Advanced Skin Lightening Cream
Advanced Skin Lightening Cream by
Drug Labeling and Warnings
Advanced Skin Lightening Cream by is a Otc medication manufactured, distributed, or labeled by Vivier Pharma, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
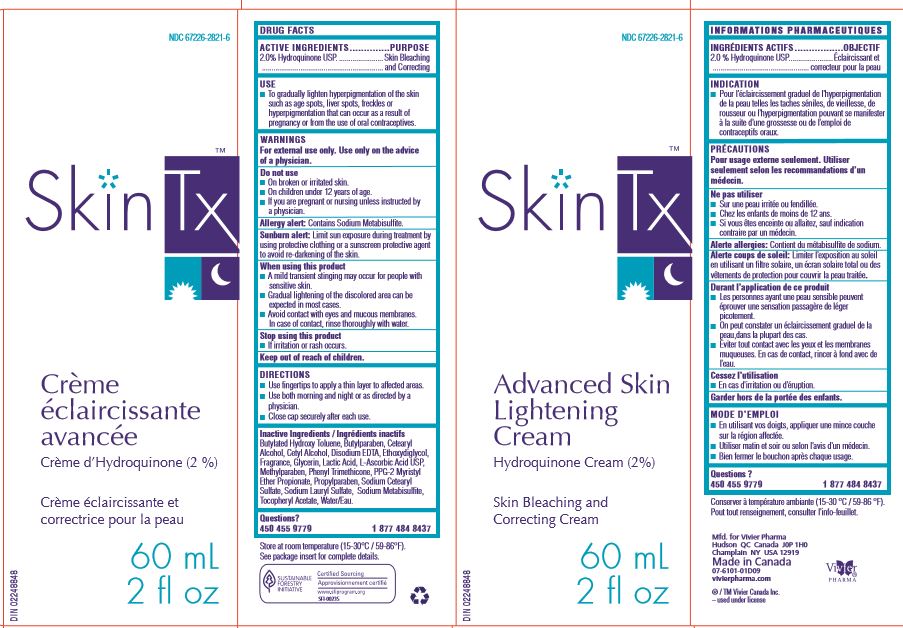
ADVANCED SKIN LIGHTENING CREAM- hydroquinone lotion
Vivier Pharma, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Advanced Skin Lightening Cream
INDICATIONS & USAGE
To gradually lighten hyperpigmentation of the skin such as age spots, liver spots, freckles or hyperpigmentation that can occur as a result of pregnancy or from the use of oral contraceptives.
DOSAGE & ADMINISTRATION
Use fingertips to apply a thin layer to the affected areas. Use both morning and night or as directed by a physician.
DESCRIPTION
Each gram of Advanced Skin Lightening Cream contains 20 mg/g Hydroquinone USP in a nonmedicinal cream base of Butylated Hydroxy Toluene, Butylparaben, Cetearyl Alcohol, Cetyl Alcohol, Disodium EDTA, Ethoxydiglycol, Fragrance, Glycerin, Lactic Acid, L-Ascorbic Acid USP, Methylparaben, Phenyl Trimethicone, PPG-2 Myristyl Ether Propionate, Propylparaben, Sodium Cetearyl Sulfate, Sodium Lauryl Sulfate, Sodium Metabisulfite, Tocopheryl Acetate, Water/Eau.
CONTRAINDICATIONS
Prior history of sensitivity or allergic reaction to this product or any of its ingredients. The safety of topical Hydroquinone use during pregnancy or in children (12 years and under) has not been established.
WARNINGS AND PRECAUTIONS
For external use only. Use only on the advice of a physician. A mild transient stinging may occur for people with sensitive skin. Do not use on broken or irritated skin. Discontinue use if irritation or rash occurs. Avoid contact with eyes and mucous membranes. In case of contact, rinse thoroughly with water. Do not use on children under 12 years of age. Keep out of reach of children. Avoid usage around the eyes and lips. Contains Sodium Metabisulfite, a sulfite that may cause allergic-type reactions (hives, itching, wheezing, anaphylaxis, severe asthma attacks) in certain susceptible persons. Do not use this product if you are pregnant or nursing unless instructed by a physician.
DIRECTIONS
Test for skin sensitivity before using by applying Advanced Skin Lightening Cream to an area of unbroken skin about 25 mm in diameter. If excessive irritation, rash or other reaction develops within 24 hours, discontinue use. Use fingertips to apply a thin layer to the affected areas. Use both morning and night or as directed by a physician. Depigmentation is a gradual process and results should be expected within 12 weeks of daily use. To maintain results, use several times a week. If no improvement is seen within 8 weeks of regular use, discontinue use. Limit sun exposure during treatment by using protective clothing or a sunscreen protective agent to avoid re-darkening of the skin. Close cap securely after each use. Store at room temperature (15-30°C / 59-86°F).
INACTIVE INGREDIENTS
Butylated Hydroxy Toluene, Butylparaben, Cetearyl Alcohol, Cetyl Alcohol, Disodium EDTA, Ethoxydiglycol, Fragrance, Glycerin, Lactic Acid, L-Ascorbic Acid USP, Methylparaben, Phenyl Trimethicone, PPG-2 Myristyl Ether Propionate, Propylparaben, Sodium Cetearyl Sulfate, Sodium Lauryl Sulfate, Sodium Metabisulfite, Tocopheryl Acetate, Water/Eau.
| ADVANCED SKIN LIGHTENING CREAM
hydroquinone lotion |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Vivier Pharma, Inc. (250996550) |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.