SPIKEVAX by Catalent Indiana, LLC
SPIKEVAX by
Drug Labeling and Warnings
SPIKEVAX by is a Other medication manufactured, distributed, or labeled by Catalent Indiana, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
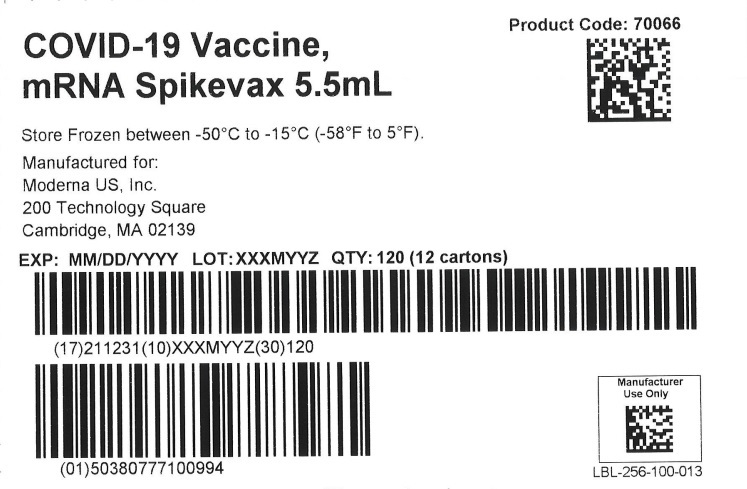
SPIKEVAX- covid-19 vaccine, mrna injection, suspension
Catalent Indiana, LLC
----------
| SPIKEVAX
covid-19 vaccine, mrna injection, suspension |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Catalent Indiana, LLC (172209277) |
| Registrant - ModernaTX, Inc. (116912313) |
Revised: 5/2022
Document Id: 05780332-1acb-4683-9849-d0cf5faa7a67
Set id: e55541c4-76f8-4425-a937-97844d4701d9
Version: 3
Effective Time: 20220511
Trademark Results [SPIKEVAX]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 SPIKEVAX 97037862 not registered Live/Pending |
ModernaTx, Inc. 2021-09-21 |
 SPIKEVAX 90247367 not registered Live/Pending |
ModernaTx, Inc. 2020-10-11 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.