NIXIFLOR- florfenicol and flunixin meglumine injection, solution
nixiFLOR by
Drug Labeling and Warnings
nixiFLOR by is a Animal medication manufactured, distributed, or labeled by Parnell Technologies Pty Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- WARNINGS
-
DESCRIPTION:
nixiFLOR™ is an injectable solution of the synthetic antibiotic florfenicol and the non-steroidal anti-inflammatory drug (NSAID) flunixin. Each milliliter of sterile nixiFLOR™ contains 300 mg florfenicol, 16.5 mg flunixin as flunixin meglumine, 300 mg 2-pyrrolidone, 35 mg malic acid, and triacetin qs.
- INDICATION:
-
DOSAGE AND ADMINISTRATION:
nixiFLOR™ should be administered once by subcutaneous injection at a dose rate of 40 mg florfenicol/kg body weight and 2.2 mg flunixin/kg body weight (6 mL/100 lb). Do not administer more than 10 mL at each site. The injection should be given only in the neck. Injection sites other than the neck have not been evaluated. For the 500 mL vial, do not puncture the stopper more than 20 times.
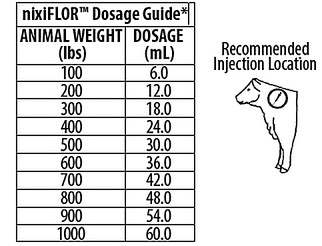
* Do not administer more than 10 mL at each site.
- CONTRAINDICATIONS:
-
WARNINGS:
WARNINGS: NOT FOR HUMAN USE. KEEP OUT OF REACH OF CHILDREN.
This product contains material that can be irritating to skin and eyes. Avoid direct contact with skin, eyes, and clothing. In case of accidental eye exposure, flush with water for 15 minutes. In case of accidental skin exposure, wash with soap and water. Remove contaminated clothing. Consult a physician if irritation persists. Accidental injection of this product may cause local irritation. Consult a physician immediately. The Safety Data Sheet (SDS) contains more detailed occupational safety information. For customer service, adverse effects reporting, and/or a copy of the SDS, contact Parnell at 1-800-887-2763. For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or http://www.fda.gov/reportanimalae.
-
PRECAUTIONS:
As a class, cyclo-oxygenase inhibitory NSAIDs may be associated with gastrointestinal, renal, and hepatic toxicity. Sensitivity to drug-associated adverse events varies with the individual patient. Patients at greatest risk for adverse events are those that are dehydrated, on diuretic therapy, or those with existing renal, cardiovascular, and/or hepatic dysfunction. Concurrent administration of potentially nephrotoxic drugs should be carefully monitored. NSAIDs may inhibit the prostaglandins that maintain normal homeostatic function. Such anti-prostaglandin effects may result in clinically significant disease in patients with underlying or pre-existing disease that have not been previously diagnosed. Since many NSAIDs possess the potential to produce gastrointestinal ulceration, concomitant use of florfenicol and flunixin meglumine with other anti-inflammatory drugs, such as NSAIDs or corticosteroids, should be avoided or closely monitored.
Flunixin is a cyclo-oxygenase inhibitory NSAID, and as with others in this class, adverse effects may occur with its use. The most frequently reported adverse effects have been gastrointestinal signs. Events involving suspected renal, hematologic, neurologic, dermatologic, and hepatic effects have also been reported for other drugs in this class.
Not for use in animals intended for breeding purposes. The effects of florfenicol and flunixin meglumine on bovine reproductive performance, pregnancy, and lactation have not been determined. Toxicity studies in dogs, rats, and mice have associated the use of florfenicol with testicular degeneration and atrophy. NSAIDs are known to have potential effects on both parturition and the estrous cycle. There may be a delay in the onset of estrus if flunixin is administered during the prostaglandin phase of the estrous cycle. Studies have associated the use of flunixin in cattle with a delay in parturition and prolonged labor (which may increase the risk of stillbirth), and interference with involution and expulsion of fetal membranes (which may increase the risk for placental retention and metritis). Florfenicol and flunixin meglumine, when administered as directed, may induce a transient reaction at the site of injection and underlying tissues that may result in trim loss of edible tissue at slaughter.

-
ADVERSE REACTIONS:
Transient inappetence, diarrhea, decreased water consumption, and injection site swelling have been associated with the use of florfenicol in cattle. In addition, anaphylaxis and collapse have been reported post-approval with the use of another formulation of florfenicol in cattle.
In cattle, rare instances of anaphylactic-like reactions, some of which have been fatal, have been reported, primarily following intravenous use of flunixin meglumine. - CLINICAL PHARMACOLOGY:
-
MICROBIOLOGY:
Florfenicol is a synthetic, broad-spectrum antibiotic active against many Gram-negative and Gram-positive bacteria isolated from domestic animals. It acts by binding to the 50S ribosomal subunit and inhibiting bacterial protein synthesis. Florfenicol is generally considered a bacteriostatic drug, but exhibits bactericidal activity against certain bacterial species.
In vitro studies demonstrate that florfenicol is active against the BRD pathogens M. haemolytica, P. multocida, and H. somni, and M. bovis, and that florfenicol exhibits bactericidal activity against strains of M. haemolytica and H. somni.
The minimum inhibitory concentrations (MICs) of florfenicol were determined for non-mycoplasmal BRD isolates obtained from calves enrolled in BRD field studies in the U.S. in 2006 using methods recommended by the Clinical and Laboratory Standards Institute (M31-A2).
MICs for M. bovis isolates were determined by an accepted method using Hayflick Broth with Alamar Blue (HBAN) medium under appropriate control. Isolates were obtained from pre-treatment nasal swabs from all calves enrolled at all four sites, post-treatment nasal swabs from treatment failures in the florfenicol and flunixin meglumine and saline control treatment groups at three sites, and lung tissue from one calf that died in the saline control treatment group.
The results are shown in Table 3 below.
Table 3. Florfenicol MIC values* of indicated pathogens isolated from cattle with naturally-occurring BRD.
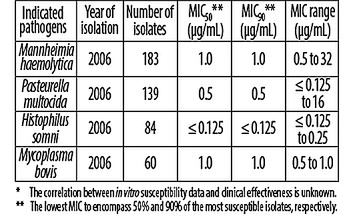
-
EFFECTIVENESS:
In a multi-site field study, calves with naturally-occurring BRD were treated with florfenicol and flunixin meglumine, florfenicol (NADA 141-265), or saline. A treatment success was defined as a calf with normal respiration to mild respiratory distress, normal attitude to mildly depressed, and a rectal temperature < 104.0 °F on Day 11.
The treatment success rate for BRD for the florfenicol and flunixin meglumine treatment group (68.4%) was statistically significantly greater (p = 0.0255) compared to the saline control treatment group (42.9%). Florfenicol and flunixin meglumine was non-inferior to florfenicol for the treatment of BRD, with a one-sided 95% lower confidence bound for the difference between the two treatments equal to -13.2%.
In the same study, the change in rectal temperature from pre-treatment to six hours post-treatment was evaluated to determine the effectiveness of florfenicol and flunixin meglumine for the control of BRD-associated pyrexia. The proportion of calves whose rectal temperatures decreased by ≥ 2.0 °F from pre-treatment to six hours post-treatment was statistically significantly greater (p = 0.0019) in the florfenicol and flunixin meglumine treatment group compared to the saline control treatment group.
The mean decrease in rectal temperature from pre-treatment to six hours post-treatment was statistically significantly greater in the florfenicol and flunixin meglumine treatment group compared to the florfenicol and saline control treatment groups (p = 0.0031 and 0.0002, respectively).
The effectiveness of florfenicol and flunixin meglumine for the treatment of BRD associated with Mycoplasma bovis was demonstrated by examining the M. bovis data from cattle enrolled in the BRD treatment study described above. There were numerically more treatment successes (6 of 8 calves, 75%) than treatment failures (2 of 8 calves, 25%) in florfenicol and flunixin meglumine-treated calves that cultured positive for M. bovis pre-treatment.
-
ANIMAL SAFETY:
A target animal safety study was conducted to evaluate the effects of florfenicol and flunixin meglumine when administered to cattle subcutaneously at 1X, 3X, or 5X the labeled dose for three consecutive days (3X the labeled duration). Decreased feed and water consumption, and decreased body weights (secondary to decreased feed consumption) were observed in the 1X, 3X, and 5X groups. Injection site swellings were noted in the 1X, 3X, and 5X groups.
A separate injection site study was conducted in cattle. The study demonstrated that florfenicol and flunixin meglumine, when administered according to the label directions, may induce a transient local reaction in the subcutaneous and underlying muscle tissue.
- STORAGE INFORMATION:
-
HOW SUPPLIED:
nixiFLOR™ is available in 100, 250, and 500 mL sterile, multiple-dose, glass vials.
Approved by FDA under ANADA # 200-828
Manufactured by:
PARNELL TECHNOLOGIES PTY. LTD.
4/476 Gardeners Road, Alexandria
NSW 2015, AustraliaDistributed by:
PARNELL U.S. 1, Inc.
7015 College Boulevard, Level 6
Overland Park, KS 66211Owner of the registered trademark nixiFLOR™.

50449-01-Oct25
-
Principal Display Panel – 250 mL Carton
Sterile
250 mL
Multiple Dose Vial
300 mg/16.5 mg/mLnixiFLOR™
(florfenicol and flunixin meglumine)Antimicrobial/Non-Steroidal Anti-Inflammatory Drug
For subcutaneous use in beef and non-lactating dairy cattle only.
Not for use in female dairy cattle 20 months of age or older or in calves to be processed for veal.
CAUTION: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Approved by FDA under ANADA # 200-828

-
INGREDIENTS AND APPEARANCE
NIXIFLOR
florfenicol and flunixin meglumine injection, solutionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC: 68504-009 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Florfenicol (UNII: 9J97307Y1H) (FLORFENICOL - UNII:9J97307Y1H) Florfenicol 300 mg in 1 mL FLUNIXIN MEGLUMINE (UNII: 8Y3JK0JW3U) (FLUNIXIN - UNII:356IB1O400) FLUNIXIN 16.5 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68504-009-01 1 in 1 CARTON 1 100 mL in 1 VIAL, MULTI-DOSE 2 NDC: 68504-009-02 1 in 1 CARTON 2 250 mL in 1 VIAL, MULTI-DOSE 3 NDC: 68504-009-03 1 in 1 CARTON 3 500 mL in 1 VIAL, MULTI-DOSE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANADA ANADA200828 01/09/2026 Labeler - Parnell Technologies Pty Ltd (742511504)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
