SK Dry Eye Relief by Synpac-Kingdom Pharmaceutical, Co., Ltd. / Synpac-Kingdom SK Dry Eye Relief
SK Dry Eye Relief by
Drug Labeling and Warnings
SK Dry Eye Relief by is a Otc medication manufactured, distributed, or labeled by Synpac-Kingdom Pharmaceutical, Co., Ltd., Synpac-Kingdom. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
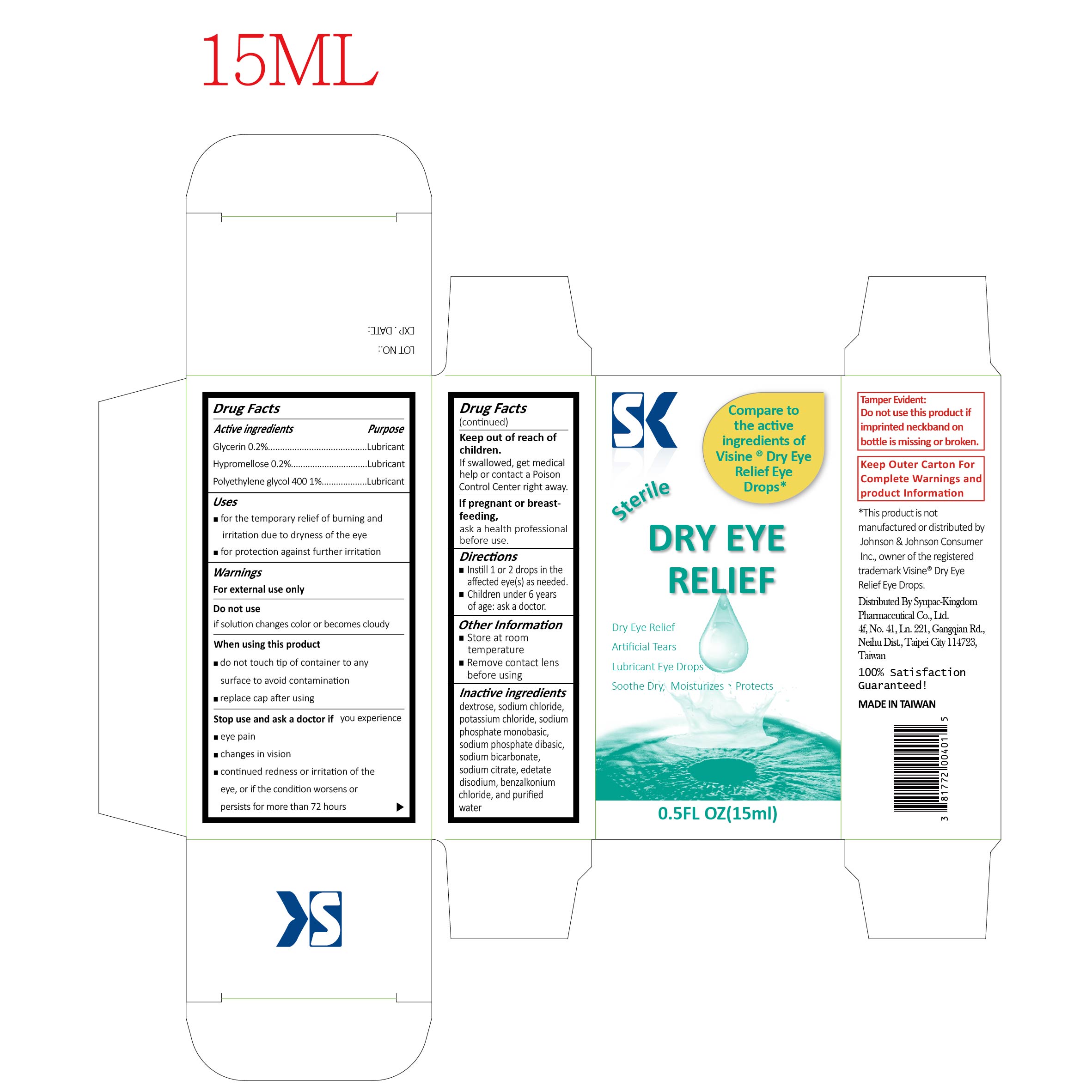
SK DRY EYE RELIEF- glycerin, hypromellose, polypropylene glycol 400 solution
Synpac-Kingdom Pharmaceutical, Co., Ltd.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
SK Dry Eye Relief
Purpose
Glycerin..........................Lubricant
Hypromellose..................Lubricant
Polyethylene glycol 400....Lubricant
Uses
- for the temporary relief of burning and irritation due to dryness of the eye
- for protection against further irritation
Warnings
For external use only
Stop use and ask a doctor if you experience
- eye pain
- changes in vision
- continued redness or irritation of the eye or if the condition worsens or persists for more than 72 hours
When using this product
- do not touch tip of container to any surface to avoid contamination
- replace cap after using
Directions
- Instill 1 or 2 drops in the affected eye(s) as needed.
- Children under 6 years of age: ask a doctor.
| SK DRY EYE RELIEF
glycerin, hypromellose, polypropylene glycol 400 solution |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - Synpac-Kingdom Pharmaceutical, Co., Ltd. (657636332) |
| Registrant - Synpac-Kingdom (657636332) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Synpac-Kingdom | 657636332 | manufacture(81772-004) , pack(81772-004) , label(81772-004) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.