TOFACITINIB tablet, extended release
Drug Labeling and Warnings
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use TOFACITINIB EXTENDED-RELEASE TABLETS safely and effectively. See full prescribing information for TOFACITINIB EXTENDED-RELEASE TABLETS.
TOFACITINIB extended-release tablets, for oral use
Initial U.S. Approval: 2012WARNING: SERIOUS INFECTIONS, MORTALITY, MALIGNANCY, MAJOR ADVERSE CARDIOVASCULAR
EVENTS, and THROMBOSIS
See full prescribing information for complete boxed warning.
- Increased risk of serious bacterial, fungal, viral, and opportunistic infections, including tuberculosis (TB), leading to hospitalization or death. Interrupt tofacitinib extended-release tablets treatment if serious infection occurs until the infection is controlled. Test for latent TB before and during therapy; treat latent TB prior to use. Monitor all patients for active TB during treatment, even patients with initial negative latent TB test. (5.1)
- Higher rate of all-cause mortality, including sudden cardiovascular (CV) death with tofacitinib vs. TNF blockers in rheumatoid arthritis (RA) patients. (5.2)
- Malignancies have occurred in patients treated with tofacitinib. Higher rate of lymphomas and lung cancers with tofacitinib vs. TNF blockers in RA patients. (5.3)
- Higher rate of major adverse CV events (defined as CV death, myocardial infarction, and stroke) with tofacitinib vs. TNF blockers in RA patients. (5.4)
- Thrombosis has occurred in patients treated with tofacitinib. Increased incidence of pulmonary embolism, venous and arterial thrombosis with tofacitinib vs. TNF blockers in RA patients. (5.5)
RECENT MAJOR CHANGES
INDICATIONS AND USAGE
Tofacitinib extended-release tablets are Janus kinase (JAK) inhibitors.
Tofacitinib extended-release tablets are indicated for the treatment of adult patients with:
- Moderately to severely active rheumatoid arthritis (RA), who have had an inadequate response or intolerance to one or more TNF blockers.
- Active psoriatic arthritis (PsA), who have had an inadequate response or intolerance to one or more TNF blockers.
- Active ankylosing spondylitis (AS), who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use:
DOSAGE AND ADMINISTRATION
Recommended Evaluations and Immunization Prior to Treatment Initiation
- Prior to initiating tofacitinib extended-release tablets, consider performing an active and latent TB evaluation, viral hepatitis screening, a complete blood count, and updating immunizations. Avoid tofacitinib extended-release tablets initiation if absolute lymphocyte count <500 cells/mm3, an absolute neutrophil count (ANC) <1000 cells/mm3 or hemoglobin <9 g/dL. (2.1)
Important Administration Instructions
- Tofacitinib extended-release tablets are not substitutable with tofacitinib tablets and oral solution. (2.2)
- Switching between tofacitinib tablets and tofacitinib extended-release tablets should be made by the healthcare provider. (2.2)
Recommended Dosage
Adult Patients with RA, PsA or AS
- Tofacitinib extended-release tablets 11 mg once daily. (2.3)
Dosage in Patients with Renal Impairment or Hepatic Impairment
- Use of tofacitinib extended-release tablets in patients with severe HI is not recommended. (2.3, 8.7)
- See full prescribing information (FPI) for recommended dosage in patients with moderate or severe RI or moderate HI. (2.3, 8.6, 8.7)
Dosage Modification
See the full prescribing information for dosage modification by indication for patients who concomitantly use CYP2C19 and/or CYP3A4 inhibitors and patients with lymphopenia, neutropenia, or anemia. (2.3, 7)
DOSAGE FORMS AND STRENGTHS
- Tofacitinib extended-release tablets: 11 mg tofacitinib (3)
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
- Serious Infections: Avoid use of tofacitinib extended-release tablets during an active serious infection, including localized infections. (5.1)
- Gastrointestinal Perforations: Promptly evaluate patients at increased risk for gastrointestinal perforation who present with new onset abdominal symptoms. (5.6)
- Laboratory Monitoring: Recommended due to potential changes in lymphocytes, neutrophils, hemoglobin, liver enzymes and lipids. (5.8)
- Vaccinations: Avoid use of live vaccines concurrently with tofacitinib extended-release tablets. (5.9)
ADVERSE REACTIONS
Most common adverse reactions are:
- RA, PsA, and AS: Reported in ≥2% of adult patients treated with tofacitinib tablets monotherapy or in combination with DMARDs: upper respiratory tract infection (URI), nasopharyngitis, diarrhea, and headache. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Aurobindo Pharma USA, Inc. at 1-866-850-2876 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
Lactation: Advise not to breastfeed. (8.2)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 3/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: SERIOUS INFECTIONS, MORTALITY, MALIGNANCY, MAJOR ADVERSE CARDIOVASCULAR EVENTS, and THROMBOSIS
1 INDICATIONS AND USAGE
1.1 Rheumatoid Arthritis
1.2 Psoriatic Arthritis
1.3 Ankylosing Spondylitis
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Evaluations and Immunization Prior to Treatment Initiation
2.2 Important Administration Instructions
2.3 Recommended Dosage in Adults with Rheumatoid Arthritis, Psoriatic Arthritis, and Ankylosing Spondylitis
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Serious Infections
5.2 Increased Risk of Mortality
5.3 Malignancy and Lymphoproliferative Disorders
5.4 Major Adverse Cardiovascular Events
5.5 Thrombosis
5.6 Gastrointestinal Perforations
5.7 Hypersensitivity Reactions
5.8 Laboratory Abnormalities
5.9 Vaccinations
5.10 Risk of Gastrointestinal Obstruction with Tofacitinib Extended-Release Tablets - A Non-Deformable Extended-Release Formulation
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Clinical Studies in Rheumatoid Arthritis
14.2 Clinical Studies in Psoriatic Arthritis
14.3 Clinical Studies in Ankylosing Spondylitis
14.6 Safety Study in Adults with Rheumatoid Arthritis (Tofacitinib Versus TNF-blocker)
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: SERIOUS INFECTIONS, MORTALITY, MALIGNANCY, MAJOR ADVERSE CARDIOVASCULAR EVENTS, and THROMBOSIS
SERIOUS INFECTIONS
Patients treated with tofacitinib extended-release tablets are at increased risk for developing serious bacterial, fungal, viral, and opportunistic infections, including tuberculosis (TB), that may lead to hospitalization or death [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)]. Most patients who developed these infections were taking concomitant immunosuppressants such as methotrexate or corticosteroids. Reported infections included:
- Active TB, which may present with pulmonary or extrapulmonary disease. Patients should be tested for latent TB before tofacitinib extended-release tablets use and during therapy. Treatment for latent infection should be initiated prior to tofacitinib extended-release tablets use.
- Invasive fungal infections, including cryptococcosis and pneumocystosis. Patients with invasive fungal infections may present with disseminated, rather than localized, disease.
- Bacterial, viral, including herpes zoster, and other infections due to opportunistic pathogens.
The risks and benefits of tofacitinib extended-release tablets treatment should be carefully considered prior to initiating therapy in patients with chronic or recurrent infection.
Patients should be closely monitored for the development of signs and symptoms of infection during and after tofacitinib extended-release tablets treatment, including the possible development of TB in patients who tested negative for latent TB infection prior to initiating therapy. If a serious infection develops, interrupt tofacitinib extended-release tablets until the infection is controlled [see Warnings and Precautions (5.1)].
MORTALITY
In a large, randomized, postmarketing safety study in rheumatoid arthritis (RA) patients 50 years of age and older with at least one cardiovascular (CV) risk factor comparing tofacitinib tablets 5 mg or 10 mg twice a day to tumor necrosis factor (TNF) blockers, a higher rate of all-cause mortality, including sudden CV death, was observed with tofacitinib tablets 5 mg or 10 mg twice a day [see Warnings and Precautions (5.2)]. Tofacitinib tablets 10 mg twice daily and tofacitinib extended-release tablets 22 mg once daily dosages are not recommended for the treatment of RA, psoriatic arthritis (PsA), or ankylosing spondylitis (AS) [see Dosage and Administration (2.3)].
MALIGNANCIESMalignancies, including lymphomas and solid tumors, have occurred in patients treated with tofacitinib and other Janus kinase inhibitors used to treat inflammatory conditions. In RA patients, a higher rate of malignancies (excluding non-melanoma skin cancer (NMSC)) was observed in patients treated with tofacitinib tablets 5 mg or 10 mg twice a day compared with TNF blockers [see Warnings and Precautions (5.3)].
Lymphomas and lung cancers were observed at a higher rate in patients treated with tofacitinib tablets 5 mg or 10 mg twice a day in RA patients compared to those treated with TNF blockers. Patients who are current or past smokers are at additional increased risk.
MAJOR ADVERSE CARDIOVASCULAR EVENTS
RA patients 50 years of age and older with at least one cardiovascular risk factor, treated with tofacitinib tablets 5 mg or 10 mg twice daily, had a higher rate of major adverse cardiovascular events (MACE) (defined as cardiovascular death, myocardial infarction, and stroke), compared to those treated with TNF blockers. Patients who are current or past smokers are at additional increased risk. Discontinue tofacitinib extended-release tablets in patients that have experienced a myocardial infarction or stroke [see Warnings and Precautions (5.4)].
THROMBOSIS
Thrombosis, including pulmonary embolism, deep venous thrombosis, and arterial thrombosis have occurred in patients treated with tofacitinib and other Janus kinase inhibitors used to treat inflammatory conditions. Many of these events were serious and some resulted in death. RA patients 50 years of age and older with at least one cardiovascular risk factor treated with tofacitinib tablets 5 mg or 10 mg twice daily compared to TNF blockers had an observed increase in incidence of these events. Avoid tofacitinib extended-release tablets in patients at risk. Discontinue tofacitinib extended-release tablets and promptly evaluate patients with symptoms of thrombosis [see Warnings and Precautions (5.5)].
-
1 INDICATIONS AND USAGE
1.1 Rheumatoid Arthritis
Tofacitinib extended-release tablets are indicated for the treatment of adult patients with moderately to severely active rheumatoid arthritis (RA), who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of tofacitinib extended-release tablets in combination with biologic disease-modifying antirheumatic drugs (DMARDs) or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
1.2 Psoriatic Arthritis
Tofacitinib extended-release tablets are indicated for the treatment of adults with active PsA who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of tofacitinib extended-release tablets in combination with biologic DMARDs or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
1.3 Ankylosing Spondylitis
Tofacitinib extended-release tablets are indicated for the treatment of adult patients with active ankylosing spondylitis (AS), who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use
Use of tofacitinib extended-release tablets in combination with biologic DMARDs or potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Evaluations and Immunization Prior to Treatment Initiation
Prior to initiating tofacitinib extended-release tablets, consider performing the following:
- Active and latent tuberculosis (TB) infection evaluation: If the patient has latent TB, treat for TB prior to tofacitinib extended-release tablets treatment [see Warnings and Precautions (5.1)].
- Viral hepatitis screening in accordance with clinical guidelines [see Warnings and Precautions (5.1)].
- A complete blood count: Avoid initiation of tofacitinib extended-release tablets treatment in patients with a lymphocyte count less than 500 cells/mm3, absolute neutrophil count less than 1000 cells/mm3, or hemoglobin level less than 9 g/dL [see Warnings and Precautions (5.8)].
- Baseline hepatic function evaluation: Tofacitinib extended-release tablets are not recommended for patients with severe hepatic impairment [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
- Update immunizations according to current immunization guidelines. The interval between live vaccinations and initiation of tofacitinib extended-release tablets should be in accordance with current vaccination guidelines regarding immunosuppressive agents [see Warnings and Precautions (5.9)].
2.2 Important Administration Instructions
- Tofacitinib extended-release tablets are not substitutable with tofacitinib tablets and oral solution. Switching between tofacitinib tablets and tofacitinib extended-release tablets should be made by the healthcare provider.
- Dose interruption is recommended for management of lymphopenia, neutropenia, and anemia [see Warnings and Precautions (5.8) and Adverse Reactions (6.1)].
- Interrupt use of tofacitinib extended-release tablets if a patient develops a serious infection until the infection is controlled [see Warnings and Precautions (5.1)].
- Take tofacitinib extended-release tablets with or without food [see Clinical Pharmacology (12.3)].
- Swallow tofacitinib extended-release tablets whole and intact. Do not crush, split, or chew the extended-release tablets [see Clinical Pharmacology (12.3)].
2.3 Recommended Dosage in Adults with Rheumatoid Arthritis, Psoriatic Arthritis, and Ankylosing Spondylitis
Table 1 displays the recommended dosage of tofacitinib extended-release tablets for adults with RA, PsA, and AS [see Indication and Usage (1.1, 1.2, 1.3)] with and without renal impairment (including those who are undergoing hemodialysis) or hepatic impairment [see Use in Specific Populations (8.6, 8.7)]. The table also displays the recommended dosage modifications for patients concomitantly using CYP2C19 and/or CYP3A4 inhibitors [see Drug Interactions (7) and Clinical Pharmacology (12.3)], and patients with lymphopenia, neutropenia, or anemia.
Table 1: Recommended Dosage of Tofacitinib Extended-Release Tablets in Adults with Rheumatoid Arthritis, Psoriatic Arthritis, or Ankylosing Spondylitis Adults
Tofacitinib Extended-Release Tablets
Patients with Normal Renal and
Hepatic Functiona
11 mg once daily
Recommended Dosage in Patients with Renal Impairment (RI)b
Mild RI
(CLcr >50 and ≤80 mL/min)
11 mg once daily
Moderate RI
(CLcr ≥30 and ≤50 mL/min)
Tofacitinib tablets 5 mg once daily
Severe RI
(CLcr <30 mL/min)
Tofacitinib tablets 5 mg once daily
For patients undergoing hemodialysis, administer the dose after the dialysis session on dialysis days. If a dose was taken before the dialysis procedure, supplemental doses are not recommended after dialysis.
Recommended Dosage in Patients with Hepatic Impairment (HI)
Mild HI
(Child-Pugh A)
11 mg once daily
Moderate HI
(Child-Pugh B)
Tofacitinib tablets 5 mg once daily
Severe HI
(Child-Pugh C)
Use of tofacitinib extended-release tablets is not recommended.
Dosage Modifications with Concomitant Use of CYP3A4 and/or CYP2C19 Inhibitor(s)
Strong CYP2C19 inhibitor(s)
11 mg once daily
Moderate CYP2C19 inhibitor(s)
Moderate CYP3A4 inhibitor(s)
Moderate CYP3A4 inhibitor(s)
with strong CYP2C19 inhibitor(s)
(e.g., fluconazole)
Tofacitinib tablets 5 mg once daily
Strong CYP3A4 inhibitor(s)
Dosage Modifications for Lymphopenia, Neutropenia, or Anemia
Patients with lymphocyte count less
than 500 cells/mm3, confirmed by
repeat testing
Discontinue dosing.
Patients with ANC less than
500 cells/mm3
Discontinue dosing.
Patients with ANC 500 to
1000 cells/mm3
Interrupt dosing. When ANC is greater than 1000, resume 11 mg once daily.
Patients with hemoglobin less than 8 g/dL
or a decrease of more than 2 g/dL
Interrupt dosing until hemoglobin values have normalized.
a Excludes patients who concomitantly use tofacitinib extended-release tablets with strong CYP3A4 inhibitor(s) or moderate CYP3A4 inhibitor(s) and strong CYP2C19 inhibitor(s), as well as patients with lymphocyte count less than 500 cells/mm3, ANC <1000 cells/mm3, or hemoglobin less than 8 g/dL or a decrease of more than 2 g/dL.
b Tofacitinib PK was evaluated in subjects with varying degrees of renal impairment, where the severity of renal impairment was defined based on creatinine clearance (CLcr) estimated using the Cockcroft-Gault equation: CLcr >80 mL/min (normal renal function); >50 and ≤80 mL/min (mild renal impairment); ≥30 and ≤50 mL/min (moderate renal impairment); <30 mL/min (severe renal impairment).
Switching from Tofacitinib Tablets to Tofacitinib Extended-Release Tablets
Patients treated with tofacitinib tablets 5 mg twice daily may be switched to tofacitinib extended-release tablets 11 mg once daily the day following the last dose of tofacitinib tablets 5 mg.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Serious Infections
Serious and sometimes fatal infections may occur with tofacitinib extended-release tablets. Serious and sometimes fatal infections due to bacterial, mycobacterial, invasive fungal, viral, or other opportunistic pathogens have been reported in patients receiving tofacitinib. The most common serious infections reported with tofacitinib included pneumonia, urinary tract infection, cellulitis, herpes zoster, bronchitis, septic shock, diverticulitis, gastroenteritis, appendicitis, and sepsis. Among opportunistic infections, tuberculosis and other mycobacterial infections, cryptococcosis, histoplasmosis, esophageal candidiasis, pneumocystosis, multi-dermatomal herpes zoster, cytomegalovirus infections, BK virus infection, and listeriosis were reported with tofacitinib. Some patients have presented with disseminated rather than localized disease, and were often taking concomitant immunomodulating agents such as methotrexate or corticosteroids.
Other serious infections that were not reported in clinical studies may also occur (e.g., coccidioidomycosis).
Avoid use of tofacitinib extended-release tablets in patients with an active, serious infection, including localized infections. The risks and benefits of treatment should be considered prior to initiating tofacitinib extended-release tablets in patients:
- with chronic or recurrent infection
- who have been exposed to tuberculosis
- with a history of a serious or an opportunistic infection
- who have resided or traveled in areas of endemic tuberculosis or endemic mycoses; or
- with underlying conditions that may predispose them to infection.
Closely monitor patients for the development of signs and symptoms of infection during and after treatment with tofacitinib extended-release tablets. Interrupt tofacitinib extended-release tablets if a patient develops a serious infection, an opportunistic infection, or sepsis. In patients who develop a new infection during treatment with tofacitinib extended-release tablets, promptly complete diagnostic testing appropriate for an immunocompromised patient; initiate appropriate antimicrobial therapy, and monitor the patients closely.
Caution is also recommended in patients with a history of chronic lung disease, or in those who develop interstitial lung disease, as they may be more prone to infections.
Risk of infection may be higher with increasing degrees of lymphopenia and consideration should be given to lymphocyte counts when assessing individual patient risk of infection. Discontinuation and monitoring criteria for lymphopenia are recommended [see Dosage and Administration (2.3)].
Tuberculosis
Evaluate and test patients for latent or active tuberculosis (TB) infection prior to and per applicable guidelines during administration of tofacitinib extended-release tablets.
Consider anti-TB therapy prior to administration of tofacitinib extended-release tablets in patients with a past history of latent or active TB in whom an adequate course of treatment cannot be confirmed, and for patients with a negative test for latent TB but who have risk factors for TB infection.
Consultation with a physician with expertise in the treatment of TB is recommended to aid in the decision about whether initiating anti-TB therapy is appropriate for an individual patient.
Monitor patients closely for the development of signs and symptoms of TB, including patients who tested negative for latent TB infection prior to initiating therapy.
Treat patients with latent TB with standard antimycobacterial therapy before administering tofacitinib extended-release tablets.
Viral Reactivation
Viral reactivation, including cases of herpes virus reactivation (e.g., herpes zoster), were observed in clinical studies with tofacitinib. Postmarketing cases of hepatitis B reactivation have been reported in patients treated with tofacitinib. The impact of tofacitinib extended-release tablets on chronic viral hepatitis reactivation is unknown. Patients who screened positive for hepatitis B or C were excluded from clinical trials. Perform screening for viral hepatitis in accordance with clinical guidelines before starting therapy with tofacitinib extended-release tablets. The risk of herpes zoster is increased in patients treated with tofacitinib extended-release tablets and appears to be higher in patients treated with tofacitinib in Japan and Korea.
5.2 Increased Risk of Mortality
Increased risk of mortality may occur with tofacitinib extended-release tablets. Adult patients with rheumatoid arthritis (RA), 50 years of age and older, with at least one cardiovascular risk factor treated with tofacitinib tablets 5 mg or 10 mg twice a day had a higher observed rate of all-cause mortality, including sudden cardiovascular death, compared to those treated with TNF blockers in a large, randomized, postmarketing safety study (RA Safety Study 1). The incidence rate of all-cause mortality per 100 patient-years was 1.23 for tofacitinib tablets 10 mg twice a day, 0.88 for tofacitinib tablets 5 mg twice a day, and 0.69 for TNF blockers [see Clinical Studies (14.6)]. Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with tofacitinib extended-release tablets.
Tofacitinib tablets 10 mg twice daily (or tofacitinib extended-release tablets 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, or AS [see Dosage and Administration (2.3)].
5.3 Malignancy and Lymphoproliferative Disorders
Malignancies and lymphoproliferative disorders may occur with tofacitinib extended-release tablets. Malignancies, including lymphomas and solid cancers, were observed in clinical studies of tofacitinib [see Adverse Reactions (6.1)].
Other malignancies were observed in tofacitinib clinical studies and the postmarketing setting, including, but not limited to, lung cancer, breast cancer, melanoma, prostate cancer, and pancreatic cancer.
In RA Safety Study 1, a higher rate of malignancies (excluding non-melanoma skin cancer (NMSC)) was observed in patients treated with tofacitinib tablets 5 mg or 10 mg twice a day compared with TNF blockers. The incidence rate of malignancies (excluding NMSC) per 100 patient-years was 1.13 for tofacitinib tablets 10 mg twice a day, 1.13 for tofacitinib tablets 5 mg twice a day, and 0.77 for TNF blockers. Patients who are current or past smokers are at additional increased risk [see Clinical Studies (14.6)].
Lymphomas and lung cancers, which are a subset of all malignancies in RA Safety Study 1, were observed at a higher rate in patients treated with tofacitinib tablets 5 mg twice a day and tofacitinib tablets 10 mg twice a day compared to those treated with TNF blockers. The incidence rate of lymphomas per 100 patient-years was 0.11 for tofacitinib tablets 10 mg twice a day, 0.07 for tofacitinib tablets 5 mg twice a day, and 0.02 for TNF blockers. The incidence rate of lung cancers per 100 patient-years among current and past smokers was 0.59 for tofacitinib tablets 10 mg twice a day, 0.48 for tofacitinib tablets 5 mg twice a day, and 0.27 for TNF blockers [see Clinical Studies (14.6)].
Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with tofacitinib extended-release tablets, particularly in patients with a known malignancy (other than a successfully treated NMSC), patients who develop a malignancy while on treatment, and patients who are current or past smokers. Tofacitinib tablets 10 mg twice daily (or tofacitinib extended-release tablets 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, or AS [see Dosage and Administration (2.3)].
Non-Melanoma Skin Cancer
Non-melanoma skin cancers (NMSCs) have been reported in patients treated with tofacitinib tablets. Periodic skin examination is recommended for patients who are at increased risk for skin cancer.
5.4 Major Adverse Cardiovascular Events
Major adverse cardiovascular events may occur with tofacitinib extended-release tablets. In RA Safety Study 1, patients with RA who were 50 years of age and older with at least one cardiovascular risk factor and treated with tofacitinib tablets 5 mg or 10 mg twice daily had a higher rate of major adverse cardiovascular events (MACE) defined as cardiovascular death, non-fatal myocardial infarction (MI), and non-fatal stroke, compared to those treated with TNF blockers. The incidence rate of MACE per 100 patient-years was 1.11 for tofacitinib tablets 10 mg twice a day, 0.91 for tofacitinib tablets 5 mg twice a day, and 0.79 for TNF blockers. The incidence rate of fatal or non-fatal myocardial infarction per 100 patient-years was 0.39 for tofacitinib tablets 10 mg twice a day, 0.36 for tofacitinib tablets 5 mg twice a day, and 0.2 for TNF blockers [see Clinical Studies (14.6)]. Patients who are current or past smokers are at additional increased risk.
Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with tofacitinib extended-release tablets, particularly in patients who are current or past smokers and patients with other cardiovascular risk factors. Inform patients about the symptoms of serious cardiovascular events and the steps to take if they occur. Discontinue tofacitinib extended-release tablets in patients that have experienced a MI or stroke. Tofacitinib tablets 10 mg twice daily (or tofacitinib extended-release tablets 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, or AS [see Dosage and Administration (2.3)].
5.5 Thrombosis
Thrombosis may occur with tofacitinib extended-release tablets. Thrombosis, including pulmonary embolism (PE), deep venous thrombosis (DVT), and arterial thrombosis, have occurred in patients treated with tofacitinib and other Janus kinase (JAK) inhibitors used to treat inflammatory conditions. Many of these events were serious and some resulted in death [see Warnings and Precautions (5.2)].
Patients with RA 50 years of age and older with at least one cardiovascular risk factor treated with tofacitinib tablets 5 mg or 10 mg twice daily compared to TNF blockers in RA Safety Study 1 had an observed increase in incidence of these thrombotic events. The incidence rate of DVT per 100 patient-years was 0.28 for tofacitinib tablets 10 mg twice a day, 0.22 for tofacitinib tablets 5 mg twice a day, and 0.16 for TNF blockers. The incidence rate of PE per 100 patient-years was 0.49 for tofacitinib tablets 10 mg twice a day, 0.18 for tofacitinib tablets 5 mg twice a day, and 0.05 for TNF blockers [see Clinical Studies (14.6)].
Tofacitinib tablets 10 mg twice daily (or tofacitinib extended-release tablets 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, or AS [see Dosage and Administration (2.3)].
Promptly evaluate patients with symptoms of thrombosis and discontinue tofacitinib extended-release tablets in patients with symptoms of thrombosis.
Avoid tofacitinib extended-release tablets in patients that may be at increased risk of thrombosis.
5.6 Gastrointestinal Perforations
Gastrointestinal perforations may occur with tofacitinib extended-release tablets. Events of gastrointestinal perforation have been reported in clinical studies with tofacitinib tablets, although the role of JAK inhibition in these events is not known. In these studies, many patients with RA received background therapy with nonsteroidal anti-inflammatory drugs (NSAIDs).
Promptly evaluate patients treated with tofacitinib extended-release tablets who may be at increased risk for gastrointestinal perforation (e.g., patients with a history of diverticulitis or taking NSAIDs) and who present with new onset abdominal symptoms for early identification of gastrointestinal perforation [see Adverse Reactions (6.1)].
5.7 Hypersensitivity Reactions
Hypersensitivity reactions may occur with tofacitinib extended-release tablets. Reactions such as angioedema and urticaria that may reflect drug hypersensitivity have been observed in patients receiving tofacitinib extended-release tablets. Some events were serious. If a serious hypersensitivity reaction occurs, promptly discontinue tofacitinib extended-release tablets while evaluating the potential cause or causes of the reaction [see Adverse Reactions (6.2)].
5.8 Laboratory Abnormalities
Laboratory abnormalities may occur with tofacitinib extended-release tablets.
Lymphocyte Abnormalities
Treatment with tofacitinib tablets was associated with initial lymphocytosis at one month of tofacitinib tablets treatment followed by a gradual decrease in mean absolute lymphocyte counts below the baseline of approximately 10% during 12 months of therapy. Lymphocyte counts less than 500 cells/mm3 in these patients were associated with an increased incidence of treated and serious infections.
- Monitor lymphocyte counts at baseline and every 3 months thereafter.
- Avoid initiation of tofacitinib extended-release tablets treatment in patients with a low lymphocyte count (i.e., less than 500 cells/mm3). In patients who develop a confirmed absolute lymphocyte count less than 500 cells/mm3, treatment with tofacitinib extended-release tablets not recommended.
Neutropenia
Treatment with tofacitinib tablets was associated with an increased incidence of neutropenia (less than 2000 cells/mm3) compared to treatment with placebo.
- Monitor neutrophil counts at baseline and after 4 to 8 weeks of treatment and every 3 months thereafter.
- Avoid initiation of tofacitinib extended-release tablets treatment in patients with a low neutrophil count (i.e., ANC less than 1000 cells/mm3). For patients who develop a persistent ANC of 500 to 1000 cells/mm3, interrupt dosing until ANC is greater than or equal to 1000 cells/mm3. In patients who develop an ANC less than 500 cells/mm3, treatment with tofacitinib extended-release tablets are not recommended.
Anemia
- Monitor hemoglobin at baseline and after 4 to 8 weeks of treatment and every 3 months thereafter.
- Avoid initiation of tofacitinib extended-release tablets treatment in patients with a low hemoglobin level (i.e., less than 9 g/dL). Interrupt treatment with tofacitinib extended-release tablets in patients who develop hemoglobin levels less than 8 g/dL or whose hemoglobin level drops greater than 2 g/dL on treatment until hemoglobin values have normalized.
Liver Enzyme Elevations
Treatment with tofacitinib tablets were associated with an increased incidence of liver enzyme elevation compared to treatment with placebo. Most of these abnormalities occurred in studies with background DMARD therapy (primarily methotrexate).
- Routine monitoring of liver tests and prompt investigation of the causes of liver enzyme elevations is recommended to identify potential cases of drug-induced liver injury.
- If drug-induced liver injury is suspected, interrupt the administration of tofacitinib extended-release tablets until this diagnosis has been excluded.
Lipid Elevations
Treatment with tofacitinib tablets were associated with dose-dependent increases in lipid parameters including total cholesterol, low-density lipoprotein (LDL) cholesterol, and high-density lipoprotein (HDL) cholesterol. Maximum changes in these lipid parameters were generally observed within 6 weeks. There were no clinically relevant changes in LDL/HDL cholesterol ratios. The effect of these lipid parameter elevations on cardiovascular morbidity and mortality has not been determined.
- Perform assessment of lipid parameters approximately 4 to 8 weeks following initiation of tofacitinib extended-release tablets therapy.
- Manage patients according to clinical guidelines [e.g., National Cholesterol Educational Program (NCEP)] for the management of hyperlipidemia.
5.9 Vaccinations
Avoid use of live vaccines concurrently with tofacitinib extended-release tablets. Prior to initiating tofacitinib extended-release tablets therapy, update immunizations in agreement with current immunization guidelines. The interval between live vaccinations and initiation of tofacitinib extended-release tablets therapy should be in accordance with current vaccination guidelines regarding immunosuppressive agents.
5.10 Risk of Gastrointestinal Obstruction with Tofacitinib Extended-Release Tablets - A Non-Deformable Extended-Release Formulation
Gastrointestinal obstruction may occur with tofacitinib extended-release tablets. Avoid use of tofacitinib extended-release tablets in patients with pre-existing severe gastrointestinal narrowing (pathologic or iatrogenic). There have been rare reports of obstructive symptoms in patients with known strictures in association with the ingestion of other drugs utilizing a non-deformable extended-release formulation.
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Serious Infections [see Warnings and Precautions (5.1)]
- Increased Risk of Mortality [see Warnings and Precautions (5.2)]
- Malignancy and Lymphoproliferative Disorders [see Warnings and Precautions (5.3)]
- Major Adverse Cardiovascular Events [see Warnings and Precautions (5.4)]
- Thrombosis [see Warnings and Precautions (5.5)]
- Gastrointestinal Perforations [see Warnings and Precautions (5.6)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
- Laboratory Abnormalities [see Warnings and Precautions (5.8)]
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not predict the rates observed in a broader patient population in clinical practice.
The clinical studies described in this subsection were conducted using tofacitinib tablets and/or tofacitinib oral solution.
Adverse Reactions in Adults with Rheumatoid Arthritis
In RA Safety Study 1, 1,455 adults were treated with tofacitinib 5 mg twice daily, 1,456 adults were treated with 10 mg twice daily, and 1,451 adults were treated with a TNF blocker for a median of 4 years [see Clinical Studies (14.6)]. A dosage of tofacitinib 10 mg twice daily is not recommended for the treatment of RA because of increased risks [see Dosage and Administration (2.3) and Warnings and Precautions (5)]. For the treatment of adults with moderately to severely active RA [see Indications and Usage (1.1)], the recommended dosage of tofacitinib is 5 mg twice daily and the recommended dosage for tofacitinib extended-release tablets is 11 mg once daily.
The safety of tofacitinib was also evaluated in two Phase 2 and five Phase 3 double-blind, placebo-controlled, multicenter trials in patients with RA. In these trials, adults were randomized to receive:
- Tofacitinib (monotherapy) 5 mg twice daily (292 patients) or 10 mg twice daily (306 patients),
- In combination with DMARDs (including methotrexate), tofacitinib 5 mg twice daily (1044 patients) or 10 mg twice daily (1043 patients) and
- Placebo (809 patients).
All seven trials included provisions for patients taking placebo to receive treatment with tofacitinib at Month 3 or Month 6 either by patient response (based on uncontrolled disease activity) or by design, so that adverse events cannot always be unambiguously attributed to a given treatment. Therefore, some analyses that follow include patients who changed treatment by design or by patient response from placebo to tofacitinib in both the placebo and tofacitinib group of a given interval. Comparisons between placebo and tofacitinib groups were based on the first 3 months of exposure, and comparisons between tofacitinib 5 mg twice daily and tofacitinib 10 mg twice daily were based on the first 12 months of exposure.
The long-term safety population includes all adults with RA who participated in a double-blind, placebo-controlled trial (including earlier development phase studies) and then participated in one of two long-term safety studies. The design of the long-term safety studies allowed for modification of tofacitinib doses according to clinical judgment. This limits the interpretation of the long-term safety data with respect to dose.
The most common serious adverse reactions were serious infections [see Warnings and Precautions (5.1)].
The proportion of patients who discontinued treatment due to any adverse reaction during the 0 to 3 months exposure in the double-blind, placebo-controlled trials was 4% for tofacitinib-treated patients and 3% for placebo-treated patients.
Overall Infections
In the seven placebo-controlled trials in patients with RA, during the 0 to 3 months exposure, the overall frequency of infections was 20% and 22% in the tofacitinib 5 mg twice daily and tofacitinib 10 mg twice daily groups, respectively, and 18% in the placebo group.
The most commonly reported infections with tofacitinib were upper respiratory tract infections, nasopharyngitis, and urinary tract infections (4%, 3%, and 2% of patients, respectively).
Serious Infections: In the seven placebo-controlled trials in patients with RA, during the 0 to 3 months exposure, serious infections were reported in 1 patient (0.5 events per 100 patient-years) who received placebo and 11 patients (1.7 events per 100 patient-years) who received tofacitinib 5 mg or 10 mg twice daily. The rate difference between treatment groups (and the corresponding 95% confidence interval) was 1.1 (-0.4, 2.5) events per 100 patient-years for the combined tofacitinib 5 mg twice daily and 10 mg twice daily group minus placebo.
In the seven placebo-controlled trials, during the 0 to 12 months exposure, serious infections were reported in 34 patients (2.7 events per 100 patient-years) who received tofacitinib 5 mg twice daily and 33 patients (2.7 events per 100 patient-years) who received tofacitinib 10 mg twice daily. The rate difference between tofacitinib doses (and the corresponding 95% confidence interval) was -0.1 (-1.3, 1.2) events per 100 patient-years for tofacitinib 10 mg twice daily minus tofacitinib 5 mg twice daily.
The most common serious infections included pneumonia, cellulitis, herpes zoster, and urinary tract infection [see Warnings and Precautions (5.1)].
Tuberculosis: In the seven placebo-controlled trials in patients with RA, during the 0 to 3 months exposure, tuberculosis (TB) was not reported in patients who received placebo, tofacitinib 5 mg twice daily, or tofacitinib 10 mg twice daily.
In the seven placebo-controlled trials, during the 0 to 12 months exposure, TB was reported in 0 patients who received tofacitinib 5 mg twice daily and 6 patients (0.5 events per 100 patient-years) who received tofacitinib 10 mg twice daily. The rate difference between tofacitinib doses (and the corresponding 95% confidence interval) was 0.5 (0.1, 0.9) events per 100 patient-years for tofacitinib 10 mg twice daily minus tofacitinib 5 mg twice daily.
Cases of disseminated TB were also reported. The median tofacitinib exposure prior to diagnosis of TB was 10 months (range from 152 to 960 days) [see Warnings and Precautions (5.1)].
Opportunistic Infections (excluding tuberculosis): In the seven placebo-controlled trials in patients with RA, during the 0 to 3 months exposure, opportunistic infections were not reported in patients who received placebo, tofacitinib 5 mg twice daily, or tofacitinib 10 mg twice daily.
In the seven placebo-controlled trials, during the 0 to 12 months exposure, opportunistic infections were reported in 4 patients (0.3 events per 100 patient-years) who received tofacitinib 5 mg twice daily and 4 patients (0.3 events per 100 patient-years) who received tofacitinib 10 mg twice daily. The rate difference between tofacitinib doses (and the corresponding 95% confidence interval) was 0 (-0.5, 0.5) events per 100 patient-years for tofacitinib 10 mg twice daily minus tofacitinib 5 mg twice daily.
The median tofacitinib exposure prior to diagnosis of an opportunistic infection was 8 months (range from 41 to 698 days) [see Warnings and Precautions (5.1)].
Malignancies
In the seven placebo-controlled trials in patients with RA, during the 0 to 3 months exposure, malignancies excluding NMSC were reported in 0 patients who received placebo and 2 patients (0.3 events per 100 patient-years) who received either tofacitinib 5 mg or 10 mg twice daily. The rate difference between treatment groups (and the corresponding 95% confidence interval) was 0.3 (-0.1, 0.7) events per 100 patient-years for the combined tofacitinib 5 mg and 10 mg twice daily group minus placebo.
In the seven placebo-controlled trials, during the 0 to 12 months exposure, malignancies excluding NMSC were reported in 5 patients (0.4 events per 100 patient-years) who received tofacitinib 5 mg twice daily and 7 patients (0.6 events per 100 patient-years) who received tofacitinib 10 mg twice daily. The rate difference between tofacitinib doses (and the corresponding 95% confidence interval) was 0.2 (-0.4, 0.7) events per 100 patient-years for tofacitinib 10 mg twice daily minus tofacitinib 5 mg twice daily. One of these malignancies was a case of lymphoma that occurred during the 0 to 12 month period in a patient treated with tofacitinib 10 mg twice daily.
The most common types of malignancy, including malignancies observed during the long-term extension in tofacitinib-treated patients, were lung and breast cancer, followed by gastric, colorectal, renal cell, prostate cancer, lymphoma, and malignant melanoma [see Warnings and Precautions (5.3)].
Laboratory Abnormalities
Lymphopenia: In the placebo-controlled clinical trials in patients with RA, confirmed decreases in absolute lymphocyte counts below 500 cells/mm3 occurred in 0.04% of patients for the tofacitinib 5 mg twice daily and 10 mg twice daily groups combined during the first 3 months of exposure.
Confirmed lymphocyte counts less than 500 cells/mm3 were associated with an increased incidence of treated and serious infections [see Warnings and Precautions (5.8)].
Neutropenia: In the placebo-controlled clinical trials in patients with RA, confirmed decreases in ANC below 1,000 cells/mm3 occurred in 0.07% of patients for the tofacitinib 5 mg twice daily and 10 mg twice daily groups combined during the first 3 months of exposure.
There were no confirmed decreases in ANC below 500 cells/mm3 observed in any treatment group. There was no clear relationship between neutropenia and the occurrence of serious infections.
In the long-term safety population, the pattern and incidence of confirmed decreases in ANC remained consistent with what was seen in the placebo-controlled clinical trials [see Warnings and Precautions (5.8)].
Liver Enzyme Elevations: Confirmed increases in liver enzymes greater than 3 times the upper limit of normal (3x ULN) were observed in patients with RA treated with tofacitinib. In patients experiencing liver enzyme elevation, modification of treatment regimen, such as reduction in the dose of concomitant DMARD, interruption of tofacitinib, or reduction in tofacitinib dosage, resulted in decrease or normalization of liver enzymes.
In the placebo-controlled monotherapy trials (0 to 3 months), no differences in the incidence of ALT or AST elevations were observed between the placebo, and tofacitinib 5 mg, and 10 mg twice daily groups.
In the placebo-controlled background DMARD trials (0 to 3 months), ALT elevations greater than 3x ULN were observed in 1.0%, 1.3% and 1.2% of patients who received placebo, tofacitinib 5 mg, and 10 mg twice daily, respectively. In these trials, AST elevations greater than 3x ULN were observed in 0.6%, 0.5% and 0.4% of patients who received placebo, tofacitinib 5 mg, and 10 mg twice daily, respectively.
One case of drug-induced liver injury was reported in a patient treated with tofacitinib 10 mg twice daily for approximately 2.5 months. The patient developed symptomatic elevations of AST and ALT greater than 3x ULN and bilirubin elevations greater than 2x ULN, which required hospitalizations and a liver biopsy.
Lipid Elevations: In the placebo-controlled clinical trials in patients with RA, dose-related elevations in lipid parameters (total cholesterol, LDL cholesterol, HDL cholesterol, triglycerides) were observed at one month of exposure and remained stable thereafter. Changes in lipid parameters during the first 3 months of exposure in the placebo-controlled clinical trials are summarized below:
- Mean LDL cholesterol increased by 15% in the tofacitinib 5 mg twice daily arm and 19% in the tofacitinib 10 mg twice daily arm.
- Mean HDL cholesterol increased by 10% in the tofacitinib 5 mg twice daily arm and 12% in the tofacitinib 10 mg twice daily arm.
- Mean LDL/HDL ratios were essentially unchanged in tofacitinib-treated patients.
In a placebo-controlled clinical trial, elevations in LDL cholesterol and ApoB decreased to pretreatment levels in response to statin therapy.
In the long-term safety population, elevations in lipid parameters remained consistent with what was seen in the placebo-controlled clinical trials.
Serum Creatinine Elevations: In the placebo-controlled clinical trials in patients with RA, dose-related elevations in serum creatinine were observed with tofacitinib treatment. The mean increase in serum creatinine was <0.1 mg/dL in the 12-month pooled safety analysis; however, with increasing duration of exposure in the long-term extensions, up to 2% of patients were discontinued from tofacitinib treatment due to the protocol-specified discontinuation criterion of an increase in creatinine by more than 50% of baseline. The clinical significance of the observed serum creatinine elevations is unknown.
Common Adverse Reactions
Table 5 displays adverse reactions that occurred in 2% or more of patients on tofacitinib 5 mg or 10 mg twice daily and at least 1% greater than in tofacitinib-treated patients that observed in placebo-treated patients with or without DMARD in the RA trials.
Table 5: Common Adverse Reactions* in Clinical Trials of Tofacitinib for the Treatment of Rheumatoid Arthritis in Adults With or Without Concomitant DMARDs (0 to 3 Months) N reflects randomized and treated patients from the seven placebo-controlled clinical trials. * reported in ≥2% of patients treated with either dose of tofacitinib and ≥1% greater than that reported for placebo.
** The recommended dose of tofacitinib for the treatment of RA is 5 mg twice daily [see Dosage and Administration (2)].
Preferred Term
Placebo
Tofacitinib
5 mg Twice Daily
Tofacitinib
10 mg Twice Daily**
N = 809
(%)
N = 1336
(%)
N = 1349
(%)
Upper respiratory tract infection
3
4
4
Nasopharyngitis
3
4
3
Diarrhea
2
4
3
Headache
2
4
3
Hypertension
1
2
2
Other adverse reactions that occurred in placebo-controlled and open-label extension studies in patients with RA included:
Blood and lymphatic system disorders: Anemia
Infections and infestations: Diverticulitis
Metabolism and nutrition disorders: Dehydration
Psychiatric disorders: Insomnia
Nervous system disorders: Paresthesia
Respiratory, thoracic and mediastinal disorders: Dyspnea, cough, sinus congestion, interstitial lung disease (cases were limited to patients with RA and some were fatal)
Gastrointestinal disorders: Abdominal pain, dyspepsia, vomiting, gastritis, nausea Hepatobiliary disorders: Hepatic steatosis
Skin and subcutaneous tissue disorders: Rash, erythema, pruritus
Musculoskeletal, connective tissue and bone disorders: Musculoskeletal pain, arthralgia, tendonitis, joint swelling
Neoplasms benign, malignant and unspecified (including cysts and polyps): Non-melanoma skin cancers
General disorders and administration site conditions: Pyrexia, fatigue, peripheral edema
Clinical Experience in Methotrexate-Naïve Patients
Study RA-VI was an active-controlled clinical trial in methotrexate-naïve patients [see Clinical Studies (14)]. The safety experience in these patients was consistent with Studies RA-I through V.
Adverse Reactions in Adults with Psoriatic Arthritis
The safety of tofacitinib was evaluated in 2 double-blind Phase 3 clinical trials in adults with active psoriatic arthritis (PsA):
- Study PsA-I (NCT01877668) had a duration of 12 months and enrolled adults who had an inadequate response to a nonbiologic DMARD and who were naïve to treatment with a TNF blocker. Study PsA-I included a 3-month placebo-controlled period and also included adalimumab 40 mg subcutaneously once every 2 weeks for 12 months.
- Study PsA-II (NCT01882439) had a duration of 6 months and enrolled adults who had an inadequate response to at least one approved TNF blocker. This clinical trial included a 3-month placebo-controlled period.
In these combined Phase 3 clinical trials, 238 patients were randomized and treated with tofacitinib 5 mg twice daily and 236 patients were randomized and treated with tofacitinib 10 mg twice daily. A dosage of tofacitinib 10 mg twice daily is not recommended for the treatment of PsA. For the treatment of adults with active PsA [see Indications and Usage (1.2)], the recommended dosage of tofacitinib is 5 mg twice daily and the recommended dosage for tofacitinib extended-release tablets is 11 mg once daily [see Dosage and Administration (2.3)].
All patients in the clinical trials in patients with PsA were required to receive treatment with a stable dose of a nonbiologic DMARD [the majority (79%) received methotrexate]. The study population randomized and treated with tofacitinib (474 patients) included 45 (10%) patients aged 65 years or older and 66 (14%) patients with diabetes at baseline.
During the 2 PsA controlled clinical trials, there were:
- 3 malignancies (excluding NMSC) in 474 patients who received tofacitinib plus non-biologic DMARD (6 to 12 months exposure)
- 0 malignancies in 236 patients who received placebo plus non-biologic DMARD group (3 months exposure) and
- 0 malignancies in 106 patients in patients who received adalimumab plus non-biologic DMARD group (12 months exposure).
No lymphomas were reported. Malignancies have also been observed in the long-term extension study in patients with PsA treated with tofacitinib.
The safety profile observed in adults with active PsA treated with tofacitinib was consistent with the safety profile observed in adults with RA.
Adverse Reactions in Adults with Ankylosing Spondylitis
The safety of tofacitinib was evaluated in adults with active ankylosing spondylitis (AS) in a double-blind placebo-controlled Phase 3 clinical trial (Study AS-I) and in a dose-ranging Phase 2 clinical trial (Study AS-II).
- Study AS-I (NCT03502616) had a duration of 48 weeks and enrolled adults who had an inadequate response to at least 2 NSAIDs. Study AS-I included a 16-week double-blind period in which patients received tofacitinib 5 mg or placebo twice daily and a 32-week open-label treatment period in which all patients received tofacitinib 5 mg twice daily.
- Study AS-II (NCT01786668) had a duration of 16 weeks and enrolled adults who had an inadequate response to at least 2 NSAIDs. This clinical trial included a 12-week treatment period in which patients received either tofacitinib 2 mg (40% of the recommended dose), 5 mg, 10 mg, or placebo twice daily. A dosage of tofacitinib 10 mg twice daily is not recommended for the treatment of AS. For the treatment of adults with active AS [see Indications and Usage (1.3)], the recommended dosage of tofacitinib is 5 mg twice daily and the recommended dosage for tofacitinib extended-release tablets is 11 mg once daily [see Dosage and Administration (2.3)].
In the combined Phase 2 and Phase 3 clinical trials, a total of 420 patients were treated with either tofacitinib 2 mg, 5 mg, or 10 mg twice daily. Of these, 316 patients were treated with tofacitinib 5 mg twice daily for up to 48 weeks. In the combined double-blind period, 185 patients were randomized to and treated with tofacitinib 5 mg twice daily and 187 to placebo for up to 16 weeks. Concomitant treatment with stable doses of nonbiologic DMARDs, NSAIDs, or corticosteroids (≤10 mg/day) was permitted. The study population randomized and treated with tofacitinib included 13 (3%) patients aged 65 years or older and 18 (4%) patients with diabetes at baseline.
The safety profile observed in adults with AS treated with tofacitinib was consistent with the safety profile observed in adults with RA and PsA.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of tofacitinib extended-release tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: Drug hypersensitivity (events such as angioedema and urticaria have been observed)
Skin and subcutaneous tissue disorders: Acne
-
7 DRUG INTERACTIONS
Table 7 includes drugs with clinically significant drug interactions when concomitantly used with tofacitinib extended-release tablets and instructions for preventing or managing them.
Table 7: Clinically Significant Interactions Affecting Tofacitinib Extended-Release Tablets When Concomitantly Used with Other Drugs Strong CYP3A4 Inhibitors (e.g., ketoconazole)
Clinical Impact
Increased exposure to tofacitinib
Intervention
Dosage modification of tofacitinib extended-release tablets is recommended [see Dosage and Administration (2), Clinical Pharmacology, Figure 3 (12.3)]
Moderate CYP3A4 Inhibitors Concomitantly Used with Strong CYP2C19 Inhibitors (e.g., fluconazole)
Clinical Impact
Increased exposure to tofacitinib
Intervention
Dosage modification of tofacitinib extended-release tablets is recommended [see Dosage and Administration (2), Clinical Pharmacology, Figure 3 (12.3)]
Strong CYP3A4 Inducers (e.g., rifampin)
Clinical Impact
Decreased exposure to tofacitinib and may result in loss of or reduced clinical response
Intervention
Concomitant use with tofacitinib extended-release tablets is not recommended [see Clinical Pharmacology, Figure 3 (12.3)]
Immunosuppressive Drugs (e.g., azathioprine, tacrolimus, cyclosporine)
Clinical Impact
Risk of added immunosuppression; concomitant use of tofacitinib extended-release tablets with biologic DMARDs or potent immunosuppressants has not been studied in patients with RA, PsA, or AS.
Intervention
Concomitant use with tofacitinib extended-release tablets is not recommended [see Indications and Usage (1), Clinical Pharmacology, Figure 3 (12.3)]
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
The available data with tofacitinib extended-release tablets from a pregnancy exposure registry that enrolled 11 exposed pregnant females, pharmacovigilance, and published literature are insufficient to draw conclusions about a drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes. There are risks to the mother and the fetus associated with RA in pregnancy (see Clinical Considerations). In animal reproduction studies, fetocidal and teratogenic effects were noted when pregnant rats and rabbits received tofacitinib during the period of organogenesis at exposures multiples of 73-times and 6.3-times the maximum recommended dose of 10 mg twice daily, respectively. Further, in a peri- and post-natal study in rats, tofacitinib resulted in reductions in live litter size, postnatal survival, and pup body weights at exposure multiples of approximately 73-times the recommended dosage of 5 mg twice daily and approximately 36 times the maximum recommended dosage of 10 mg twice daily, respectively (see Data).
The background risks of major birth defects and miscarriage for the indicated populations are unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. The background risks in the U.S. general population of major birth defects and miscarriages are 2 to 4% and 15 to 20% of clinically recognized pregnancies, respectively.
Clinical ConsiderationsDisease-Associated Maternal and/or Embryo/Fetal Risk: Published data suggest that increased disease activity is associated with the risk of developing adverse pregnancy outcomes in women with RA. Adverse pregnancy outcomes include preterm delivery (before 37 weeks of gestation), low birth weight (less than 2500 grams) infants, and small for gestational age at birth.
Data
Animal Data: In a rat embryofetal developmental study, in which pregnant rats received tofacitinib during organogenesis, tofacitinib was teratogenic at exposure levels approximately 146 times the recommended dose of 5 mg twice daily, and approximately 73 times the maximum recommended dose of 10 mg twice daily (on an AUC basis at oral doses of 100 mg/kg/day in rats). Teratogenic effects consisted of external and soft tissue malformations of anasarca and membranous ventricular septal defects, respectively; and skeletal malformations or variations (absent cervical arch; bent femur, fibula, humerus, radius, scapula, tibia, and ulna; sternoschisis; absent rib; misshapen femur; branched rib; fused rib; fused sternebra; and hemicentric thoracic centrum). In addition, there was an increase in post-implantation loss, consisting of early and late resorptions, resulting in a reduced number of viable fetuses. Mean fetal body weight was reduced. No developmental toxicity was observed in rats at exposure levels approximately 58 times the recommended dose of 5 mg twice daily, and approximately 29 times the maximum recommended dose of 10 mg twice daily (on an AUC basis at oral doses of 30 mg/kg/day in pregnant rats).
In a rabbit embryofetal developmental study in which pregnant rabbits received tofacitinib during the period of organogenesis, tofacitinib was teratogenic at exposure levels approximately 13 times the recommended dose of 5 mg twice daily, and approximately 6.3 times the maximum recommended dose of 10 mg twice daily (on an AUC basis at oral doses of 30 mg/kg/day in rabbits) in the absence of signs of maternal toxicity. Teratogenic effects included thoracogastroschisis, omphalocele, membranous ventricular septal defects, and cranial/skeletal malformations (microstomia, microphthalmia), mid-line and tail defects. In addition, there was an increase in post-implantation loss associated with late resorptions. No developmental toxicity was observed in rabbits at exposure levels approximately 3 times the recommended dose of 5 mg twice daily, and approximately 1.5 times the maximum recommended dose of 10 mg twice daily (on an AUC basis at oral doses of 10 mg/kg/day in pregnant rabbits).
In a peri- and postnatal development study in pregnant rats that received tofacitinib from gestation day 6 through day 20 of lactation, there were reductions in live litter size, postnatal survival, and pup body weights at exposure levels approximately 73 times the recommended dose of 5 mg twice daily, and approximately 36 times the maximum recommended dose of 10 mg twice daily (on an AUC basis at oral doses of 50 mg/kg/day in rats). There was no effect on behavioral and learning assessments, sexual maturation or the ability of the F1 generation rats to mate and produce viable F2 generation fetuses in rats at exposure levels approximately 17 times the recommended dose of 5 mg twice daily, and approximately 8.3 times the maximum recommended dose of 10 mg twice daily (on an AUC basis at oral doses of 10 mg/kg/day in rats).
8.2 Lactation
Risk Summary
Based on published data, tofacitinib is present in human milk. Data on the effects of tofacitinib on the breastfed infant is limited to a small number of cases with no reported adverse effects. There are no data on the effects on milk production. Given the serious adverse reactions seen in patients treated with tofacitinib extended-release tablets, such as increased risk of serious infections, advise patients that breastfeeding is not recommended during treatment and for at least 36 hours after the last dose of tofacitinib extended-release tablets (approximately 6 elimination half-lives).
Data
Following administration of tofacitinib to lactating rats, concentrations of tofacitinib in milk over time paralleled those in serum and were approximately 2 times higher in milk relative to maternal serum at all time points measured.
8.3 Females and Males of Reproductive Potential
Contraception
Females
In an animal reproduction study, tofacitinib at AUC multiples of 13 times the recommended dosage of 5 mg twice daily and 6.3 times the maximum recommended dosage of 10 mg twice daily demonstrated adverse embryo-fetal findings [see Use in Specific Populations (8.1)]. However, there is uncertainty as to how these animal findings relate to females of reproductive potential treated with the recommended clinical dosage. Consider pregnancy planning and prevention for females of reproductive potential.
Infertility
Females
Based on findings in rats, treatment with tofacitinib extended-release tablets may result in reduced fertility in females of reproductive potential. It is not known if this effect is reversible [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
The safety and effectiveness of tofacitinib extended-release tablets in pediatric patients have not been established.
8.5 Geriatric Use
Of the 3315 adults who were enrolled in clinical trials with RA (Studies RA-I to V), a total of 505 patients were 65 years of age and older, including 71 patients 75 years and older. The frequency of serious infection among tofacitinib tablets-treated patients 65 years of age and older was higher than among those adults under the age of 65.
Of the 783 tofacitinib tablet-treated patients in clinical trials of patients with PsA, a total of 72 (9.2%) patients were 65 years of age and older, including 2 (0.3%) patients 75 years and older. These clinical studies did not include sufficient numbers of patients aged 65 years and older with PsA to determine if they respond differently from younger adult patients.
Of the 420 tofacitinib tablet-treated patients in clinical trials of patients with AS, a total of 12 (2.9%) patients were 65 years of age and older, including 1 (0.2%) patient 75 years and older. These clinical studies did not include sufficient numbers of patients aged 65 years and older with AS to determine if they respond differently from younger adult patients.
8.6 Renal Impairment
Moderate and Severe Renal Impairment
Tofacitinib-treated patients with moderate renal impairment (RI) (CLcr ≥30 and ≤50 mL/minute) or severe RI (<30 mL/minute) had greater tofacitinib blood concentrations than tofacitinib-treated patients with normal renal function (CLcr >80 mL/minute). The recommended dosage of tofacitinib extended-release tablets in patients with moderate or severe RI (including those with severe RI who are undergoing hemodialysis) is lower than the recommended dosage in patients with normal renal function [see Dosage and Administration (2.3)].
Mild Renal Impairment
The recommended dosage in patients with mild RI (CLcr >50 and ≤80 mL/minute) is the same as patients with normal renal function.
8.7 Hepatic Impairment
Severe Hepatic Impairment
Tofacitinib extended-release tablets have not been studied in patients with severe hepatic impairment (HI) (Child-Pugh C); therefore, use of tofacitinib extended-release tablets in patients with severe HI is not recommended.
Moderate Hepatic Impairment
Tofacitinib-treated patients with moderate hepatic impairment (Child-Pugh B) had greater tofacitinib blood concentration than tofacitinib-treated patients with normal hepatic function [see Clinical Pharmacology (12.3)]. Higher blood concentrations may increase the risk of some adverse reactions. The recommended tofacitinib extended-release tablets dosage in patients with moderate HI is lower than the recommended dosage in patients with normal hepatic function [see Dosage and Administration (2.3)].
Mild Hepatic Impairment
The recommended dosage of tofacitinib extended-release tablets in patients with mild hepatic impairment (Child-Pugh A) is the same as patients with normal hepatic function.
Hepatitis B or C Serology
The safety and efficacy of tofacitinib extended-release tablets have not been studied in patients with positive hepatitis B virus or hepatitis C virus serology.
- DRUG ABUSE AND DEPENDENCE
-
10 OVERDOSAGE
There is no specific antidote for overdose with tofacitinib extended-release tablets. In case of an overdose, it is recommended that the patient be monitored for signs and symptoms of adverse reactions.
In a study in patients with end-stage renal disease (ESRD) undergoing hemodialysis, plasma tofacitinib concentrations declined more rapidly during the period of hemodialysis and dialyzer efficiency, calculated as dialyzer clearance/blood flow entering the dialyzer, was high [mean (SD) = 0.73 (0.15)]. However, due to the significant non-renal clearance of tofacitinib, the fraction of total elimination occurring by hemodialysis was small, and thus, limits the value of hemodialysis for treatment of overdose with tofacitinib extended-release tablets.
Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
-
11 DESCRIPTION
Tofacitinib extended-release tablets are formulated with the citrate salt of tofacitinib, a JAK inhibitor.
Tofacitinib citrate is a white to off-white crystalline powder with the following chemical name: (3R,4R)-4-methyl-3-(methyl-7H-pyrrolo [2,3-d]pyrimidin-4-ylamino)-ß-oxo-1-piperidinepropanenitrile, 2-hydroxy-1,2,3-propanetricarboxylate (1:1).
The solubility of tofacitinib citrate in water is 2.9 mg/mL.
Tofacitinib citrate has a molecular weight of 504.5 Daltons (or 312.4 Daltons as the tofacitinib free base) and a molecular formula of C16H20N6OC6H8O7. The chemical structure of tofacitinib citrate is:
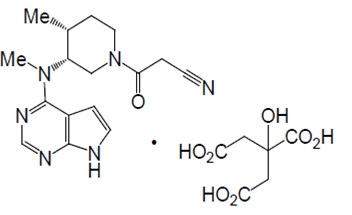
Tofacitinib extended-release tablets are supplied for oral administration as a 11 mg pink, oval, bevel edged, mat finished film-coated extended-release tablets imprinted with “TI11” on one side and plain on other side. Each 11 mg tablet of tofacitinib extended-release tablet contains 11 mg tofacitinib (equivalent to 17.77 mg tofacitinib citrate) and the following inactive ingredients: butylated hydroxytoluene, cellulose acetate, colloidal silicon dioxide, copovidone, hydroxyethyl cellulose, hydroxypropyl cellulose, hypromellose, iron oxide red, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene oxide, titanium dioxide and triacetin. Printing ink contains, iron oxide black, propylene glycol, and shellac glaze.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Tofacitinib is a Janus kinase (JAK) inhibitor. JAKs are intracellular enzymes which transmit signals arising from cytokine or growth factor-receptor interactions on the cellular membrane to influence cellular processes of hematopoiesis and immune cell function. Within the signaling pathway, JAKs phosphorylate and activate Signal Transducers and Activators of Transcription (STATs) which modulate intracellular activity including gene expression. Tofacitinib modulates the signaling pathway at the point of JAKs, preventing the phosphorylation and activation of STATs. JAK enzymes transmit cytokine signaling through pairing of JAKs (e.g., JAK1/JAK3, JAK1/JAK2, JAK1/TyK2, JAK2/JAK2). Tofacitinib inhibited the in vitro activities of JAK1/JAK2, JAK1/JAK3, and JAK2/JAK2 combinations with IC50 of 406, 56, and 1377 nM, respectively. However, the relevance of specific JAK combinations to therapeutic effectiveness is not known.
12.2 Pharmacodynamics
Treatment with tofacitinib was associated with dose-dependent reductions of circulating CD16/56+ natural killer cells, with estimated maximum reductions occurring at approximately 8 to 10 weeks after initiation of therapy. These changes generally resolved within 2 to 6 weeks after discontinuation of treatment. Treatment with tofacitinib was associated with dose-dependent increases in B cell counts. Changes in circulating T-lymphocyte counts and T-lymphocyte subsets (CD3+, CD4+ and CD8+) were small and inconsistent. The clinical significance of these changes is unknown.
Total serum IgG, IgM, and IgA levels after 6-month dosing in patients with rheumatoid arthritis (RA) were lower than in patients who received placebo; however, changes were small and not dose-dependent.
After treatment with tofacitinib in patients with RA, rapid decreases in serum C-reactive protein (CRP) were observed and maintained throughout dosing. Changes in CRP observed with tofacitinib treatment do not reverse fully within 2 weeks after discontinuation, indicating a longer duration of pharmacodynamic activity compared to the pharmacokinetic half-life.
Similar changes in T cells, B cells, and serum CRP have been observed in patients with active psoriatic arthritis (PsA) although reversibility was not assessed. Total serum immunoglobulins were not assessed in patients with active PsA.
12.3 Pharmacokinetics
Following oral administration of tofacitinib tablets, peak plasma concentrations were reached within 0.5 hour to 1 hour, elimination half-life was about 3 hours and a dose-proportional increase in systemic exposure was observed in the therapeutic dosage range. Steady state concentrations were achieved in 24 to 48 hours with negligible accumulation after twice daily administration.
Following oral administration of tofacitinib extended-release tablets, peak plasma concentrations were reached at 4 hours and half-life was about 6 to 8 hours. Steady state concentrations were achieved within 48 hours with negligible accumulation after once daily administration.
Table 8 describes the pharmacokinetic parameters of tofacitinib tablets and tofacitinib extended-release tablets.
Table 8: Pharmacokinetic Parameters of Tofacitinib Tablets/Tofacitinib Extended-Release Tablets Following Multiple Oral Dosing Abbreviations: AUC24 = area under the concentration time profile from time 0 to 24 hours; Cmax = maximum plasma concentration; Cmin = minimum plasma concentration; Tmax = time to Cmax; CV = Coefficient of variation.
a Values represent the geometric mean, except T max, for which is the median (range) is shown.
b Values beyond 12 hours were after the evening dose which was administered 12 hours after the morning dose of twice-daily tofacitinib.
PK Parametersa (CV%)
Tofacitinib Tablets
Tofacitinib Extended-Release Tablets
Dosing Regimen
5 mg
Twice Daily
10 mg
Twice Daily
11 mg
Once Daily
22 mg
Once Daily
AUC24 (ng.hr/mL)
263.4 (15)
539.6 (22)
269.0 (18)
596.6 (19)
Cmax (ng/mL)
42.7 (26)
84.7 (18)
38.2 (15)
83.8 (25)
Cmin (ng/mL)
1.41 (40)
3.10 (54)
1.07 (69)
3.11 (43)
Tmax (hours)
1.0
(0.5 to14.0b)
0.8
(0.5 to 14.0b)
4.0
(3.0 to 4.0)
4.0
(2.0 to 4.0)
Absorption
Tofacitinib Tablets
The absolute oral bioavailability of tofacitinib tablets is 74%. Coadministration of tofacitinib tablets with a high-fat meal resulted in no changes in AUC while Cmax was reduced by 32%. In clinical trials, tofacitinib tablets were administered without regard to meals [see Dosage and Administration (2.2)].
Tofacitinib Extended-Release Tablets
Coadministration of tofacitinib extended-release tablets 11 and 22 mg with a high-fat meal resulted in no changes in AUC while Cmax was increased by 27% and 19% respectively. Tmax was extended by approximately 1 hour for tofacitinib extended-release tablets 11 and 22 mg.
Distribution
After intravenous administration, the volume of distribution was 87 L. The protein binding of tofacitinib is approximately 40%. Tofacitinib binds predominantly to albumin and does not appear to bind to α1-acid glycoprotein. Tofacitinib distributes equally between red blood cells and plasma.
Metabolism and Excretion
Clearance mechanisms for tofacitinib are approximately 70% hepatic metabolism and 30% renal excretion of the parent drug. The metabolism of tofacitinib is primarily mediated by CYP3A4 with minor contribution from CYP2C19. In a human radiolabeled study, more than 65% of the total circulating radioactivity was accounted for by unchanged tofacitinib, with the remaining 35% attributed to 8 metabolites, each accounting for less than 8% of total radioactivity. The pharmacologic activity of tofacitinib is attributed to the parent molecule.
Pharmacokinetics in Patients with RA, PsA, and AS
Population pharmacokinetic (PK) analyses indicated that PK characteristics were similar between patients with RA, PsA, and ankylosing spondylitis. The coefficient of variation (%) in AUC of tofacitinib were generally similar across different disease patients, ranging from 22% to 34% (Table 9).
Table 9: Tofacitinib Exposure in Patients with RA, PsA, and AS After Administration of Tofacitinib 5 mg Twice Daily Abbreviations: AUC0-24,ss = area under the plasma concentration-time curve over 24 hours at steady state;
CV = coefficient of variation.
a Pharmacokinetic parameters estimated based on population pharmacokinetic analysis.
Pharmacokinetic Parametersa Geometric Mean
(CV%)
Tofacitinib 5 mg
Twice Daily
Rheumatoid Arthritis
Psoriatic Arthritis
Ankylosing
Spondylitis
AUC0-24,ss
(ng·h/mL)
504
(22.0%)
419
(34.1%)
381
(25.4%)
Specific Populations
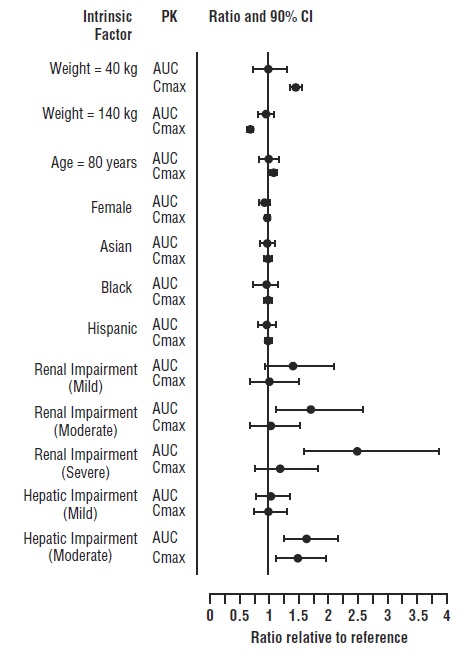
Covariate evaluation as part of population PK analyses in adult patient populations indicated no clinically relevant change in tofacitinib exposure, after accounting for differences in renal function (i.e., creatinine clearance) between patients, based on age, weight, biological sex and race (Figure 1). An approximately linear relationship between body weight and volume of distribution was observed, resulting in higher peak (Cmax) and lower trough (Cmin) concentrations in lighter patients. However, this difference is not considered to be clinically relevant.
The effect of renal and hepatic impairment and other intrinsic factors on the PK of tofacitinib is shown in Figure 1.
Figure 1: Impact of Intrinsic Factors on Tofacitinib Pharmacokinetics
Note: Reference values for weight, age, biological sex, and race comparisons are 70 kg, 55 years, male, and white, respectively; reference groups for renal and hepatic impairment data are patients with normal renal and hepatic function. Renal function was estimated using creatinine clearance by Cockcroft-Gault method and hepatic function was estimated using Child-Pugh scoring method.
In patients with end-stage renal disease maintained on hemodialysis, mean AUC was approximately 40% higher compared with historical healthy subject data, consistent with approximately 30% contribution of renal clearance to the total clearance of tofacitinib [see Dosage and Administration (2.3) and Use in Specific Populations (8.6)].
Drug Interaction Studies
Potential for Tofacitinib Extended-Release Tablets to Influence the PK of Other Drugs
In vitro studies indicate that tofacitinib does not significantly inhibit or induce the activity of the major human drug-metabolizing CYPs (CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP3A4) at concentrations corresponding to the steady state Cmax of a 10 mg twice daily dose. These in vitro results were confirmed by a human drug interaction study showing no changes in the pharmacokinetics of midazolam, a highly sensitive CYP3A4 substrate, when concomitantly administered with tofacitinib.
In vitro studies indicate that tofacitinib does not significantly inhibit the activity of the major human drug-metabolizing uridine 5'-diphospho-glucuronosyltransferases (UGTs) [UGT1A1, UGT1A4, UGT1A6, UGT1A9, and UGT2B7] at concentrations exceeding 250 times the steady state Cmax of a 10 mg twice daily dose.
In patients with RA, the oral clearance of tofacitinib does not vary with time, indicating that tofacitinib does not normalize CYP enzyme activity in patients with RA. Therefore, concomitant use with tofacitinib extended-release tablets is not expected to result in clinically relevant increases in the metabolism of CYP substrates in patients with RA.
In vitro data indicate that the potential for tofacitinib to inhibit transporters such as P-glycoprotein, organic anionic or cationic transporters at therapeutic concentrations is low.
The impact of tofacitinib on the PK of other drugs for the concomitant drugs are shown in Figure 2.
Figure 2: Impact of Tofacitinib on the Pharmacokinetics of Other Drugs
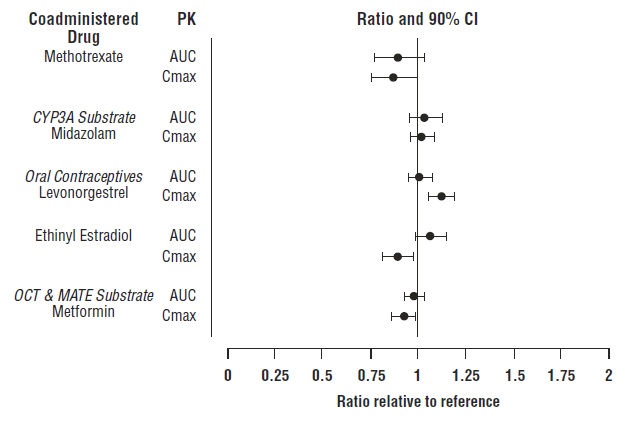
Note: Reference group is administration of concomitant medication alone; OCT = Organic Cationic Transporter; MATE = Multidrug and Toxic Compound Extrusion.
Potential for Other Drugs to Influence the Pharmacokinetics of Tofacitinib
Since tofacitinib is metabolized by CYP3A4, interaction with drugs that inhibit or induce CYP3A4 is likely. Inhibitors of CYP2C19 alone or P-glycoprotein are unlikely to substantially alter the pharmacokinetics of tofacitinib (see Figure 3).
Figure 3: Impact of Other Drugs on the Pharmacokinetics of Tofacitinib
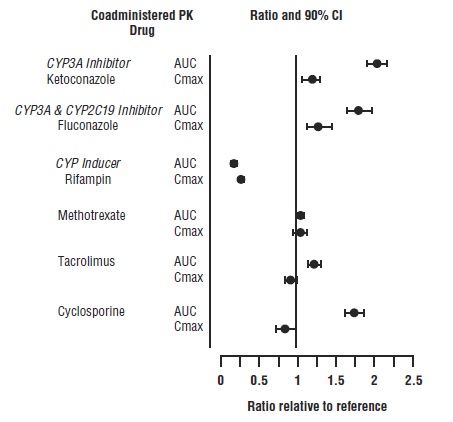
Note: Reference group is administration of tofacitinib alone.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 39-week toxicology study in monkeys, tofacitinib at exposure levels approximately 6 times the recommended dose of 5 mg twice daily, and approximately 3 times the 10 mg twice daily dose (on an AUC basis at oral doses of 5 mg/kg twice daily) produced lymphomas. No lymphomas were observed in this study at exposure levels 1 times the recommended dose of 5 mg twice daily, and approximately 0.5 times the 10 mg twice daily dose (on an AUC basis at oral doses of 1 mg/kg twice daily).
The carcinogenic potential of tofacitinib was assessed in 6-month rasH2 transgenic mouse carcinogenicity and 2-year rat carcinogenicity studies. Tofacitinib, at exposure levels approximately 34 times the recommended dose of 5 mg twice daily, and approximately 17 times the 10 mg twice daily dose (on an AUC basis at oral doses of 200 mg/kg/day) was not carcinogenic in mice.
In the 24-month oral carcinogenicity study in Sprague-Dawley rats, tofacitinib caused benign Leydig cell tumors, hibernomas (malignancy of brown adipose tissue), and benign thymomas at doses greater than or equal to 30 mg/kg/day (approximately 42 times the exposure levels at the recommended dose of 5 mg twice daily, and approximately 21 times the 10 mg twice daily dose on an AUC basis). The relevance of benign Leydig cell tumors to human risk is not known.
Tofacitinib was not mutagenic in the bacterial reverse mutation assay. It was positive for clastogenicity in the in vitro chromosome aberration assay with human lymphocytes in the presence of metabolic enzymes, but negative in the absence of metabolic enzymes. Tofacitinib was negative in the in vivo rat micronucleus assay and in the in vitro CHO-HGPRT assay and the in vivo rat hepatocyte unscheduled DNA synthesis assay.
In rats, tofacitinib at exposure levels approximately 17 times the recommended dose of 5 mg twice daily, and approximately 8.3 times the 10 mg twice daily dose (on an AUC basis at oral doses of 10 mg/kg/day) reduced female fertility due to increased post-implantation loss. There was no impairment of female rat fertility at exposure levels of tofacitinib equal to the recommended dose of 5 mg twice daily, and approximately 0.5 times the 10 mg twice daily dose (on an AUC basis at oral doses of 1 mg/kg/day). Tofacitinib exposure levels at approximately 133 times the recommended dose of 5 mg twice daily, and approximately 67 times the 10 mg twice daily dose (on an AUC basis at oral doses of 100 mg/kg/day) had no effect on male fertility, sperm motility, or sperm concentration.
-
14 CLINICAL STUDIES
14.1 Clinical Studies in Rheumatoid Arthritis
The rheumatoid arthritis (RA) clinical development program with tofacitinib tablets included six randomized controlled trials in adults with moderate to severe active RA.
Trial Design
- Study RA-I (NCT00814307) was a 6-month monotherapy trial in which 610 patients with moderate to severe active RA who had an inadequate response to a DMARD (nonbiologic or biologic) received tofacitinib 5 mg or 10 mg twice daily or placebo added to their background DMARD. At the Month 3 visit, all patients randomized to placebo treatment were switched in a blinded fashion to a second predetermined treatment of tofacitinib 5 mg or 10 mg twice daily. The primary endpoints at Month 3 were the proportion of patients who achieved an ACR20 response, changes in Health Assessment Questionnaire – Disability Index (HAQ-DI), and rates of Disease Activity Score DAS28-4(ESR) less than 2.6.
- Study RA-II (NCT00856544) was a 12-month trial in which 792 patients with moderate to severe active RA who had an inadequate response to a nonbiologic DMARD received tofacitinib 5 mg or 10 mg twice daily or placebo added to background DMARD treatment (excluding potent immunosuppressive treatments such as azathioprine or cyclosporine). At the Month 3 visit, nonresponding patients were switched in a blinded fashion to a second predetermined treatment of tofacitinib 5 mg or 10 mg twice daily. At the end of Month 6, all patients treated with placebo were switched to their second predetermined tofacitinib treatment in a blinded fashion. The primary endpoints were the proportion of patients who achieved an ACR20 response at Month 6, changes in HAQ-DI at Month 3, and rates of DAS28-4(ESR) less than 2.6 at Month 6.
- Study RA-III (NCT00853385) was a 12-month trial in 717 patients with moderate to severe active RA who had an inadequate response to methotrexate (MTX). Patients received tofacitinib 5 mg or 10 mg orally twice daily, adalimumab 40 mg subcutaneously every other week, or placebo added to background MTX. Patients treated with placebo were switched as in Study RA-II. The primary endpoints were the proportion of patients who achieved an ACR20 response at Month 6, HAQ-DI at Month 3, and DAS28-4(ESR) less than 2.6 at Month 6.
- Study RA-IV (NCT00847613) was a 2-year trial with a planned analysis at 1 year in which 797 patients with moderate to severe active RA who had an inadequate response to MTX received tofacitinib 5 mg or 10 mg twice daily or placebo added to background MTX. Patients treated with placebo were switched as in Study RA-II. The primary endpoints were the proportion of patients who achieved an ACR20 response at Month 6, mean change from baseline in van der Heijde-modified total Sharp Score (mTSS) at Month 6, HAQ-DI at Month 3, and DAS28-4(ESR) less than 2.6 at Month 6.
- Study RA-V (NCT00960440) was a 6-month trial in which 399 patients with moderate to severe active RA who had an inadequate response to at least one approved TNF blocking biological product received tofacitinib 5 mg or 10 mg twice daily or placebo added to background MTX. At the Month 3 visit, all patients randomized to placebo treatment were switched in a blinded fashion to a second predetermined treatment of tofacitinib 5 or 10 mg twice daily. The primary endpoints at Month 3 were the proportion of patients who achieved an ACR20 response, HAQ-DI, and DAS28-4(ESR) less than 2.6.
- Study RA-VI (NCT01039688) was a 2-year monotherapy trial with a planned analysis at 1 year in which 952 MTX-naïve patients with moderate to severe active RA received tofacitinib 5 or 10 mg twice daily or MTX dose-titrated over 8 weeks to 20 mg weekly. The primary endpoints were mean change from baseline in van der Heijde-modified Total Sharp Score (mTSS) at Month 6 and the proportion of patients who achieved an ACR70 response at Month 6.
Although other dosages have been studied, the recommended dosage of tofacitinib is 5 mg twice daily. Tofacitinib 10 mg twice daily is not recommended for the treatment of RA [see Dosage and Administration (2.3)].
Clinical Response
The percentages of tofacitinib-treated patients who achieved ACR20, ACR50, and ACR70 responses in Studies RA-I, IV, and V are shown in Table 10. Similar results were observed with Studies RA-II and III. In trials RA-I through V, patients treated with 5 mg twice daily tofacitinib had higher ACR20, ACR50, and ACR70 response rates versus patients treated with placebo, with or without background DMARD treatment, at Month 3 and Month 6. Higher ACR20 response rates were observed within 2 weeks compared to placebo. In the 12-month trials, ACR response rates in tofacitinib-treated patients were consistent at 6 and 12 months.
Table 10: Proportion of Adults with Moderate to Severe Active RA with an ACR Response at Months 3 and 6 in Studies RA-I, IV, and V a N is number of randomized and treated patients.
b NA (not applicable), as data for placebo treatment is not available beyond 3 months in Studies RA-I and RA-V due to placebo advancement.
c Inadequate response to at least one DMARD (biologic or nonbiologic) due to lack of efficacy or toxicity.
d Inadequate response to MTX defined as the presence of sufficient residual disease activity to meet the entry criteria.
e Inadequate response to a least one TNF blocker due to lack of efficacy and/or intolerance.
Monotherapy in Nonbiologic or Biologic DMARD Inadequate Respondersc
MTX Inadequate Respondersd
TNF Blocker Inadequate Responderse
Study RA-I
Study RA-IV
Study RA-V
Na
Placebo + background DMARD
122
Tofacitinib
5 mg Twice Daily + background DMARD
243
Placebo + background MTX
160
Tofacitinib
5 mg Twice Daily + background MTX
321
Placebo + background MTX
132
Tofacitinib
5 mg Twice Daily + background MTX
133
ACR20 Month 3 Month 6
26%
NAb
59%
69%
27%
25%
55%
50%
24%
NA
41%
51%
ACR50 Month 3 Month 6
12%
NA
31%
42%
8%
9%
29%
32%
8%
NA
26%
37%
ACR70 Month 3 Month 6
6%
NA
15%
22%
3%
1%
11%
14%
2%
NA
14%
16%
In Study RA-IV, a greater proportion of patients treated with tofacitinib 5 mg twice daily plus MTX achieved a low level of disease activity as measured by a DAS28-4(ESR) less than 2.6 at 6 months compared to those treated with MTX alone (Table 11).
Table 11: Proportion and Numbers of Adults with Moderate to Severe Active RA with DAS28-4(ESR) Less Than 2.6 with Number of Residual Active Joints at Month 6 in Study RA-IV
Study RA-IV
DAS28-4(ESR) Less Than 2.6
Placebo + MTX
160
Tofacitinib
5 mg Twice Daily + MTX
321
Proportion of responders at Month 6 (n)
1% (2)
6% (19)
Of responders, proportion with 0 active
joints (n)
50% (1)
42% (8)
Of responders, proportion with 1 active
joint (n)
0
5% (1)
Of responders, proportion with 2 active
joints (n)
0
32% (6)
Of responders, proportion with 3 or more active joints (n)
50% (1)
21% (4)
The results of the components of the ACR response criteria for Study RA-IV are shown in Table 12. Similar results were observed for tofacitinib in Studies RA-I, II, III, V, and VI.
Table 12: Components of ACR Response in Adults with Moderate to Severe Active RA at Baseline and Month 3 in Study RA-IV a Data shown is mean (Standard Deviation) at Month 3.
b Visual analog scale: 0 = best, 100 = worst.
c Health Assessment Questionnaire Disability Index: 0 = best, 3 = worst; 20 questions; categories: dressing and grooming, arising, eating, walking, hygiene, reach, grip, and activities.
Study RA-IV
Placebo + MTX
N=160
Tofacitinib 5 mg
Twice Daily + MTX
N=321
Component (mean)a
Baseline
Month 3a
Baseline
Month 3a
Number of tender joints
(0 to 68)
23
(13)
18
(14)
24
(14)
13
(14)
Number of swollen joints
(0 to 66)
14
(9)
10
(9)
14
(8)
6
(8)
Painb
55
(24)
47
(24)
58
(23)
34
(23)
Patient global assessmentb
54
(23)
47
(24)
58
(24)
35
(23)
Disability index (HAQ-DI)c
1.32
(0.67)
1.19
(0.68)
1.41
(0.68)
0.99
(0.65)
Physician global assessmentb
56
(18)
43
(22)
59
(16)
30
(19)
CRP (mg/L)
13.7
(14.9)
14.6
(18.7)
15.3
(19.0)
7.1
(19.1)
The percent of ACR20 responders by visit for Study RA-IV is shown in Figure 4. Similar responses were observed for tofacitinib in Studies RA-I, II, III, V, and VI.
Figure 4: Percentage of ACR20 Responders by Visit Through Month 6 in Study RA-IV
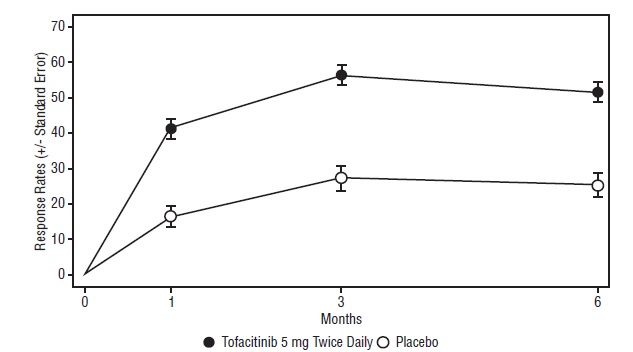
Non-responder imputation was used. Patients who withdrew from the study were counted as failures, as were patients who failed to have at least a 20% improvement in joint counts at Month 3.
Radiographic Response
Two studies were conducted to evaluate the effect of tofacitinib on structural joint damage. In Study RA-IV and Study RA-VI, progression of structural joint damage was assessed radiographically and expressed as change from baseline in mTSS and its components, the erosion score and joint space narrowing score, at Months 6 and 12. The proportion of patients with no radiographic progression (mTSS change less than or equal to 0) was also assessed.
In Study RA-IV, tofacitinib 5 mg twice daily reduced the mean progression of structural damage (not statistically significant) as shown in Table 13. Analyses of erosion and joint space narrowing scores were consistent with the overall results.
In the placebo plus MTX group, 74% of patients experienced no radiographic progression at Month 6 compared to 84% of patients treated with tofacitinib plus MTX 5 mg twice daily.
In Study RA-VI, tofacitinib monotherapy inhibited the progression of structural damage compared to MTX at Months 6 and 12 as shown in Table 13. Analyses of erosion and joint space narrowing scores were consistent with the overall results.
In the MTX group, 55% of patients experienced no radiographic progression at Month 6 compared to 73% of patients treated with tofacitinib 5 mg twice daily.
Table 13: Radiographic Changes in Adults with Moderate to Severe Active RA at Months 6 and 12 in Studies RA-IV and VI a SD = Standard Deviationb Difference between least squares means tofacitinib minus placebo or MTX (95% CI = 95% confidence interval)
c Month 6 and Month 12 data are mean change from baseline.
Study RA-IV
Placebo
N=139
Mean (SD)a
Tofacitinib 5 mg
Twice Daily
N=277
Mean (SD)a
Tofacitinib 5 mg
Twice Daily
Mean Difference from
Placebob (CI)
mTSScBaseline Month 6
33 (42)
0.5 (2.0)
31 (48)
0.1 (1.7)
-
-0.3 (-0.7, 0.0)
Study RA-VI
MTX
N=166
Mean (SD)a
Tofacitinib 5 mg
Twice Daily N=346
Mean (SD)a
Tofacitinib 5 mg Twice Daily Mean Difference from MTXb (CI)
mTSScBaseline Month 6 Month 12
17 (29)
0.8 (2.7)
1.3 (3.7)
20 (40)
0.2 (2.3)
0.4 (3.0)
-
-0.7 (-1.0, -0.3)
-0.9 (-1.4, -0.4)
Physical Function Response
Improvement in physical functioning was measured by the HAQ-DI. Patients who received tofacitinib 5 mg twice daily demonstrated greater improvement from baseline in physical functioning compared to patients who received placebo at Month 3.
The mean (95% CI) difference from placebo in HAQ-DI improvement from baseline at Month 3 in Study RA-III was -0.22 (-0.35, -0.10) in patients who received 5 mg tofacitinib twice daily. Similar results were obtained in Studies RA-I, II, IV and V. In the 12-month trials, HAQ-DI results in tofacitinib-treated patients were consistent at 6 and 12 months.
Other Health-Related Outcomes
General health status was assessed by the Short Form health survey (SF-36). In Studies RA-I, IV, and V, patients who received tofacitinib 5 mg twice daily demonstrated greater improvement from baseline compared to placebo in physical component summary (PCS), mental component summary (MCS) scores and in all 8 domains of the SF-36 at Month 3.
14.2 Clinical Studies in Psoriatic Arthritis
The psoriatic arthritis (PsA) clinical development program with tofacitinib tablets included 2 multicenter, randomized, double-blind, placebo-controlled trials in 816 adults with active PsA (Studies PsA-I and PsA-II).
Trial Designs and Population
All patients had active PsA for at least 6 months based upon the Classification Criteria for Psoriatic Arthritis (CASPAR), at least 3 tender/painful joints and at least 3 swollen joints, and active plaque psoriasis. Patients randomized and treated across the 2 clinical trials represented different PsA subtypes at screening, including <5 joints or asymmetric involvement (21%), ≥5 joints involved (90%), distal interphalangeal (DIP) joint involvement (61%), arthritis mutilans (8%), and spondylitis (19%). Patients in these clinical trials had a diagnosis of PsA for a mean (SD) of 7.7 (7.2) years. At baseline, 80% and 53% of patients had enthesitis and dactylitis, respectively. At baseline, all patients were required to receive treatment with a stable dose of a nonbiologic DMARD (79% received methotrexate, 13% received sulfasalazine, 7% received leflunomide, 1% received other nonbiologic DMARDs). In both clinical trials, the primary endpoints were the ACR20 response and the change from baseline in HAQ-DI at Month 3.
- Study PsA-I was a 12-month clinical trial in 422 patients who had an inadequate response to a nonbiologic DMARD (67% and 33% were inadequate responders to 1 nonbiologic DMARD and ≥2 nonbiologic DMARDs, respectively) and who were naïve to treatment with a TNF blocker. Although Study PsA-1 included patients who are TNF blocker-naïve, tofacitinib tablets and tofacitinib extended-release tablets are not approved for use in TNF blocker-naïve patients [see Indications and Usage (1.2)]. Patients were randomized in a 2:2:2:1:1 ratio to receive tofacitinib 5 mg twice daily, tofacitinib 10 mg twice daily, adalimumab 40 mg subcutaneously once every 2 weeks, placebo to tofacitinib 5 mg twice daily treatment sequence, or placebo to tofacitinib 10 mg twice daily treatment sequence, respectively; study drug was added to background nonbiologic DMARD treatment. At the Month 3 visit, all patients randomized to placebo treatment were switched in a blinded fashion to a predetermined tofacitinib dosage of 5 mg or 10 mg twice daily. Study PsA-I was not designed to demonstrate non-inferiority or superiority to adalimumab.
- Study PsA-II was a 6-month clinical trial in 394 patients who had an inadequate response to at least 1 approved TNF blocker (66%, 19%, and 15% were inadequate responders to 1 TNF blocker, 2 TNF blockers and ≥3 TNF blockers, respectively). Patients were randomized in a 2:2:1:1 ratio to receive tofacitinib 5 mg twice daily, tofacitinib 10 mg twice daily, placebo to tofacitinib 5 mg twice daily treatment sequence, or placebo to tofacitinib 10 mg twice daily treatment sequence, respectively; study drug was added to background nonbiologic DMARD treatment. At the Month 3 visit, placebo patients were switched in a blinded fashion to a predetermined tofacitinib dosage of 5 mg or 10 mg twice daily as in Study PsA-I.
Although other dosages have been studied, the recommended dosage of tofacitinib is 5 mg twice daily. Tofacitinib 10 mg twice daily is not recommended for treatment of PsA [see Dosage and Administration (2.3)].
Clinical Response
At Month 3, patients treated with tofacitinib 5 mg twice daily had higher (p≤0.05) response rates versus placebo for ACR20, ACR50, and ACR70 in Study PsA-I and for ACR20 and ACR50 in Study PsA-II; ACR70 response rates were also higher for tofacitinib 5 mg twice daily versus placebo in Study PsA-II, although the differences versus placebo were not statistically significant (p>0.05) (Tables 14 and 15).
Table 14: Proportion of Adults with Active PsA with an ACR Response at Month 3 in Study PsA-I* [Nonbiologic DMARD Inadequate Responders (TNF Blocker-Naïve)]** Patients with missing data were treated as non-responders.
* Patients received one concomitant nonbiologic DMARD.
** Tofacitinib tablets and tofacitinib extended-release tablets are not approved for use in TNF blocker-naïve patients [see Indications and Usage (1.2)].
a N is number of randomized and treated patients.Treatment
Group
Placebo
Tofacitinib 5 mg Twice Daily +
Background Nonbiologic DMARD
Na
105
107
Response Rate
Response Rate
Difference (%)
95% CI from Placebo
Month 3
ACR20
ACR50
ACR70
33%
10%
5%
50%
28%
17%
17.1 (4.1, 30.2)
18.5 (8.3, 28.7)
12.1 (3.9, 20.2)
Table 15: Proportion of Adults with Active PsA with an ACR Response at Month 3 in Study PsA-II* (TNF Blocker Inadequate Responders)
Patients with missing data were treated as non-responders.
Patients received one concomitant nonbiologic DMARD.
a N is number of randomized and treated patients.Treatment
Group
Placebo
Tofacitinib
5 mg
Twice Daily
Na
131
131
Response Rate
Response Rate
Difference (%)
95% CI from Placebo
Month 3
ACR20
ACR50
ACR70
24%
15%
10%
50%
30%
17%
26.0 (14.7, 37.2)
15.3 (5.4, 25.2)
6.9 (-1.3, 15.1)
Improvements from baseline in the ACR response criteria components for both studies are shown in Table 16.
Table 16: Components of ACR Response in Adults with Active PsA at Baseline and Month 3 in Studies PsA-I and PsA-II * Patients received one concomitant nonbiologic DMARD.
** Tofacitinib tablets and tofacitinib extended-release tablets are not approved for use in TNF blocker-naïve patients [see Indications and Usage (1.2)].
a Data shown are mean value at baseline and at Month 3.
b Visual analog scale (VAS): 0 = best, 100 = worst.
c HAQ-DI = Health Assessment Questionnaire – Disability Index: 0 = best, 3 = worst; 20 questions;
categories: dressing and grooming, arising, eating, walking, hygiene, reach, grip, and activities.
Nonbiologic DMARD
Inadequate Responders
(TNF Blocker-Naïve)
TNF Blocker Inadequate
Responders
Study PsA-I*,**
Study PsA-II*
Treatment Group
Placebo
Tofacitinib
5 mg
Twice Daily
Placebo
Tofacitinib
5 mg
Twice Daily
N at Baseline
105
107
131
131
ACR Componenta
Number of tender/painful
joints (0 to 68)
Baseline
Month 3
20.6
14.6
20.5
12.2
19.8
15.1
20.5
11.5
Number of
swollen joints
(0 to 66)
Baseline
Month 3
11.5
7.1
12.9
6.3
10.5
7.7
12.1
4.8
Patient
assessment of arthritis painb
Baseline
Month 3
53.2
44.7
55.7
34.7
54.9
48.0
56.4
36.1
Patient global assessment of arthritisb
Baseline
Month 3
53.9
44.4
54.7
35.5
55.8
49.2
57.4
36.9
HAQ-DIc
Baseline
Month 3
1.11
0.95
1.16
0.81
1.25
1.09
1.26
0.88
Physician’s
Global Assessment of Arthritisb
Baseline
Month 3
53.8
35.4
54.6
29.5
53.7
36.4
53.5
27.0
CRP (mg/L)
Baseline
Month 3
10.4
8.6
10.5
4.0
12.1
11.4
13.8
7.7
The percentage of ACR20 responders by visit for Study PsA-I is shown in Figure 5. Similar responses were observed in Study PsA-II. In both studies, improvement in ACR20 response on tofacitinib was observed at the first visit after baseline (Week 2).
Figure 5: Percentage of ACR20 Responders by Visit Through Month 3 in Study PsA-I*,**
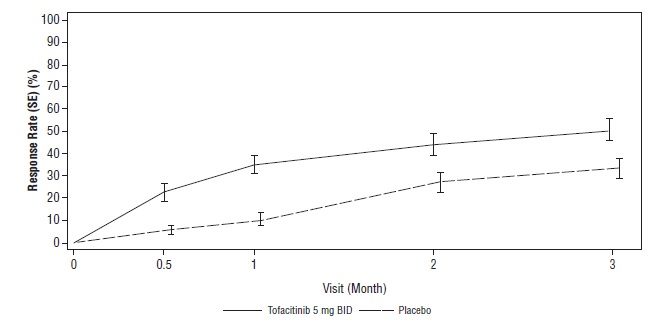
BID = twice daily; SE = standard error.
Patients with missing data were treated as non-responders.
* Subjects received one concomitant nonbiologic DMARD.
** Tofacitinib tablets and tofacitinib extended-release tablets are not approved for use in TNF blocker-naïve patients [see Indications and Usage (1.2)].
In patients with active PsA evidence of benefit in enthesitis and dactylitis was observed with tofacitinib treatment.
Physical Function
Improvement in physical functioning was measured by the HAQ-DI. Patients receiving tofacitinib 5 mg twice daily demonstrated significantly greater improvement (p ≤0.05) from baseline in physical functioning compared to placebo at Month 3 (Table 17).
Table 17: Change from Baseline in HAQ-DI in Adults with Active PsA at Month 3 Studies PsA-I and PsA-II * Patients received one concomitant nonbiologic DMARD.
** Tofacitinib tablets and tofacitinib extended-release tablets are not approved for use in TNF blocker-naïve patients [see Indications and Usage (1.2)].
a N is the total number of patients in the statistical analysis.
b Inadequate response to at least one nonbiologic DMARD due to lack of efficacy and/or intolerability.
c Inadequate response to at least one TNF blocker due to lack of efficacy and/or intolerability.
Least Squares Mean Change from Baseline In HAQ-DI at Month 3
Nonbiologic DMARD Inadequate Respondersb (TNF Blocker-Naïve)
TNF Blocker Inadequate Respondersc
Study PsA-I*,**
Study PsA-II*
Treatment
Group
Placebo
Tofacitinib 5 mg Twice Daily
Placebo
Tofacitinib 5 mg Twice Daily
Na
104
107
131
129
LSM Change from Baseline
-0.18
-0.35
-0.14
-0.39
Difference
from Placebo
(95% CI)
-
-0.17
(-0.29, -0.05)
-
-0.25
(-0.38, -0.13)
In Study PsA-I, the HAQ-DI responder rate (response defined as having improvement from baseline of ≥0.35) at Month 3 was 53% in patients receiving tofacitinib 5 mg twice daily and 31% in patients receiving placebo. Similar responses were observed in Study
PsA-II.
Other Health-Related Outcomes
General health status was assessed by the Short Form health survey (SF-36). In Studies PsA-I and PsA-II, patients receiving tofacitinib 5 mg twice daily had greater improvement from baseline compared to placebo in Physical Component Summary (PCS) score, but not in Mental Component Summary (MCS) score at Month 3. Patients receiving tofacitinib 5 mg twice daily reported consistently greater improvement relative to placebo in the domains of Physical Functioning, Bodily Pain, Vitality, and Social Functioning, but not in Role-Physical, General Health, Role-Emotional, or Mental Health.
Radiographic Response
Treatment effect on inhibition of radiographic progression in PsA could not be established from the results of Study PsA-I.
14.3 Clinical Studies in Ankylosing Spondylitis
The ankylosing spondylitis (AS) clinical development program with tofacitinib tablets included one placebo-controlled trial (Study AS-I) in adults with active AS. Patients had active disease as defined by both Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) and back pain score (BASDAI question 2) of greater or equal to 4 despite non-steroidal anti-inflammatory drug (NSAID), corticosteroid or disease modifying anti-rheumatic drug (DMARD) therapy.
Trial Design
Study AS-I was a randomized, double-blind, placebo-controlled, 48-week clinical trial in 269 adult patients who had an inadequate response (inadequate clinical response or intolerance) to at least 2 NSAIDs. Although Study AS-I included some patients who are TNF blocker-naïve, tofacitinib tablets and tofacitinib extended-release tablets are not approved for use in TNF blocker-naïve patients [see Indications and Usage (1.3)]. Patients were randomized and treated with tofacitinib 5 mg twice daily or placebo for 16 weeks of blinded treatment and then all received treatment of tofacitinib 5 mg twice daily for additional 32 weeks. The primary endpoint was to evaluate the proportion of patients who achieved an ASAS20 response at Week 16.
Approximately 7% and 21% of patients used concomitant methotrexate or sulfasalazine, respectively from baseline to Week 16. Twenty-two percent of patients had an inadequate response to 1 or 2 TNF blockers.
Clinical Response
Patients treated with tofacitinib 5 mg twice daily achieved greater improvements in ASAS20 and ASAS40 responses compared to patients treated with placebo at Week 16 (Table 18). Consistent results were observed in the subgroup of patients who had an inadequate response to TNF blockers for both the ASAS20 (primary endpoint) and ASAS40 (secondary endpoint) at Week 16 (Table 18).
Table 18: ASAS20 and ASAS40 Responses in Adults with Active AS at Week 16 in Study AS-I * type I error-controlled.
** p-value <0.0001.
Placebo
Tofacitinib 5 mg Twice Daily
Difference from Placebo (95% CI)
All patients (N)
N=136
N=133
ASAS20 response*, %
29
56
27 (16, 38)**
ASAS40 response*, %
13
41
28 (18, 38)**
TNFi-IR patients (N)
N=30
N=29
ASAS20 response, %
17
41
25 (2, 47)
ASAS40 response, %
7
28
21 (2, 39)
Abbreviations: CI = confidence interval; TNFi-IR = tumor necrosis factor inhibitor inadequate response.
The improvements in the components of the ASAS response and other measures of disease activity were greater in the tofacitinib 5 mg twice daily group compared to the placebo group as shown in Table 19.
Table 19: ASAS Components and Other Measures of Disease Activity in Adults with Active AS at Week 16 in Study AS-I * type I error-controlled.
** p <0.0001.
a Measured on a numerical rating scale with 0 = not active or no pain, 10 = very active or most severe pain.
b Bath Ankylosing Spondylitis Functional Index measured on a numerical rating scale with 0 = easy and 10 = impossible.
c Inflammation is the mean of two patient-reported stiffness self-assessments in BASDAI.
d BASDAI total score.
e Bath Ankylosing Spondylitis Metrology Index.
f High sensitivity C-reactive protein.
g Estimates are generated based on a mixed model for repeated measures using both on-treatment and off-treatment data.
LSM = least squares mean.
Placebo
(N=136)
Tofacitinib 5 mg Twice Daily
(N=133)
Baseline
(mean)
Week 16
(LSM
change
from
Baseline)g
Baseline
(mean)
Week 16
(LSM
change
from
Baseline)g
Difference from
Placebo
(95% CI)g
ASAS Components
− Patient Global Assessment of Disease Activity (0 to 10)a,*
7.0
-1.0
6.9
-2.5
-1.5 (-2.00, -0.97)**
− Total spinal pain (0 to 10)a,*
6.9
-1.1
6.9
-2.6
-1.5 (-2.00, -1.03)**
− BASFI (0 to 10)b,*
5.9
-0.8
5.8
-2.0
-1.2 (-1.64, -0.79)**
− Inflammation (0 to 10)c,*
6.8
-1.1
6.6
-2.8
-1.7 (-2.13, -1.18)**
BASDAI Scored
6.5
-1.2
6.4
-2.6
-1.4 (-1.86, -0.98)**
BASMIe,*
4.4
-0.1
4.5
-0.6
-0.5 (-0.66, -0.36)**
hsCRPf,* (mg/dL)
1.8
-0.1
1.6
-1.1
-0.9 (-1.17, -0.69)**
The percentage of patients with active AS who achieved ASAS20 response by visit is shown in Figure 6.
Figure 6: Percentage of ASAS20 Responders Over Time Up to Week 16 in Patients with Active AS in Study AS-I
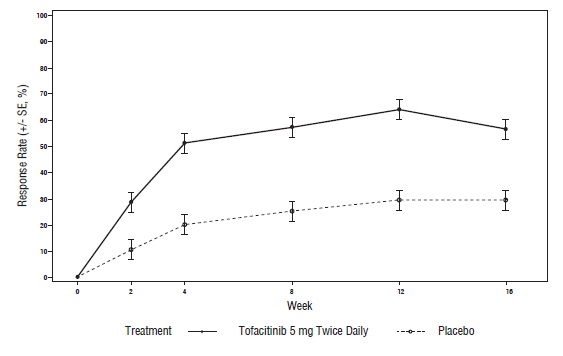
SE=standard error.
Patients with missing data were treated as non-responders.
Other Health-Related Outcomes
Patients treated with tofacitinib 5 mg twice daily achieved greater improvements from baseline in Ankylosing Spondylitis Quality of Life (ASQoL) (-4.0 vs -2.0) compared to patients treated with placebo at Week 16.
14.6 Safety Study in Adults with Rheumatoid Arthritis (Tofacitinib Versus TNF-blocker)
A randomized open-label trial (RA Safety Study 1; NCT02092467) was conducted to evaluate safety with tofacitinib tablets at two doses, 5 mg twice daily (N=1455) and 10 mg twice daily (N=1456), versus the TNF-blocker control (N=1451) in RA patients 50 years of age and older with at least one cardiovascular risk factor. The co-primary endpoints were adjudicated MACE (defined as cardiovascular death, non-fatal MI, and non-fatal stroke) and adjudicated malignancy (excluding non-melanoma skin cancer). The study was designed to exclude a prespecified risk margin of 1.8 for the hazard ratio of combined tofacitinib regimens versus the TNF-blocker control for each co-primary endpoint. An independent committee conducted a blinded evaluation of the co-primary endpoints according to predefined criteria (adjudication). The study was event-driven and patients were followed until a sufficient number of primary outcome events accrued. Other endpoints included mortality, serious infections, and thromboembolic events. The median on-study follow-up time was 4 years.
The mean age of the population was 61 years (range: 50 to 88 years). Most patients were female (78%) and Caucasian (77%). Patients had a diagnosis of RA for a mean of 10 years, and a median swollen and tender joint count of 11 and 15 respectively. Cardiovascular risk factors included cigarette smoking (current or past) (48%), hypertension (66%), high density lipoprotein <40 mg/dL (12%), diabetes mellitus (17%), family history of premature coronary heart disease (15%), extra-articular disease associated with RA (37%), and history of coronary artery disease (11%).
The non-inferiority criterion was not met for the primary comparison of the combined tofacitinib dosages to TNF blockers since the upper limit of the 95% CI exceeded the pre-specified non-inferiority criterion of 1.8 (for MACE, the upper limit of the 95% CI was 1.94; for malignancies excluding NMSC, the upper limit of the 95% CI was 2.09).
Table 22 shows the study results for each of the co-primary endpoints, and other endpoints. There was an increased risk of death, MACE, malignancies, serious infections, thromboembolic events, and fractures associated with both dosages of tofacitinib.
Table 22: Results of RA Safety Study 1 in Adults with Rheumatoid Arthritis 50 years of Age and Older with at Least One Cardiovascular Risk Factor Note: Tofacitinib 10 mg twice daily was discontinued by the Data Monitoring Committee due to safety concerns, and ongoing patients switched from tofacitinib 10 mg to tofacitinib 5 mg. The column “tofacitinib 10 mg Twice Daily” includes all events and follow-up for patients randomized to tofacitinib 10 mg twice daily. A tofacitinib tablets 10 mg twice daily (or a tofacitinib extended-release tablets 22 mg once daily) dosage is not recommended for the treatment of RA, PsA, or AS [see Dosage and Administration (2.3)].
N indicates number of patients; n indicates number of patients with events.
IR indicates incidence rate per 100 person-year (PY).
†MI and Stroke include fatal and non-fatal events.
††Data and analyses for Malignancies excluding NMSC for current and ex-smokers are included. There were 720 current and ex-smokers randomized to tofacitinib 5 mg, 704 to tofacitinib 10 mg, and 679 to TNF blockers.
*HR (95%) CI for tofacitinib vs. TNF Blocker (Univariate Cox Proportional Hazard Model).
NMSC: Non-melanoma Skin Cancer; MACE: Major Adverse Cardiac Events; HR: Hazard Ratio; DVT: Deep Vein Thrombosis; PE: Pulmonary Embolism; VTE: Venous Thromboembolism, first occurrence of a VTE, defined as the composite of adjudicated DVT and adjudicated PE; ATE: Arterial Thromboembolism; TE: Thromboembolism, first occurrence of a TE, defined as the composite of adjudicated VTE and unadjudicated ATE.Endpoint
TNF Blocker
N=1451
PY=5526
Tofacitinib
5 mg
Twice Daily
N=1455
PY=5551
Tofacitinib
10 mg
Twice Daily
N=1456
PY=5371
MACE, n [IR]
HR (95% CI)*
43 [0.79]
50 [0.91]
1.16 (0.77, 1.74)
59 [1.11]
1.41 (0.95, 2.10)
MI,† n [IR]
HR (95% CI)*
11 [0.20]
20 [0.36]
1.81 (0.87, 3.79)
21 [0.39]
1.97 (0.95, 4.09)
Stroke,† n [IR]
HR (95% CI)*
20 [0.36]
18 [0.33]
0.89 (0.47, 1.69)
21 [0.39]
1.08 (0.59, 2.00)
Cardiovascular Death, n [IR]
HR (95% CI)*
15 [0.27]
18 [0.32]
1.20 (0.60, 2.37)
25 [0.47]
1.71 (0.90, 3.24)
Malignancies Excl. NMSC, n [IR]
HR (95% CI)*
42 [0.77]
62 [1.13]
1.47 (1.00, 2.18)
60 [1.13]
1.48 (1.00, 2.19)
Malignancies Excl. NMSC
(among current and past smokers)††
HR (95% CI)*
25 [0.99]
41 [1.53]
1.55 (0.94, 2.55)
48 [1.91]
1.94 (1.19, 3.14)
All Death
HR (95% CI)*
38 [0.69]
49 [0.88]
1.29 (0.84, 1.96)
66 [1.23]
1.79 (1.20, 2.66)
Serious Infections
HR (95% CI)*
133 [2.52]
155 [2.95]
1.17 (0.93, 1.47)
184 [3.65]
1.44 (1.15, 1.80)
DVT
HR (95% CI)*
9 [0.16]
12 [0.22]
1.33 (0.56, 3.15)
15 [0.28]
1.72 (0.75, 3.92)
PE
HR (95% CI)*
3 [0.05]
10 [0.18]
3.32 (0.91, 12.08)
26 [0.49]
8.95 (2.71, 29.56)
VTE
HR (95% CI)*
12 [0.22]
18 [0.33]
1.50 (0.72, 3.10)
36 [0.68]
3.10 (1.61, 5.96)
ATE
HR (95% CI)*
45 [0.83]
51 [0.93]
1.13 (0.76, 1.69)
55 [1.04]
1.26 (0.85, 1.87)
TE
HR (95% CI)*
56 [1.03]
67 [1.23]
1.19 (0.84, 1.70)
86 [1.65]
1.60 (1.14, 2.23)
All Fractures
HR (95% CI)*
121 [2.32]
138 [2.64]
1.14 (0.90, 1.46)
141 [2.80]
1.21 (0.95, 1.54)
Osteoporotic Fractures
HR (95% CI)*
49 [0.91]
58 [1.07]
1.18 (0.81, 1.73)
70 [1.34]
1.48 (1.03, 2.13)
Lymphomas and lung cancers, which are a subset of all malignancies in RA Safety Study 1, were observed at a higher rate in patients treated with tofacitinib 5 mg twice a day and tofacitinib 10 mg twice a day compared to those treated with TNF blockers. Lymphoma was reported for 4 patients who received tofacitinib 5 mg twice a day, 6 patients who received tofacitinib 10 mg twice a day, and 1 patient who received TNF blockers (Incidence Rate [IR] of 0.07, 0.11, and 0.02 per 100 patient-years, respectively). Among current and past smokers, lung cancer was reported for 13 patients who received tofacitinib 5 mg twice a day, 15 patients who received tofacitinib 10 mg twice a day, and 7 patients who received TNF blockers (IR of 0.48, 0.59, and 0.27 per 100 patient-years, respectively).
Given these increased risks, tofacitinib tablets 10 mg twice daily (or tofacitinib extended-release tablets 22 mg once daily) dosages are not recommended for the treatment of RA, PsA, or AS [see Dosage and Administration (2.3)].
The overall fractures and osteoporotic fractures were observed at a higher rate in both tofacitinib treatment groups compared to the TNF blocker treatment group. The observed incidence rate of osteoporotic fractures was higher in women than men, and was highest in women on tofacitinib 10 mg twice daily (1.56 per 100 patient-years), followed by tofacitinib 5 mg twice daily (1.26 per 100 patient-years), and TNF blockers (1.01 per 100 patient-years).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Tofacitinib Extended-Release Tablets 11 mg are pink, oval, bevel edged, mat finished film-coated extended-release tablets imprinted with “TI11” on one side and plain on other side.
Bottles of 30 NDC: 59651-685-30
Store at 20º to 25ºC (68º to 77ºF); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
Do not repackage. -
17 PATIENT COUNSELING INFORMATION
Advise patients to read the FDA-approved patient labeling (Medication Guide).
Serious Infections
Inform patients that tofacitinib extended-release tablets may lower the ability of their immune system to fight infections. Advise patients not to start taking tofacitinib extended-release tablets if they have an active infection. Instruct patients to contact their healthcare provider immediately during treatment if symptoms suggesting infection appear to ensure rapid evaluation and appropriate treatment [see Warnings and Precautions (5.1)].
Advise patients that the risk of herpes zoster, some cases of which can be serious, is increased in patients treated with tofacitinib extended-release tablets [see Warnings and Precautions (5.1)].
Malignancies and Lymphoproliferative Disorders
Inform patients that tofacitinib extended-release tablets may increase their risk of certain cancers, and that lymphoma and other cancers have been observed in patients taking tofacitinib. Instruct patients to inform their healthcare provider if they have ever had any type of cancer [see Warnings and Precautions (5.3)].
Major Adverse Cardiovascular Events
Inform patients that tofacitinib extended-release tablets may increase their risk of major adverse cardiovascular events (MACE) defined as myocardial infarction, stroke, and cardiovascular death. Instruct all patients, especially current or past smokers or patients with other cardiovascular risk factors, to be alert for the development of signs and symptoms of cardiovascular events [see Warnings and Precautions (5.4)].
Thrombosis
Advise patients to stop taking tofacitinib extended-release tablets and to call their healthcare provider right away if they experience any symptoms of thrombosis (sudden shortness of breath, chest pain worsened with breathing, swelling of leg or arm, leg pain or tenderness, red or discolored skin in the affected leg or arm) [see Warnings and Precautions (5.5)].
Hypersensitivity
Advise patients to stop taking tofacitinib extended-release tablets and to call their healthcare provider right away if they experience any symptoms of allergic reactions while taking tofacitinib extended-release tablets [see Warnings and Precautions (5.7)].
Important Information on Laboratory Abnormalities
Inform patients that tofacitinib extended-release tablets may affect certain lab test results, and that blood tests are required before and during tofacitinib extended-release tablets treatment [see Warnings and Precautions (5.8)].
Pregnancy
Advise pregnant females and females of reproductive potential of the potential risk to a fetus. Advise females to inform their prescriber of a known or suspected pregnancy [see Use in Specific Populations (8.1)].
Lactation
Advise women not to breastfeed during treatment with tofacitinib extended-release tablets and for at least 36 hours after the last dose of tofacitinib extended-release tablets [see Use in Specific Populations (8.2)].
Infertility
Advise females of reproductive potential that tofacitinib extended-release tablets may impair fertility [see Use in Specific Populations (8.3), Nonclinical Toxicology (13.1)]. It is not known if this effect is reversible.
Residual Tablet Shell
Patients receiving tofacitinib extended-release tablets may notice an inert tablet shell passing in the stool or via colostomy. Patients should be informed that the active medication has already been absorbed by the time the patient sees the inert tablet shell.
Distributed by:Aurobindo Pharma USA, Inc.
279 Princeton-Hightstown Road
East Windsor, NJ 08520
Manufactured by:
Aurobindo Pharma Limited
Hyderabad-500 032, India
Revised: 03/2026 -
MEDICATION GUIDE
MEDICATION GUIDE
Tofacitinib (toe″ fa sye′ ti nib)
Extended-Release Tablets, for oral useWhat is the most important information I should know about tofacitinib extended-release tablets?
Tofacitinib extended-release tablets may cause serious side effects including:
1. Serious infections. Tofacitinib extended-release tablets are medicines that affect your immune system. Tofacitinib extended-release tablets can lower the ability of your immune system to fight infections. Some people can have serious infections while taking tofacitinib extended-release tablets including tuberculosis (TB), and infections caused by bacteria, fungi, or viruses that can spread throughout the body. Some people have died from these infections.
- Your healthcare provider should test you for TB before starting tofacitinib extended-release tablets and during treatment.
- Your healthcare provider should monitor you closely for signs and symptoms of TB infection during treatment with tofacitinib extended-release tablets.
You should not start taking tofacitinib extended-release tablets if you have any kind of infection unless your healthcare provider tells you it is okay. You may be at a higher risk of developing shingles (herpes zoster).
Before starting tofacitinib extended-release tablets, tell your healthcare provider if you:
- think you have an infection or have symptoms of an infection such as:
- fever, sweating, or chills
- cough
- blood in phlegm
- warm, red, or painful skin or sores on your body
- burning when you urinate or urinating more often than normal
- muscle aches
- shortness of breath
- weight loss
- diarrhea or stomach pain
- feeling very tired
- are being treated for an infection.
- get a lot of infections or have infections that keep coming back.
- have diabetes, chronic lung disease, HIV, or a weak immune system. People with these conditions have a higher chance for infections.
- have TB, or have been in close contact with someone with TB.
- live or have lived, or have traveled to certain parts of the country (such as the Ohio and Mississippi River valleys and the Southwest) where there is an increased chance for getting certain kinds of fungal infections (histoplasmosis, coccidioidomycosis, or blastomycosis). These infections may happen or become more severe if you take tofacitinib extended-release tablets. Ask your healthcare provider if you do not know if you have lived in an area where these infections are common.
- have or have had hepatitis B or C.
After starting tofacitinib extended-release tablets, call your healthcare provider right away if you have any symptoms of an infection. Tofacitinib extended-release tablets can make you more likely to get infections or make worse any infection that you have.
2. Increased risk of death in people 50 years of age and older who have at least 1 heart disease (cardiovascular) risk factor and are taking tofacitinib tablets 5 mg or 10 mg twice daily.
3. Cancer and immune system problems. Tofacitinib extended-release tablets may increase your risk of certain cancers by changing the way your immune system works.
- Lymphoma and other cancers including skin cancers can happen in people taking tofacitinib extended-release tablets. People taking tofacitinib tablets 5 mg twice daily or tofacitinib tablets 10 mg twice daily have a higher risk of certain cancers including lymphoma and lung cancer, especially if you are a current or past smoker.
4. Increased risk of major cardiovascular events such as heart attack, stroke or death in people 50 years of age and older who have at least 1 heart disease (cardiovascular) risk factor and are taking tofacitinib tablets 5 mg or 10 mg twice daily, especially if you are a current or past smoker.
Get emergency help right away if you have any symptoms of a heart attack or stroke while taking tofacitinib extended-release tablets, including:
- discomfort in the center of your chest that lasts for more than a few minutes, or that goes away and comes back
- severe tightness, pain, pressure, or heaviness in your chest, throat, neck, or jaw
- pain or discomfort in your arms, back, neck, jaw, or stomach
- shortness of breath with or without chest discomfort
- breaking out in a cold sweat
- nausea or vomiting
- feeling lightheaded
- weakness in one part or on one side of your body
- slurred speech
5. Blood clots in the lungs, veins of the legs or arms, and arteries. Blood clots in the lungs (pulmonary embolism, PE), veins of the legs (deep vein thrombosis, DVT) and arteries (arterial thrombosis) have happened more often in people who are 50 years of age and older and with at least 1 heart disease (cardiovascular) risk factor taking tofacitinib tablets 5 mg or 10 mg twice daily. Some people have died from these blood clots.
- Stop taking tofacitinib extended-release tablets and tell your healthcare provider right away if you develop signs and symptoms of a blood clot, such as sudden shortness of breath or difficulty breathing, chest pain, swelling of the leg or arm, leg pain or tenderness, or redness or discoloration in the leg or arm.
6. Tears (perforation) in the stomach or intestines.
- Tell your healthcare provider if you have had diverticulitis (inflammation in parts of the large intestine) or ulcers in your stomach or intestines. Some people taking tofacitinib extended-release tablets can get tears in their stomach or intestines. This happens most often in people who also take nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, or methotrexate.
Tell your healthcare provider right away if you have fever and stomach-area pain that does not go away, and a change in your bowel habits.
7. Allergic reactions.
- Symptoms such as swelling of your lips, tongue, or throat, or hives (raised, red patches of skin that are often very itchy) that may mean you are having an allergic reaction have been seen in people taking tofacitinib extended-release tablets. Some of these reactions were serious. If any of these symptoms occur while you are taking tofacitinib extended-release tablets, stop tofacitinib extended-release tablets and call your healthcare provider right away.
8. Changes in certain laboratory test results. Your healthcare provider should do blood tests before you start taking tofacitinib extended-release tablets and while you take tofacitinib extended-release tablets to check for the following side effects:
- changes in lymphocyte counts. Lymphocytes are white blood cells that help the body fight off infections.
- low neutrophil counts. Neutrophils are white blood cells that help the body fight off infections.
- low red blood cell count. This may mean that you have anemia, which may make you feel weak and tired.
Your healthcare provider should routinely check certain liver tests.
You should not take tofacitinib extended-release tablets if your lymphocyte count, neutrophil count, or red blood cell count is too low or your liver tests are too high.
Your healthcare provider may stop your tofacitinib extended-release tablets treatment for a period of time if needed because of changes in these blood test results.
You may also have changes in other laboratory tests, such as your blood cholesterol levels. Your healthcare provider should do blood tests to check your cholesterol levels 4 to 8 weeks after you start taking tofacitinib extended-release tablets, and as needed after that. Normal cholesterol levels are important to good heart health.
See “What are the possible side effects of tofacitinib extended-release tablets?” for more information about side effects.
What are tofacitinib extended-release tablets?
- Tofacitinib extended-release tablets are a prescription medicine called a Janus kinase (JAK) inhibitor. Tofacitinib extended-release tablets are used to treat adults with moderately to severely active rheumatoid arthritis when 1 or more medicines called tumor necrosis factor (TNF) blockers have been used and did not work well or cannot be tolerated.
- Tofacitinib extended-release tablets are used to treat adults with active psoriatic arthritis when 1 or more TNF blocker medicines have been used, and did not work well or cannot be tolerated.
- Tofacitinib extended-release tablets are used to treat adults with active ankylosing spondylitis when 1 or more TNF blocker medicines have been used and did not work well or cannot be tolerated.
Tofacitinib extended-release tablets are not recommended for people with severe liver problems.
It is not known if tofacitinib extended-release tablets are safe and effective in children.
What should I tell my healthcare provider before taking tofacitinib extended-release tablets?
Before taking tofacitinib extended-release tablets, tell your healthcare provider about all of your medical conditions, including if you:
- have an infection. See “What is the most important information I should know about tofacitinib extended-release tablets?”
- are a current or past smoker.
- have had any type of cancer.
- have had a heart attack, other heart problems or stroke.
- have had blood clots in the veins of your legs, arms, or lungs, or clots in the arteries in the past.
- have liver problems.
- have kidney problems.
- have any stomach-area (abdominal) pain or been diagnosed with diverticulitis or ulcers in your stomach or intestines.
- have had a reaction to tofacitinib or any of the ingredients in tofacitinib extended-release tablets.
- have recently received or are scheduled to receive a vaccine. People who take tofacitinib extended-release tablets should not receive live vaccines. People taking tofacitinib extended-release tablets can receive non-live vaccines.
- plan to become pregnant or are pregnant. Tofacitinib extended-release tablets may affect the ability of females to get pregnant. It is not known if this will change after stopping tofacitinib extended-release tablets. It is not known if tofacitinib extended-release tablets will harm an unborn baby.
- plan to breastfeed or are breastfeeding. You and your healthcare provider should decide if you will take tofacitinib extended-release tablets or breastfeed. You should not do both. After you stop your treatment with tofacitinib extended-release tablets do not start breastfeeding again until:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Tofacitinib extended-release tablets and other medicines may affect each other causing side effects.
Especially tell your healthcare provider if you take:
- any other medicines to treat your rheumatoid arthritis, psoriatic arthritis, or ankylosing spondylitis. You should not take tocilizumab (Actemra), etanercept (Enbrel), adalimumab (Humira), infliximab (Remicade), rituximab (Rituxan), abatacept (Orencia), anakinra (Kineret), certolizumab (Cimzia), golimumab (Simponi), ustekinumab (Stelara), secukinumab (Cosentyx), vedolizumab (Entyvio), ixekizumab (Taltz), azathioprine, cyclosporine, or other immunosuppressive drugs while you are taking tofacitinib extended-release tablets. Taking tofacitinib extended-release tablets with these medicines may increase your risk of infection.
- medicines that affect the way certain liver enzymes work. Ask your healthcare provider if you are not sure if your medicine is one of these.
How should I take tofacitinib extended-release tablets?
Take tofacitinib extended-release tablets exactly as your healthcare provider tells you to take it.
- Take tofacitinib extended-release tablets 1 time a day with or without food.
- Swallow tofacitinib extended-release tablets whole and intact. Do not crush, split, or chew.
- When you take tofacitinib extended-release tablets, you may see something in your stool that looks like a tablet. This is the empty shell from the tablet after the medicine has been absorbed by your body.
- If you take too much tofacitinib, call your healthcare provider or go to the nearest hospital emergency room right away.
- For the treatment of psoriatic arthritis, take tofacitinib extended-release tablets in combination with methotrexate, sulfasalazine or leflunomide as instructed by your healthcare provider.
- Tofacitinib extended-release tablets should not be used instead of tofacitinib oral solution.
What are the possible side effects of tofacitinib extended-release tablets?
Tofacitinib extended-release tablets may cause serious side effects, including:
- See “What is the most important information I should know about tofacitinib extended-release tablets?”
-
Hepatitis B or C activation infection in people who carry the virus in their blood. If you are a carrier of the hepatitis B or C virus (viruses that affect the liver), the virus may become active while you use tofacitinib extended-release tablets. Your healthcare provider may do blood tests before you start treatment with tofacitinib extended-release tablets and while you are taking tofacitinib extended-release tablets. Tell your healthcare provider if you have any of the following symptoms of a possible hepatitis B or C infection:
- feel very tired
- little or no appetite
- clay-colored bowel movements
- chills
- muscle aches
- skin rash
- skin or eyes look yellow
- vomiting
- fevers
- stomach discomfort
- dark urine
Common side effects of tofacitinib extended-release tablets in people with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis include:
- upper respiratory tract infections (common cold, sinus infections)
- headache
- diarrhea
- nasal congestion, sore throat, and runny nose (nasopharyngitis)
- high blood pressure (hypertension)
- acne
These are not all the possible side effects of tofacitinib extended-release tablets. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
You may also report side effects to Aurobindo Pharma USA, Inc. at 1-866-850-2876.
How should I store tofacitinib extended-release tablets?
- Store tofacitinib extended-release tablets at room temperature between 20° to 25°C (68° to 77°F).
General information about the safe and effective use of tofacitinib extended-release tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use tofacitinib extended-release tablets for a condition for which it was not prescribed. Do not give tofacitinib extended-release tablets to other people, even if they have the same symptoms you have. They may harm them.
This Medication Guide summarizes the most important information about tofacitinib extended-release tablets. If you would like more information, talk to your healthcare provider. You can ask your pharmacist or healthcare provider for information about tofacitinib extended-release tablets that is written for health professionals.
What are the ingredients in tofacitinib extended-release tablets 11 mg?
Active ingredient: tofacitinib citrate
Inactive ingredients: butylated hydroxytoluene, cellulose acetate, colloidal silicon dioxide, copovidone, hydroxyethyl cellulose, hydroxypropyl cellulose, hypromellose, iron oxide red, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene oxide, titanium dioxide, triacetin. Printing ink contains iron oxide black, propylene glycol, and shellac glaze.
Distributed by:
Aurobindo Pharma USA, Inc.
279 Princeton-Hightstown Road
East Windsor, NJ 08520
Manufactured by:
Aurobindo Pharma Limited
Hyderabad-500 032, India
The brands listed are trademarks of their respective owners and are not trademarks of Aurobindo Pharma Limited.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: 03/2026
-
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 11 mg - (30 Tablets Bottle)
NDC: 59651-685-30
Tofacitinib
Extended-Release Tablets
11 mg*
Pharmacist: Dispense the Medication Guide
provided separately to each patient.
Rx only 30 Tablets
AUROBINDO

-
INGREDIENTS AND APPEARANCE
TOFACITINIB
tofacitinib tablet, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59651-685 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TOFACITINIB CITRATE (UNII: O1FF4DIV0D) (TOFACITINIB - UNII:87LA6FU830) TOFACITINIB 11 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) CELLULOSE ACETATE (UNII: 3J2P07GVB6) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) COPOVIDONE K25-31 (UNII: D9C330MD8B) HYDROXYETHYL CELLULOSE (140 MPA.S AT 5%) (UNII: 8136Y38GY5) HYDROXYPROPYL CELLULOSE (110000 WAMW) (UNII: 5Y0974F5PW) HYPROMELLOSE 2208 (4000 MPA.S) (UNII: 39J80LT57T) HYPROMELLOSE 2910 (15 MPA.S) (UNII: 36SFW2JZ0W) FERRIC OXIDE RED (UNII: 1K09F3G675) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE OXIDE 200000 (UNII: 11628IH70O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) FERROSOFERRIC OXIDE (UNII: XM0M87F357) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) Product Characteristics Color PINK Score no score Shape OVAL (bevel edged) Size 11mm Flavor Imprint Code TI11 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59651-685-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 05/14/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA218462 05/14/2026 Labeler - Aurobindo Pharma Limited (650082092) Establishment Name Address ID/FEI Business Operations Aurobindo Pharma Limited 650381903 ANALYSIS(59651-685) , MANUFACTURE(59651-685)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.