontinuation rate was similar for the vehicle and active comparators.
All adverse events (regardless of relationship to treatment) reported by 5% or more of adult and adolescent patients ages 12 years and older who received mometasone furoate nasal spray 50 mcg, 200 mcg/day vs. placebo and that were more common with mometasone furoate nasal spray 50 mcg than placebo, are displayed in TABLE 1 below.
TABLE 1: ADULT AND ADOLESCENT PATIENTS 12 YEARS AND OLDER – ADVERSE EVENTS FROM CONTROLLED CLINICAL TRIALS IN SEASONAL ALLERGIC AND PERENNIAL ALLERGIC RHINITIS (PERCENT OF PATIENTS REPORTING) Mometasone Furoate Nasal Spray
200 mcg
(n = 2103)Vehicle Placebo
(n = 1671)Headache 26 22 Viral Infection 14 11 Pharyngitis 12 10 Epistaxis/Blood-Tinged Mucus 11 6 Coughing 7 6 Upper Respiratory Tract Infection 6 2 Dysmenorrhea 5 3 Musculoskeletal Pain 5 3 Sinusitis 5 3 Other adverse events which occurred in less than 5% but greater than or equal to 2% of adult and adolescent patients (ages 12 years and older) treated with mometasone furoate nasal spray 50 mcg, 200-mcg/day (regardless of relationship to treatment), and more frequently than in the placebo group included: arthralgia, asthma, bronchitis, chest pain, conjunctivitis, diarrhea, dyspepsia, earache, flu-like symptoms, myalgia, nausea, and rhinitis.
Pediatric patients <12 years of age
In controlled US and international studies, a total of 990 pediatric patients (ages 3 to 11 years) with allergic rhinitis received treatment with mometasone furoate nasal spray 50 mcg, at doses of 25 to 200 mcg/day. The majority of pediatric patients (n = 720) were treated with 100 mcg/day. A total of 163 pediatric patients have been treated for one year or longer. Two percent or less of patients in clinical trials who received mometasone furoate nasal spray 50 mcg discontinued treatment because of adverse events and the discontinuation rate was similar for the placebo and active comparators.
Adverse events which occurred in ≥5% of pediatric patients (ages 3 to 11 years) treated with mometasone furoate nasal spray 50 mcg, 100 mcg/day vs. placebo (regardless of relationship to treatment) and more frequently than in the placebo group included upper respiratory tract infection (5% in mometasone furoate nasal spray 50 mcg group vs. 4% in placebo) and vomiting (5% in mometasone furoate nasal spray 50 mcg group vs. 4% in placebo).
Other adverse events which occurred in less than 5% but greater than or equal to 2% of pediatric patients (ages 3 to 11 years) treated with mometasone furoate nasal spray 50 mcg, 100 mcg/day vs. placebo (regardless of relationship to treatment) and more frequently than in the placebo group included: diarrhea, nasal irritation, otitis media, and wheezing.
The adverse event (regardless of relationship to treatment) reported by 5% of pediatric patients ages 2 to 5 years who received mometasone furoate nasal spray, 50 mcg, 100 mcg/day in a clinical trial vs. placebo including 56 subjects (28 each mometasone furoate nasal spray, 50 mcg and placebo) and that was more common with mometasone furoate nasal spray, 50 mcg than placebo, included: upper respiratory tract infection (7% vs. 0%, respectively). The other adverse event which occurred in less than 5% but greater than or equal to 2% of mometasone furoate pediatric patients ages 2 to 5 years treated with 100 mcg doses vs. placebo (regardless of relationship to treatment) and more frequently than in the placebo group included: skin trauma.
Nasal Polyps
Adults 18 years of age and older
In controlled clinical studies, the types of adverse events observed in patients with nasal polyps were similar to those observed for patients with allergic rhinitis. A total of 594 adult patients (ages 18 to 86 years) received mometasone furoate nasal spray 50 mcg at doses of 200 mcg once or twice daily for up to 4 months for treatment of nasal polyps. The overall incidence of adverse events for patients treated with mometasone furoate nasal spray 50 mcg was comparable to patients with the placebo except for epistaxis, which was 9% for 200 mcg once daily, 13% for 200 mcg twice daily, and 5% for the placebo.
Nasal ulcers and nasal and oral candidiasis were also reported in patients treated with mometasone furoate nasal spray 50 mcg primarily in patients treated for longer than 4 weeks.
Nasal Congestion Associated with Seasonal Allergic Rhinitis
A total of 1008 patients aged 12 years and older received mometasone furoate nasal spray 50 mcg 200 mcg/day (n = 506) or placebo (n = 502) for 15 days. Adverse events that occurred more frequently in patients treated with mometasone furoate nasal spray 50 mcg than in patients with the placebo included sinus headache (1.2% in mometasone furoate nasal spray 50 mcg group vs. 0.2% in placebo) and epistaxis (1% in mometasone furoate nasal spray 50 mcg group vs. 0.2% in placebo) and the overall adverse event profile was similar to that observed in the other allergic rhinitis trials.
6.2 Post-Marketing Experience
The following adverse reactions have been identified during the post-marketing period for mometasone furoate nasal spray 50 mcg: nasal burning and irritation, anaphylaxis and angioedema, disturbances in taste and smell, nasal septal perforation, and vision blurred. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
-
7. DRUG INTERACTIONS
No formal drug-drug interaction studies have been conducted with mometasone furoate nasal spray 50 mcg.
Inhibitors of Cytochrome P450 3A4: Studies have shown that mometasone furoate is primarily and extensively metabolized in the liver of all species investigated and undergoes extensive metabolism to multiple metabolites. In vitro studies have confirmed the primary role of cytochrome CYP 3A4 in the metabolism of this compound.
Concomitant administration of CYP3A4 inhibitors may inhibit the metabolism of, and increase the systemic exposure to, mometasone furoate and potentially increase the risk for systemic corticosteroid side effects. Caution should be exercised when considering the coadministration of mometasone furoate nasal spray 50 mcg with long-term ketoconazole and other known strong CYP3A4 inhibitors (e.g., ritonavir, cobicistat-containing products, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, telithromycin) [see Clinical Pharmacology ( 12.3)]. Consider the benefit of coadministration versus the potential risk of systemic corticosteroid effects, in which case patients should be monitored for systemic corticosteroid side effects.
-
8. USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects: Pregnancy Category C: There are no adequate and well-controlled studies in pregnant women. Mometasone furoate nasal spray 50 mcg, like other corticosteroids, should be used during pregnancy only if the potential benefits justify the potential risk to the fetus. Experience with oral corticosteroids since their introduction in pharmacologic, as opposed to physiologic, doses suggests that rodents are more prone to teratogenic effects from corticosteroids than humans. In addition, because there is a natural increase in corticosteroid production during pregnancy, most women will require a lower exogenous corticosteroid dose and many will not need corticosteroid treatment during pregnancy.
In mice, mometasone furoate caused cleft palate at subcutaneous doses (less than the MRDID in adults on a mcg/m 2 basis). Fetal survival was reduced at approximately 2 times the MRDID in adults on a mcg/m 2 basis. No toxicity was observed at less than the MRDID in adults on a mcg/m 2 basis.
In rats, mometasone furoate produced umbilical hernia at topical dermal doses approximately 10 times the MRDID in adults on a mcg/m 2 basis. A topical dermal dose approximately 6 times the MRDID in adults on a mcg/m 2 basis produced delays in ossification, but no malformations.
In rabbits, mometasone furoate caused multiple malformations (e.g., flexed front paws, gallbladder agenesis, umbilical hernia, and hydrocephaly) at topical dermal doses approximately 6 times the MRDID in adults on a mcg/m 2 basis. In an oral study, mometasone furoate increased resorptions and caused cleft palate and/or head malformations (hydrocephaly or domed head) at approximately 30 times the MRDID in adults on a mcg/m 2 basis. At approximately 110 times the MRDID in adults on a mcg/m 2 basis, most litters were aborted or resorbed. No toxicity was observed at approximately 6 times the MRDID in adults on a mcg/m 2 basis.
When rats received subcutaneous doses of mometasone furoate throughout pregnancy or during the later stages of pregnancy, a dose less than the MRDID in adults on a mcg/m 2 basis caused prolonged and difficult labor and reduced the number of live births, birth weight, and early pup survival.
8.3 Nursing Mothers
It is not known if mometasone furoate is excreted in human milk. Because other corticosteroids are excreted in human milk, caution should be used when mometasone furoate nasal spray, 50 mcg is administered to nursing women.
8.4 Pediatric Use
The safety and effectiveness of mometasone furoate nasal spray 50 mcg for allergic rhinitis in children 12 years of age and older have been established [ see Adverse Reactions (6.1) and Clinical Studies (14.1)]. Use of mometasone furoate nasal spray 50 mcg for allergic rhinitis in pediatric patients 2 to 11 years of age is supported by safety and efficacy data from clinical studies. Seven hundred and twenty (720) patients 3 to 11 years of age with allergic rhinitis were treated with mometasone furoate nasal spray 50 mcg (100 mcg total daily dose) in controlled clinical trials [ see Adverse Reactions (6.1) and Clinical Studies (14.2)]. Twenty-eight (28) patients 2 to 5 years of age with allergic rhinitis were treated with mometasone furoate nasal spray 50 mcg (100 mcg total daily dose) in a controlled trial to evaluate safety [ see Adverse Reactions (6.1)]. Safety and effectiveness of mometasone furoate nasal spray 50 mcg for allergic rhinitis in children less than 2 years of age have not been established.
The safety and effectiveness of mometasone furoate nasal spray for the treatment of nasal polyps in children less than 18 years of age have not been established. One 4-month trial was conducted to evaluate the safety and efficacy of mometasone furoate nasal spray in the treatment of nasal polyps in pediatric patients 6 to 17 years of age. The primary objective of the study was to evaluate safety; efficacy parameters were collected as secondary endpoints. A total of 127 patients with nasal polyps were randomized to placebo or mometasone furoate nasal spray 100 mcg once or twice daily (patients 6 to 11 years of age) or 200 mcg once or twice daily (patients 12 to 17 years of age). The results of this trial did not support the efficacy of mometasone furoate nasal spray in the treatment of nasal polyps in pediatric patients. The adverse events reported in this trial were similar to the adverse events reported in patients 18 years of age and older with nasal polyps.
Controlled clinical studies have shown intranasal corticosteroids may cause a reduction in growth velocity in pediatric patients. This effect has been observed in the absence of laboratory evidence of hypothalamic-pituitary-adrenal (HPA) axis suppression, suggesting that growth velocity is a more sensitive indicator of systemic corticosteroid exposure in pediatric patients than some commonly used tests of HPA axis function. The long-term effects of this reduction in growth velocity associated with intranasal corticosteroids, including the impact on final adult height, are unknown. The potential for "catch up" growth following discontinuation of treatment with intranasal corticosteroids has not been adequately studied. The growth of pediatric patients receiving intranasal corticosteroids, including mometasone furoate nasal spray, 50 mcg, should be monitored routinely (e.g., via stadiometry). The potential growth effects of prolonged treatment should be weighed against clinical benefits obtained and the availability of safe and effective noncorticosteroid treatment alternatives. To minimize the systemic effects of intranasal corticosteroids, including mometasone furoate nasal spray, 50 mcg, each patient should be titrated to his/her lowest effective dose.
A clinical study to assess the effect of mometasone furoate nasal spray 50 mcg (100 mcg total daily dose) on growth velocity has been conducted in pediatric patients 3 to 9 years of age with allergic rhinitis. No statistically significant effect on growth velocity was observed for mometasone furoate nasal spray 50 mcg compared to placebo following one year of treatment. No evidence of clinically relevant HPA axis suppression was observed following a 30-minute cosyntropin infusion.
The potential of mometasone furoate nasal spray 50 mcg to cause growth suppression in susceptible patients or when given at higher doses cannot be ruled out.
8.5 Geriatric Use
A total of 280 patients above 64 years of age with allergic rhinitis or nasal polyps (age range 64 to 86 years) have been treated with mometasone furoate nasal spray 50 mcg for up to 3 or 4 months, respectively. The adverse reactions reported in this population were similar in type and incidence to those reported by younger patients.
8.6 Hepatic Impairment
Concentrations of mometasone furoate appear to increase with severity of hepatic impairment [see Clinical Pharmacology (12.3)] .
-
10. OVERDOSAGE
There are no data available on the effects of acute or chronic overdosage with mometasone furoate nasal spray 50 mcg. Because of low systemic bioavailability, and an absence of acute drug-related systemic findings in clinical studies, overdose is unlikely to require any therapy other than observation. Intranasal administration of 1600 mcg (4 times the recommended dose of mometasone furoate nasal spray 50 mcg for the treatment of nasal polyps in patients 18 years of age and older) daily for 29 days, to healthy human volunteers, showed no increased incidence of adverse events. Single intranasal doses up to 4000 mcg and oral inhalation doses up to 8000 mcg have been studied in human volunteers with no adverse effects reported. Chronic over dosage with any corticosteroid may result in signs or symptoms of hypercorticism [ see Warnings and Precautions ( 5.5) ]. Acute overdosage with this dosage form is unlikely since one bottle of mometasone furoate nasal spray 50 mcg contains approximately 8500 mcg of mometasone furoate.
-
11. DESCRIPTION
Mometasone furoate, the active component of mometasone furoate nasal spray, 50 mcg, is an anti-inflammatory corticosteroid having the chemical name, 9,21-Dichloro-11ß,17-dihydroxy-16α-methylpregna-1,4-diene-3,20-dione17-(2 furoate), and the following chemical structure:
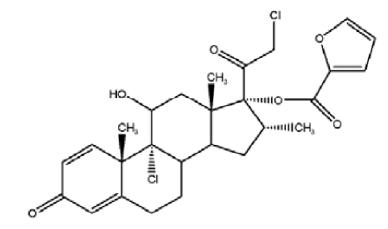
Mometasone furoate is a white to off-white powder, with an molecular formula of C 27H 30C l2O 6, and a molecular weight of 521.43 g/mol. It is practically insoluble in water; slightly soluble in methanol, ethanol, and isopropanol; soluble in acetone and chloroform; and freely soluble in tetrahydrofuran. Its partition coefficient between octanol and water is greater than 5000.
Mometasone furoate nasal spray 50 mcg is a metered-dose, manual pump spray unit containing an aqueous suspension of 0.05% w/w mometasone furoate in an aqueous medium containing benzalkonium chloride, citric acid, glycerin, microcrystalline cellulose and carboxymethylcellulose sodium, polysorbate 80 and sodium citrate. The pH is between 4.3 and 4.9.
-
12. CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Mometasone furoate nasal spray 50 mcg is a corticosteroid demonstrating potent anti-inflammatory properties. The precise mechanism of corticosteroid action on allergic rhinitis is not known. Corticosteroids have been shown to have a wide range of effects on multiple cell types (e.g., mast cells, eosinophils, neutrophils, macrophages, and lymphocytes) and mediators (e.g., histamine, eicosanoids, leukotrienes, and cytokines) involved in inflammation.
In two clinical studies utilizing nasal antigen challenge, mometasone furoate nasal spray, 50 mcg decreased some markers of the early- and late-phase allergic response. These observations included decreases (vs. placebo) in histamine and eosinophil cationic protein levels, and reductions (vs. baseline) in eosinophils, neutrophils, and epithelial cell adhesion proteins. The clinical significance of these findings is not known.
The effect of mometasone furoate nasal spray, 50 mcg on nasal mucosa following 12 months of treatment was examined in 46 patients with allergic rhinitis. There was no evidence of atrophy and there was a marked reduction in intraepithelial eosinophilia and inflammatory cell infiltration (e.g., eosinophils, lymphocytes, monocytes, neutrophils, and plasma cells).
12.2 Pharmacodynamics
Adrenal Function in Adults: Four clinical pharmacology studies have been conducted in humans to assess the effect of mometasone furoate nasal spray, 50 mcg at various doses on adrenal function. In one study, daily doses of 200 and 400 mcg of mometasone furoate nasal spray, 50 mcg and 10 mg of prednisone were compared to placebo in 64 patients (22 to 44 years of age) with allergic rhinitis. Adrenal function before and after 36 consecutive days of treatment was assessed by measuring plasma cortisol levels following a 6-hour Cortrosyn (ACTH) infusion and by measuring 24-hour urinary free cortisol levels. Mometasone furoate nasal spray, 50 mcg, at both the 200- and 400-mcg dose, was not associated with a statistically significant decrease in mean plasma cortisol levels post-Cortrosyn infusion or a statistically significant decrease in the 24-hour urinary free cortisol levels compared to placebo. A statistically significant decrease in the mean plasma cortisol levels post-Cortrosyn infusion and 24-hour urinary free cortisol levels was detected in the prednisone treatment group compared to placebo.
A second study assessed adrenal response to mometasone furoate nasal spray, 50 mcg (400 and 1600 mcg/day), prednisone (10 mg/day), and placebo, administered for 29 days in 48 male volunteers (21 to 40 years of age). The 24-hour plasma cortisol area under the curve (AUC 0–24), during and after an 8-hour Cortrosyn infusion and 24-hour urinary free cortisol levels were determined at baseline and after 29 days of treatment. No statistically significant differences in adrenal function were observed with mometasone furoate nasal spray, 50 mcg compared to placebo.
A third study evaluated single, rising doses of mometasone furoate nasal spray, 50 mcg (1000, 2000, and 4000 mcg/day), orally administered mometasone furoate (2000, 4000, and 8000 mcg/day), orally administered dexamethasone (200, 400, and 800 mcg/day), and placebo (administered at the end of each series of doses) in 24 male volunteers (22 to 39 years of age). Dose administrations were separated by at least 72 hours. Determination of serial plasma cortisol levels at 8 AM and for the 24-hour period following each treatment were used to calculate the plasma cortisol area under the curve (AUC 0–24). In addition, 24-hour urinary free cortisol levels were collected prior to initial treatment administration and during the period immediately following each dose. No statistically significant decreases in the plasma cortisol AUC, 8 AM cortisol levels, or 24-hour urinary free cortisol levels were observed in volunteers treated with either mometasone furoate nasal spray, 50 mcg or oral mometasone, as compared with placebo treatment. Conversely, nearly all volunteers treated with the three doses of dexamethasone demonstrated abnormal 8 AM cortisol levels (defined as a cortisol level <10 mcg/dL), reduced 24-hour plasma AUC values, and decreased 24-hour urinary free cortisol levels, as compared to placebo treatment.
In a fourth study, adrenal function was assessed in 213 patients (18 to 81 years of age) with nasal polyps before and after 4 months of treatment with either mometasone furoate nasal spray, 50 mcg, (200 mcg once or twice daily) or placebo by measuring 24-hour urinary free cortisol levels. Mometasone furoate nasal spray, 50 mcg, at both doses (200 and 400 mcg/day), was not associated with statistically significant decreases in the 24-hour urinary free cortisol levels compared to placebo.
Three clinical pharmacology studies have been conducted in pediatric patients to assess the effect of mometasone furoate nasal spray on the adrenal function at daily doses of 50, 100, and 200 mcg vs. placebo. In one study, adrenal function before and after 7 consecutive days of treatment was assessed in 48 pediatric patients with allergic rhinitis (ages 6 to 11 years) by measuring morning plasma cortisol and 24-hour urinary free cortisol levels. Mometasone furoate nasal spray, at all three doses, was not associated with a statistically significant decrease in mean plasma cortisol levels or a statistically significant decrease in the 24-hour urinary free cortisol levels compared to placebo. In the second study, adrenal function before and after 14 consecutive days of treatment was assessed in 48 pediatric patients (ages 3 to 5 years) with allergic rhinitis by measuring plasma cortisol levels following a 30-minute Cortrosyn infusion. Mometasone furoate nasal spray, 50 mcg, at all three doses (50, 100, and 200 mcg/day), was not associated with a statistically significant decrease in mean plasma cortisol levels post-Cortrosyn infusion compared to placebo. All patients had a normal response to Cortrosyn. In the third study, adrenal function before and after up to 42 consecutive days of once-daily treatment was assessed in 52 patients with allergic rhinitis (ages 2 to 5 years), 28 of whom received mometasone furoate nasal spray, 50 mcg per nostril (total daily dose 100 mcg), by measuring morning plasma cortisol and 24-hour urinary free cortisol levels. Mometasone furoate nasal spray was not associated with a statistically significant decrease in mean plasma cortisol levels or a statistically significant decrease in the 24-hour urinary free cortisol levels compared to placebo.
12.3 Pharmacokinetics
Absorption
Mometasone furoate monohydrate administered as a nasal spray suspension has very low bioavailability (<1%) in plasma using a sensitive assay with a lower quantitation limit (LOQ) of 0.25 pcg/mL.
Distribution
The in vitro protein binding for mometasone furoate was reported to be 98% to 99% in concentration range of 5 to 500 ng/mL.
Metabolism
Studies have shown that any portion of a mometasone furoate dose which is swallowed and absorbed undergoes extensive metabolism to multiple metabolites. There are no major metabolites detectable in plasma. Upon in vitro incubation, one of the minor metabolites formed is 6ß-hydroxy-mometasone furoate. In human liver microsomes, the formation of the metabolite is regulated by cytochrome P-450 3A4 (CYP3A4).
Elimination
Following intravenous administration, the effective plasma elimination half-life of mometasone furoate is 5.8 hours. Any absorbed drug is excreted as metabolites mostly via the bile, and to a limited extent, into the urine.
Specific Populations
Hepatic Impairment: Administration of a single inhaled dose of 400 mcg mometasone furoate to subjects with mild (n=4), moderate (n=4), and severe (n=4) hepatic impairment resulted in only 1 or 2 subjects in each group having detectable peak plasma concentrations of mometasone furoate (ranging from 50 to 105 pcg/mL). The observed peak plasma concentrations appear to increase with severity of hepatic impairment, however, the numbers of detectable levels were few.
Renal Impairment: The effects of renal impairment on mometasone furoate pharmacokinetics have not been adequately investigated.
Pediatric: Mometasone furoate pharmacokinetics have not been investigated in the pediatric population [ see Use in Specific Populations (8.4)].
Drug-Drug Interactions
Inhibitors of Cytochrome P450 3A4: In a drug interaction study, an inhaled dose of mometasone furoate 400 mcg was given to 24 healthy subjects twice daily for 9 days and ketoconazole 200 mg (as well as placebo) were given twice daily concomitantly on Days 4 to 9. Mometasone furoate plasma concentrations were <150 pcg/mL on Day 3 prior to coadministration of ketoconazole or placebo. Following concomitant administration of ketoconazole, 4 out of 12 subjects in the ketoconazole treatment group (n=12) had peak plasma concentrations of mometasone furoate >200 pcg/mL on Day 9 (211–324 pcg/mL).
-
13. NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year carcinogenicity study in Sprague Dawley rats, mometasone furoate demonstrated no statistically significant increase in the incidence of tumors at inhalation doses up to 67 mcg/kg (approximately 1 and 2 times the maximum recommended daily intranasal dose [MRDID] in adults [400 mcg] and children [100 mcg], respectively, on a mcg/m 2 basis). In a 19-month carcinogenicity study in Swiss CD-1 mice, mometasone furoate demonstrated no statistically significant increase in the incidence of tumors at inhalation doses up to 160 mcg/kg (approximately 2 times the MRDID in adults and children, respectively, on a mcg/m 2 basis).
Mometasone furoate increased chromosomal aberrations in an in vitro Chinese hamster ovary-cell assay, but did not increase chromosomal aberrations in an in vitro Chinese hamster lung cell assay. Mometasone furoate was not mutagenic in the Ames test or mouse-lymphoma assay, and was not clastogenic in an in vivo mouse micronucleus assay and a rat bone marrow chromosomal aberration assay or a mouse male germ-cell chromosomal aberration assay. Mometasone furoate also did not induce unscheduled DNA synthesis in vivo in rat hepatocytes.
In reproductive studies in rats, impairment of fertility was not produced by subcutaneous doses up to 15 mcg/kg (less than the MRDID in adults on a mcg/m 2 basis).
13.2 Animal Toxicology and/or Pharmacology
Reproduction Toxicology Studies
In mice, mometasone furoate caused cleft palate at subcutaneous doses of 60 mcg/kg and above (less than the MRDID in adults on a mcg/m 2 basis). Fetal survival was reduced at 180 mcg/kg (approximately 2 times the MRDID in adults on a mcg/m 2 basis). No toxicity was observed at 20 mcg/kg (less than the MRDID in adults on a mcg/m 2 basis).
In rats, mometasone furoate produced umbilical hernia at topical dermal doses of 600 mcg/kg and above (approximately 10 times the MRDID in adults on a mcg/m 2 basis). A dose of 300 mcg/kg (approximately 6 times the MRDID in adults on a mcg/m 2 basis) produced delays in ossification, but no malformations. In rabbits, mometasone furoate caused multiple malformations (e.g., flexed front paws, gallbladder agenesis, umbilical hernia, hydrocephaly) at topical dermal doses of 150 mcg/kg and above (approximately 6 times the MRDID in adults on a mcg/m 2 basis). In an oral study, mometasone furoate increased resorptions and caused cleft palate and/or head malformations (hydrocephaly or domed head) at 700 mcg/kg (approximately 30 times the MRDID in adults on a mcg/m 2 basis). At 2800 mcg/kg (approximately 110 times the MRDID in adults on a mcg/m 2 basis), most litters were aborted or resorbed. No toxicity was observed at 140 mcg/kg (approximately 6 times the MRDID in adults on a mcg/m 2 basis).
When rats received subcutaneous doses of mometasone furoate throughout pregnancy or during the later stages of pregnancy, 15 mcg/kg (less than the MRDID in adults on a mcg/m 2 basis) caused prolonged and difficult labor and reduced the number of live births, birth weight, and early pup survival. Similar effects were not observed at 7.5 mcg/kg (less than the MRDID in adults on a mcg/m 2 basis).
-
14. CLINICAL STUDIES
14.1 Allergic Rhinitis in Adults and Adolescents
The efficacy and safety of mometasone furoate nasal spray, 50 mcg in the prophylaxis and treatment of seasonal allergic rhinitis and the treatment of perennial allergic rhinitis have been evaluated in 18 controlled trials, and one uncontrolled clinical trial, in approximately 3000 adults (ages 17 to 85 years) and adolescents (ages 12 to 16 years). Of the total number of patients, there were 1757 males and 1453 females, including a total of 283 adolescents (182 boys and 101 girls) with seasonal allergic or perennial allergic rhinitis. Patients were treated with mometasone furoate nasal spray 50 mcg at doses ranging from 50 to 800 mcg/day. The majority of patients were treated with 200 mcg/day. The allergic rhinitis trials evaluated the total nasal symptom scores that included stuffiness, rhinorrhea, itching, and sneezing. Patients treated with mometasone furoate nasal spray 50 mcg, 200 mcg/day had a statistically significant decrease in total nasal symptom scores compared to placebo-treated patients. No additional benefit was observed for mometasone furoate doses greater than 200 mcg/day. A total of 350 patients have been treated with mometasone furoate nasal spray 50 mcg for 1 year or longer.
In patients with seasonal allergic rhinitis, mometasone furoate nasal spray 50 mcg, demonstrated improvement in nasal symptoms (vs. placebo) within 11 hours after the first dose based on one single-dose, parallel-group study of patients in an outdoor "park" setting (park study) and one environmental exposure unit (EEU) study, and within 2 days in two randomized, double-blind, placebo-controlled, parallel-group seasonal allergic rhinitis studies. Maximum benefit is usually achieved within 1 to 2 weeks after initiation of dosing.
Prophylaxis of seasonal allergic rhinitis for patients 12 years of age and older with mometasone furoate nasal spray 50 mcg, given at a dose of 200 mcg/day, was evaluated in two clinical studies in 284 patients. These studies were designed such that patients received 4 weeks of prophylaxis with mometasone furoate nasal spray 50 mcg prior to the anticipated onset of the pollen season; however, some patients received only 2 to 3 weeks of prophylaxis. Patients receiving 2 to 4 weeks of prophylaxis with mometasone furoate nasal spray 50 mcg demonstrated a statistically significantly smaller mean increase in total nasal symptom scores with onset of the pollen season as compared to placebo patients.
14.2 Allergic Rhinitis in Pediatrics
The efficacy and safety of mometasone furoate nasal spray 50 mcg in the treatment of seasonal allergic and perennial allergic rhinitis in pediatric patients (ages 3 to 11 years) have been evaluated in four controlled trials. This included approximately 990 pediatric patients ages 3 to 11 years (606 males and 384 females) with seasonal allergic or perennial allergic rhinitis treated with mometasone furoate nasal spray at doses ranging from 25 to 200 mcg/day. Pediatric patients treated with mometasone furoate nasal spray 50 mcg (100 mcg total daily dose, 374 patients) had a significant decrease in total nasal symptom (nasal congestion, rhinorrhea, itching, and sneezing) scores, compared to placebo-treated patients. No additional benefit was observed for the 200-mcg mometasone furoate total daily dose in pediatric patients (ages 3 to 11 years). A total of 163 pediatric patients have been treated for 1 year.
14.3 Nasal Polyps in Adults 18 Years of Age and Older
Two studies were performed to evaluate the efficacy and safety of mometasone furoate nasal spray in the treatment of nasal polyps. These studies involved 664 patients with nasal polyps, 441 of whom received mometasone furoate nasal spray. These studies were randomized, double-blind, placebo-controlled, parallel-group, multicenter studies in patients 18 to 86 years of age with bilateral nasal polyps. Patients were randomized to receive mometasone furoate nasal spray 200 mcg once daily, 200 mcg twice daily or placebo for a period of 4 months. The co-primary efficacy endpoints were 1) change from baseline in nasal congestion/obstruction averaged over the first month of treatment; and 2) change from baseline to last assessment in bilateral polyp grade during the entire 4 months of treatment as assessed by endoscopy. Efficacy was demonstrated in both studies at a dose of 200 mcg twice daily and in one study at a dose of 200 mcg once a day (see TABLE 2 below).
TABLE 2: EFFECT OF MOMETASONE FUROATE NASAL SPRAY IN TWO RANDOMIZED, PLACEBO-CONTROLLED TRIALS IN PATIENTS WITH NASAL POLYPS Mometasone Furoate Nasal Spray 200
mcg qdMometasone Furoate Nasal Spray 200
mcg bidPlacebo P value for Mometasone Furoate Nasal Spray 200 mcg qd vs. placebo P value for Mometasone Furoate Nasal Spray 200 mcg bid vs. placebo - * polyps in each nasal fossa were graded by the investigator based on endoscopic visualization, using a scale of 0–3 where 0 = no polyps; 1 = polyps in the middle meatus, not reaching below the inferior border of the middle turbinate; 2 = polyps reaching below the inferior border of the middle turbinate but not the inferior border of the inferior turbinate; 3 = polyps reaching to or below the border of the inferior turbinate, or polyps medial to the middle turbinate (score reflects sum of left and right nasal fossa grades).
- † nasal congestion/obstruction was scored daily by the patient using a 0–3 categorical scale where 0 = no symptoms, 1 = mild symptoms, 2 = moderate symptoms and 3 = severe symptoms.
Study 1 N=115 N=122 N=117 Baseline bilateral polyp grade * 4.21 4.27 4.25 Mean change from baseline in bilateral polyps grade -1.15 -0.96 -0.50 <0.001 0.01 Baseline nasal congestion † 2.29 2.35 2.28 Mean change from baseline in nasal congestion -0.47 -0.61 -0.24 0.001 <0.001 Study 2 N=102 N=102 N=106 Baseline bilateral polyp grade * 4.00 4.10 4.17 Mean change from baseline in bilateral polyps grade -0.78 -0.96 -0.62 0.33 0.04 Baseline nasal congestion † 2.23 2.20 2.18 Mean change from baseline in nasal congestion -0.42 -0.66 -0.23 0.01 <0.001 There were no clinically relevant differences in the effectiveness of mometasone furoate nasal spray, 50 mcg, in the studies evaluating treatment of nasal polyps across subgroups of patients defined by gender, age, or race.
14.4 Nasal Congestion Associated with Seasonal Allergic Rhinitis
The efficacy and safety of mometasone furoate nasal spray 50 mcg for nasal congestion associated with seasonal allergic rhinitis were evaluated in three randomized, placebo-controlled, double blind clinical trials of 15 days duration. The three trials included a total of 1008 patients 12 years of age and older with nasal congestion associated with seasonal allergic rhinitis, of whom 506 received mometasone furoate nasal spray 200 mcg daily and 502 received placebo. Of the 1008 patients, the majority 784 (78%) were Caucasians. The majority of the patients were between 18 to < 65 years of age with a mean age of 38.8 years and were predominantly women (66%). The primary efficacy endpoint was the change from baseline in average morning and evening reflective nasal congestion score over treatment day 1 to day 15. The key secondary efficacy endpoint was the change from baseline in average morning and evening reflective total nasal symptom score (TNSS = rhinorrhea [nasal discharge/runny nose or postnasal drip], nasal congestion/stuffiness, nasal itching, sneezing) averaged over treatment day 1 to 15. Two out of three studies demonstrated that treatment with mometasone furoate nasal spray significantly reduced the nasal congestion symptom score and the TNSS compared to placebo in patients 12 years of age and older with seasonal allergic rhinitis (see TABLE 3 and 4 below).
TABLE 3: EFFECT OF MOMETASONE FUROATE NASAL SPRAY IN TWO RANDOMIZED, PLACEBO-CONTROLLED TRIALS ON NASAL CONGESTION IN PATIENTS WITH SEASONAL ALLERGIC RHINITIS Treatment (Patient Number) Baseline *
LS Mean †Change from Baseline
LS Mean †Difference from
Placebo
LS Mean †P value for
Mometasone Furoate Nasal Spray 200
mcg qd vs. placebo- * nasal congestion/obstruction was scored daily by the patient using a 0–3 categorical scale where 0 = no symptoms, 1 = mild symptoms, 2 = moderate symptoms and 3 = severe symptoms.
- † LS Mean and p- value was from an ANCOVA model with treatment, baseline value, and center effects.
Study 1
Mometasone Furoate Nasal Spray 200 mcg qd (N=176)
2.63
-0.64
-0.15
0.006
Placebo (N=175)
2.62
-0.49
Study 2
Mometasone Furoate Nasal Spray 200 mcg qd (N=168)
2.62
-0.71
-0.31
<0.001
Placebo (N=164)
2.60
-0.40
TABLE 4: EFFECT OF MOMETASONE FUROATE NASAL SPRAY ON TNSS IN TWO RANDOMIZED, PLACEBO-CONTROLLED TRIALS IN PATIENTS WITH SEASONAL ALLERGIC RHINITIS Treatment (Patient Number) Baseline *
LS Mean †Change from Baseline
LS Mean †Difference from
Placebo
LS Mean †P value for
Mometasone Furoate Nasal Spray 200
mcg qd vs. placebo- * TNSS was the sum of four individual symptom scores: rhinorrhea, nasal congestion/stuffiness, nasal itching and sneezing. Each symptom was to be rated on a scale of 0 = none, 1 = mild, 2 = moderate, 3 = severe.
- † LS Mean and p- value was from an ANCOVA model with treatment, baseline value, and center effects.
Study 1
Mometasone Furoate Nasal Spray 200 mcg qd (N=176)
9.60
-2.68
-0.83
<0.001
Placebo (N=175)
9.66
-1.85
Study 2
Mometasone Furoate Nasal Spray 200 mcg qd (N=168)
9.39
-3.00
-1.27
<0.001
Placebo (N=164)
9.50
-1.73
Based on results in other studies with mometasone furoate nasal spray in pediatric patients, effects on nasal congestion associated with seasonal allergic rhinitis in patients below 12 years of age is similar to those seen in adults and adolescents [ see Clinical Studies (14.2)].
-
16. HOW SUPPLIED/STORAGE AND HANDLING
Mometasone Furoate Nasal Spray, 50 mcg is supplied in a white, high-density, polyethylene bottle fitted with a white metered-dose, manual spray pump, and translucent cap. It contains 17 g of product formulation, 120 sprays, each delivering 50 mcg of mometasone furoate per actuation.
(NDC: 60429-664-17).
Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Protect from light.
When mometasone furoate nasal spray, 50 mcg is removed from its cardboard container, prolonged exposure of the product to direct light should be avoided. Brief exposure to light, as with normal use, is acceptable.
SHAKE WELL BEFORE EACH USE.
Keep out of reach of children.
-
17. PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
17.1 Local Nasal Effect
Patients should be informed that treatment with mometasone furoate nasal spray 50 mcg may be associated with adverse reactions which include epistaxis (nose bleed) and nasal septum perforation. Candida infection may also occur. Because of the inhibitory effect of corticosteroids on wound healing, patients who have experienced recent nasal septum ulcers, nasal surgery, or nasal trauma should not use a nasal corticosteroid until healing has occurred [ see Warnings and Precautions (5.1)]. Patients should be cautioned not to spray mometasone furoate nasal spray 50 mcg directly onto the nasal septum.
17.2 Glaucoma and Cataracts
Advise patients that long-term use of nasal and inhaled corticosteroids may increase the risk of some eye problems (glaucoma or cataracts); regular eye examinations should be considered. Patients should be cautioned not to spray mometasone furoate nasal spray 50 mcg into the eyes [ see Warnings and Precautions ( 5.2) ].
17.3 Immunosuppression
Persons who are on immunosuppressant doses of corticosteroids should be warned to avoid exposure to chickenpox or measles, and patients should also be advised that if they are exposed, medical advice should be sought without delay [ see Warnings and Precautions (5.4)].
17.4 Use Regularly for Best Effect
Patients should use mometasone furoate nasal spray 50 mcg on a regular basis for optimal effect. Improvement in nasal symptoms of allergic rhinitis has been shown to occur within 1 to 2 days after initiation of dosing. Maximum benefit is usually achieved within 1 to 2 weeks after initiation of dosing. Patients should not increase the prescribed dosage but should contact their physician if symptoms do not improve, or if the condition worsens. Administration to young children should be aided by an adult.
If a patient missed a dose, the patient should be advised to take the dose as soon as they remember. The patient should not take more than the recommended dose for the day.
- SPL UNCLASSIFIED SECTION
-
PATIENT PACKAGE INSERT
PATIENT INFORMATION
Mometasone Furoate Nasal Spray, 50 mcg
(moe met' a sone fure' oh ate)
For Intranasal Use Only
Read the Patient Information that comes with Mometasone Furoate Nasal Spray before you start using it and each time you get a refill. There may be new information. This Patient Information does not take the place of talking to your healthcare provider about your medical condition or treatment. If you have any questions about Mometasone Furoate Nasal Spray, ask your healthcare provider.
What is Mometasone Furoate Nasal Spray?
Mometasone Furoate Nasal Spray is a man-made (synthetic) corticosteroid medicine that is used to:
- treat the nasal symptoms of seasonal and year-round allergic rhinitis (inflammation of the lining of the nose) in adults and children 2 years of age and older.
- treat nasal congestion that happens with seasonal allergic rhinitis in adults and children 2 years of age and older.
- prevent nasal symptoms of seasonal allergic rhinitis in people 12 years of age and older.
- treat nasal polyps in people 18 years and older.
- It is not known if Mometasone Furoate Nasal Spray is safe and effective in children under:
- 2 years of age to treat allergic rhinitis.
- 18 years of age to treat nasal polyps.
Who should not use Mometasone Furoate Nasal Spray?
Do not use Mometasone Furoate Nasal Spray if you are allergic to mometasone furoate or any of the ingredients in Mometasone Furoate Nasal Spray. See the end of this leaflet for a complete list of ingredients in Mometasone Furoate Nasal Spray.
What should I tell my healthcare provider before and during treatment with Mometasone Furoate Nasal Spray?
Before you take Mometasone Furoate Nasal Spray, tell your healthcare provider about all of your medical conditions, including if you:
- have had recent nasal sores, nasal surgery, or nasal injury.
- have eye or vision problems, such as cataracts, glaucoma (increased pressure in your eye), and blurred vision, or other changes in your vision.
- have tuberculosis or any untreated fungal, bacterial, viral infections, or eye infections caused by herpes.
- have been near someone who has chickenpox or measles.
- are not feeling well or have any other symptoms that you do not understand.
- are pregnant or planning to become pregnant. It is not known if Mometasone Furoate Nasal Spray will harm your unborn baby. Talk to your healthcare provider if you are pregnant or plan to become pregnant.
- are breastfeeding or planning to breastfeed. It is not known whether Mometasone Furoate Nasal Spray passes into your breast milk.
Tell your healthcare provider about all the medicines you take including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Especially tell your healthcare provider if you take:
- certain medicines for HIV (such as ritonavir, atazanavir, indinavir, nelfinavir, and saquinavir)
- cobicistat-containing products
- certain antifungals (such as ketoconazole and itraconazole)
- certain antibiotics (such as clarithromycin and telithromycin)
- certain antidepressants (such as nefazodone)
If you take these medicines with Mometasone Furoate Nasal Spray, your healthcare provider should monitor you for side effects.
Mometasone Furoate Nasal Spray may affect the way other medicines work, and other medicines may affect how Mometasone Furoate Nasal Spray works.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I use Mometasone Furoate Nasal Spray?
- Use Mometasone Furoate Nasal Spray exactly as prescribed by your healthcare provider.
- This medicine is for use in the nose only. Do not spray it into your mouth or eyes.
- An adult should help a young child use this medicine.
- For best results, you should keep using Mometasone Furoate Nasal Spray regularly each day without missing a dose. If you do miss a dose of Mometasone Furoate Nasal Spray, take it as soon as you remember. However, do not take more than the daily dose prescribed by your healthcare provider.
- Do not use Mometasone Furoate Nasal Spray more often than prescribed. Ask your healthcare provider if you have any questions.
- For detailed instructions on how to use Mometasone Furoate Nasal Spray, see the " Patient Instructions for Use" at the end of this leaflet.
- See your healthcare provider regularly to check your symptoms while taking Mometasone Furoate Nasal Spray and to check for side effects.
What should I avoid while taking Mometasone Furoate Nasal Spray?
If you are taking other corticosteroid medicines for allergy, by mouth or injection, your healthcare provider may advise you to stop taking them after you begin using Mometasone Furoate Nasal Spray.
What are the possible side effects of Mometasone Furoate Nasal Spray?
Mometasone Furoate Nasal Spray may cause serious side effects, including:
- thrush (candida), a fungal infection in your nose and throat. Tell your healthcare provider if you have any redness or white colored patches in your nose or throat.
- hole in the cartilage of the nose (nasal septal perforation). A whistling sound when you breathe may be a symptom of nasal septum perforation.
- slow wound healing. Do not use Mometasone Furoate Nasal Spray until your nose has healed if you have a sore in your nose, if you have surgery on your nose, or if your nose has been injured.
- eye problems, including glaucoma, cataracts, and blurred vision. You should have regular eye exams.
- allergic reactions. Allergic reactions including wheezing may happen after using Mometasone Furoate Nasal Spray. If wheezing happens stop using Mometasone Furoate Nasal Spray. Tell your healthcare provider or get medical help right away .
- immune system problems that may increase your risk of infections. You are more likely to get infections if you take medicines that weaken your immune system. Avoid contact with people who have contagious diseases such as chicken pox or measles while using Mometasone Furoate Nasal Spray. Symptoms of infection may include: fever, pain, aches, chills, feeling tired, nausea, and vomiting. Tell your healthcare provider about any signs of infection while you are using Mometasone Furoate Nasal Spray.
- adrenal insufficiency. Adrenal insufficiency is a condition in which the adrenal glands do not make enough steroid hormones. Symptoms of adrenal insufficiency can include: tiredness, weakness, nausea and vomiting and low blood pressure.
- slowed growth in children. Your child’s growth should be checked regularly while using Mometasone Furoate Nasal Spray.
The most common side effects of Mometasone Furoate Nasal Spray include:
- headache
- viral infection
- sore throat
- nosebleeds
- cough
These are not all the possible side effects of Mometasone Furoate Nasal Spray. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store Mometasone Furoate Nasal Spray?
- Store Mometasone Furoate Nasal Spray at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature]. Protect from light.
- Avoid prolonged exposure of Mometasone Furoate Nasal Spray container to direct light.
- Shake well before each use.
Keep Mometasone Furoate Nasal Spray and all medicines out of the reach of children.
General information about the safe and effective use of Mometasone Furoate Nasal Spray
Medicines are sometimes prescribed for conditions that are not listed in a Patient Information leaflet. Do not use Mometasone Furoate Nasal Spray for a condition for which it was not prescribed. Do not give Mometasone Furoate Nasal Spray to other people even if they have the same symptoms you have. It may harm them.
You can ask your healthcare provider or pharmacist for information about Mometasone Furoate Nasal Spray that is written for health professionals.
For more information, go to www.apotexcorp.com/products or call 1-800-706-5575.
What are the ingredients in Mometasone Furoate Nasal Spray?
Active ingredients: mometasone furoate
Inactive ingredients: benzalkonium chloride, citric acid, glycerin, microcrystalline cellulose and carboxymethylcellulose sodium, polysorbate 80 and sodium citrate.
This Patient Information has been approved by the U.S. Food and Drug Administration
APOTEX INC.
MOMETASONE FUROATE NASAL SPRAY
50 mcg
Manufactured by: Manufactured for: Apotex Inc.
Toronto, Ontario
M9L 1T9Apotex Corp.
Weston, FL
33326Marketed by:
GSMS, Inc.
Camarillo, CA USA 93012
Revised: July 2018
Patient Instructions for Use
Mometasone Furoate Nasal Spray, 50 mcg
(moe met' a sone fure' oh ate)
For use in your nose only.
Read the Patient Instructions for Use carefully before you start to use your Mometasone Furoate Nasal Spray. If you have any questions, ask your healthcare provider.
Shake the bottle well before each use.
- Remove the plastic cap ( See Figure 1).
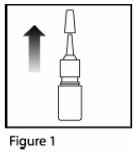
- Before you use Mometasone Furoate Nasal Spray for the first time, prime the pump by pressing downward on the shoulders of the white nasal applicator using your index finger and middle finger while holding the base of the bottle with your thumb ( See Figure 2). Do Not pierce the nasal applicator. Press down and release the pump 10 times or until a fine spray appears. Do Not spray into eyes. The pump is now ready to use. The pump may be stored unused for up to 1 week without repriming. If unused for more than 1 week, reprime by spraying 2 times or until a fine spray appears.
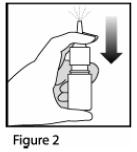
- Gently blow your nose to clear the nostrils. Close 1 nostril. Tilt your head forward slightly, keep the bottle upright, carefully insert the nasal applicator into the other nostril ( See Figure 3). Do Not spray directly onto the nasal septum (the wall between the two nostrils).
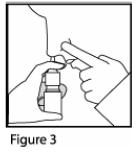
- For each spray, hold the spray bottle upright and press firmly downward 1 time on the shoulders of the white nasal applicator using your index and middle fingers while supporting the base of the bottle with your thumb. Breathe gently inward through the nostril (
See Figure 4).
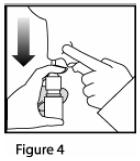
- Note: It is important to keep the Mometasone Furoate Nasal Spray unit in an upright orientation (as seen in Figure 4). Failure to do so may result in an incomplete or non-existent spray.
- Then breathe out through the mouth.
- Repeat in the other nostril.
- Wipe the nasal applicator with a clean tissue and replace the plastic cap.
Each bottle of Mometasone Furoate Nasal Spray contains enough medicine for you to spray medicine from the bottle 120 times. Do not use the bottle of Mometasone Furoate Nasal Spray after 120 sprays. Additional sprays after the 120 sprays may not contain the right amount of medicine, you should keep track of the number of sprays used from each bottle of Mometasone Furoate Nasal Spray, and throw away the bottle even if it has medicine still left in. Do not count any sprays used for priming the device. Talk with your healthcare provider before your supply runs out to see if you should get a refill of your medicine.
Pediatric Use: Administration to young children should be done by an adult. Steps 1 through 7 from the Patient Instructions for Use, should be followed.
Cleaning: Do not try to unblock the nasal applicator with a sharp object. Please see Patient Instructions for Cleaning Applicator.
Patient Instructions for Cleaning Applicator
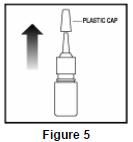
- To clean the nasal applicator, remove the plastic cap ( See Figure 5).
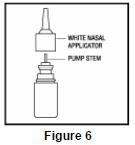
- Pull gently upward on the white nasal applicator to remove ( See Figure 6).
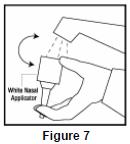
- Soak the nasal applicator in cold tap water and rinse both ends of the nasal applicator under cold tap water and dry ( See Figure 7). Do not try to unblock the nasal applicator by inserting a pin or other sharp object as this will damage the applicator and cause you not to get the right dose of medicine.
- Rinse the plastic cap under cold water and dry.
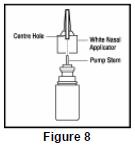
- Put the nasal applicator back together making sure the pump stem is reinserted into the applicator's center hole ( See Figure 8).
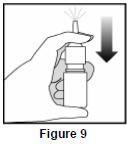
- Reprime the pump by pressing downward on the shoulders of the white nasal applicator using your index and middle fingers while holding the base of the bottle with your thumb. Press down and release the pump 2 times or until a fine spray appears. Do Not spray into eyes. The pump is now ready to use. The pump may be stored unused for up to 1 week without repriming. If unused for more than 1 week, reprime by spraying 2 times or until a fine spray appears ( See Figure 9).
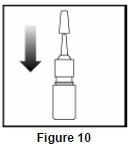
- Replace the plastic cap ( See Figure 10).
This Patient Information and Instructions for Use has been approved by the U.S. Food and Drug Administration.
APOTEX INC.
MOMETASONE FUROATE NASAL SPRAY
50 mcg
Manufactured by: Manufactured for: Apotex Inc. Apotex Corp. Toronto, Ontario Weston, FL M9L 1T9 33326 Marketed by:
GSMS, Inc.Camarillo, CA USA 93012
Revised: July 2018
-
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - Bottle Label
NDC: 60429-664-17
Net Contents: 17 g
120 Metered Sprays
For Intranasal Use Only
Rx Only

-
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - Carton Label
NDC: 60429-664-17
Net Contents: 17 g
120 Metered Sprays
For Intranasal Use Only
Rx Only
-
INGREDIENTS AND APPEARANCE
MOMETASONE FUROATE
mometasone furoate spray, meteredProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60429-664(NDC:60505-0830) Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MOMETASONE (UNII: 8HR4QJ6DW8) (MOMETASONE - UNII:8HR4QJ6DW8) MOMETASONE 50 ug Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM (UNII: K679OBS311) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) POLYSORBATE 80 (UNII: 6OZP39ZG8H) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60429-664-17 1 in 1 CARTON 03/29/2016 1 120 in 1 BOTTLE, PUMP; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA091161 03/22/2016 Labeler - Golden State Medical Supply, Inc. (603184490) Establishment Name Address ID/FEI Business Operations Golden State Medical Supply, Inc. 603184490 relabel(60429-664)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.