CASPOFUNGIN ACETATE injection, powder, lyophilized, for solution
Caspofungin Acetate by
Drug Labeling and Warnings
Caspofungin Acetate by is a Prescription medication manufactured, distributed, or labeled by Fresenius Kabi USA, LLC , Fresenius Kabi USA, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use CASPOFUNGIN ACETATE FOR INJECTION safely and effectively. See full prescribing information for CASPOFUNGIN ACETATE FOR INJECTION.
CASPOFUNGIN ACETATE for injection, for intravenous use
Initial U.S. Approval: 2001
INDICATIONS AND USAGE
Caspofungin acetate for injection is an echinocandin antifungal indicated in adults and pediatric patients (3 months of age and older) for:
- Empirical therapy for presumed fungal infections in febrile, neutropenic patients. (1)
- Treatment of candidemia and the following Candida infections: intra-abdominal abscesses, peritonitis and pleural space infections. (1)
- Treatment of esophageal candidiasis. (1)
- Treatment of invasive aspergillosis in patients who are refractory to or intolerant of other therapies. (1)
DOSAGE AND ADMINISTRATION
Important Administration Instructions for All Patients (2.1):
- Administer by slow intravenous (IV) infusion over approximately 1 hour. Do not administer by intravenous (IV) bolus administration.
- Do not mix or co-infuse caspofungin acetate for injection with other medications. Do not use diluents containing dextrose (α-D-glucose).
Dosage in Adults [18 years of age and older] (2.2):
- Administer a single 70 mg loading dose on Day 1, followed by 50 mg once daily for all indications except esophageal candidiasis.
- For esophageal candidiasis, use 50 mg once daily with no loading dose.
Dosage in Pediatric Patients [3 months to 17 years of age] (2.3):
- Dosing should be based on the patient’s body surface area.
- For all indications, administer a single 70 mg/m2 loading dose on Day 1, followed by 50 mg/m2 once daily thereafter.
- Maximum loading dose and daily maintenance dose should not exceed 70 mg, regardless of the patient's calculated dose.
Dosage Adjustments in Patients with Hepatic Impairment (2.4):
Reduce dosage for adult patients with moderate hepatic impairment (35 mg once daily, with a 70 mg loading dose on Day 1 where appropriate).
Dosage Adjustments in Patients Receiving Concomitant Inducers of Hepatic CYP Enzymes (2.5):
- Use 70 mg once daily dose for adult patients on rifampin.
- Consider dose increase to 70 mg once daily for adult patients on nevirapine, efavirenz, carbamazepine, dexamethasone, or phenytoin.
- Pediatric patients receiving these same concomitant medications may also require an increase in dose to 70 mg/m2 once daily (maximum daily dose not to exceed 70 mg).
DOSAGE FORMS AND STRENGTHS
For Injection: 50 or 70 mg lyophilized powder (plus allowance for overfill) in a single-dose vial for reconstitution. (3)
CONTRAINDICATIONS
Caspofungin is contraindicated in patients with known hypersensitivity to any component of this product. (4)
WARNINGS AND PRECAUTIONS
- Hypersensitivity: Anaphylaxis has been reported. If this occurs, discontinue caspofungin and administer appropriate treatment. Possible histamine-mediated adverse reactions, including rash, facial swelling, angioedema, pruritus, sensation of warmth or bronchospasm have been reported and may require discontinuation and/or administration of appropriate treatment. (5.1)
- Hepatic Effects: Can cause abnormalities in liver enzymes. Isolated cases of hepatic dysfunction, hepatitis, or hepatic failure have been reported. Monitor patients who develop abnormal liver enzymes for evidence of worsening hepatic function, and evaluate risk/benefit of continuing caspofungin. (5.2)
- Abnormal Liver Enzymes during Concomitant use with Cyclosporine: Limit use to patients for whom potential benefit outweighs potential risk. Monitor patients who develop abnormal liver function tests (LFTs) during concomitant use with caspofungin. (5.3)
ADVERSE REACTIONS
- Adults: Most common adverse reactions (incidence 10% or greater) are diarrhea, pyrexia, ALT/AST increased, blood alkaline phosphatase increased, and blood potassium decreased. (6.1)
- Pediatric patients: Most common adverse reactions (incidence 10% or greater) are pyrexia, diarrhea, rash, ALT/AST increased, blood potassium decreased, hypotension, and chills. (6.2)
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
-
Pregnancy: Based on animal data, may cause fetal harm. (8.1)
Pediatric Use: Safety and efficacy in neonates and infants less than 3 months old have not been established. (8.4) - Hepatic Impairment: Reduce dose for adult patients with moderate hepatic impairment (35 mg once daily, with a 70 mg loading dose on Day 1 where appropriate). No data are available in adults with severe impairment or in pediatric patients with any degree of hepatic impairment. (2.4, 8.6,12.3)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 8/2018
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Empirical Therapy for Presumed Fungal Infections in Febrile, Neutropenic Patients
1.2 Treatment of Candidemia and Other Candida Infections
1.3 Treatment of Esophageal Candidiasis
1.4 Treatment of Invasive Aspergillosis in Patients Who Are Refractory to or Intolerant of Other Therapies
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions for Use in All Patients
2.2 Recommended Dosage in Adult Patients [18 years of age and older]
2.3 Recommended Dosing in Pediatric Patients [3 months to 17 years of age]
2.4 Dosage Adjustments in Patients with Hepatic Impairment
2.5 Dosage Adjustments in Patients Receiving Concomitant Inducers of Hepatic CYP Enzymes
2.6 Preparation for Administration
2.7 Drug Incompatibilities
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity
5.2 Hepatic Effects
5.3 Elevated Liver Enzymes During Concomitant Use with Cyclosporine
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Patients with Hepatic Impairment
8.7 Patients with Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
14.1 Empirical Therapy in Febrile, Neutropenic Patients
14.2 Candidemia and the Following Other Candida Infections: Intra-Abdominal Abscesses, Peritonitis and Pleural Space Infections
14.3 Esophageal Candidiasis (and information on oropharyngeal candidiasis)
14.4 Invasive Aspergillosis
14.5 Pediatric Patients
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Empirical Therapy for Presumed Fungal Infections in Febrile, Neutropenic Patients
Caspofungin acetate for injection is indicated as empirical therapy for presumed fungal infections in febrile, neutropenic adult and pediatric patients (3 months of age and older) [see Clinical Studies (14.1,14.5)].
1.2 Treatment of Candidemia and Other Candida Infections
Caspofungin acetate for injection is indicated for the treatment of candidemia and the following Candida infections: intra-abdominal abscesses, peritonitis, and pleural space infections in adult and pediatric patients (3 months of age and older) [see Clinical Studies (14.2, 14.5)].
Limitation of Use: Caspofungin acetate for injection has not been studied in endocarditis, osteomyelitis, and meningitis due to Candida.
1.3 Treatment of Esophageal Candidiasis
Caspofungin acetate for injection is indicated for the treatment of esophageal candidiasis in adult and pediatric patients (3 months of age and older) [see Clinical Studies (14.3, 14.5)].
Limitation of Use: Caspofungin acetate for injection has not been approved for the treatment of oropharyngeal candidiasis (OPC). In the study that evaluated the efficacy of caspofungin acetate for injection in the treatment of esophageal candidiasis, patients with concomitant OPC had higher relapse rate of the OPC [see Clinical Studies (14.3)].
1.4 Treatment of Invasive Aspergillosis in Patients Who Are Refractory to or Intolerant of Other Therapies
Caspofungin acetate for injection is indicated for the treatment of invasive aspergillosis in adult and pediatric patients (3 months of age and older) who are refractory to or intolerant of other therapies [see Clinical Studies (14.4,14.5)].
Limitation of Use: Caspofungin acetate for injection has not been studied as initial therapy for invasive aspergillosis.
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions for Use in All Patients
Administer caspofungin acetate for injection by slow intravenous (IV) infusion over approximately 1 hour. Do not administer caspofungin acetate for injection by intravenous (IV) bolus administration.
2.2 Recommended Dosage in Adult Patients [18 years of age and older]
The dosage and duration of caspofungin acetate for injection treatment for each indication are as follows:
Empirical Therapy for Presumed Fungal Infections in Febrile Neutropenic Patients
Administer a single 70 mg loading dose on Day 1, followed by 50 mg once daily thereafter. Duration of treatment should be based on the patient’s clinical response. Continue empirical therapy until resolution of neutropenia. In general, treat patients found to have a fungal infection for a minimum of 14 days after the last positive culture and continue treatment for at least 7 days after both neutropenia and clinical symptoms are resolved. If the 50 mg dose is well tolerated but does not provide an adequate clinical response, the daily dose can be increased to 70 mg.
Candidemia and Other Candida Infections
Administer a single 70 mg loading dose on Day 1, followed by 50 mg once daily thereafter. Duration of treatment should be dictated by the patient’s clinical and microbiological response. In general, continue antifungal therapy for at least 14 days after the last positive culture. Patients with neutropenia who remain persistently neutropenic may warrant a longer course of therapy pending resolution of the neutropenia.
Esophageal Candidiasis
The dose is 50 mg once daily for 7 to 14 days after symptom resolution. A 70 mg loading dose has not been studied for this indication. Because of the risk of relapse of oropharyngeal candidiasis in patients with HIV infections, suppressive oral therapy could be considered [see Clinical Studies (14.3)].
Invasive Aspergillosis
Administer a single 70 mg loading dose on Day 1, followed by 50 mg once daily thereafter. Duration of treatment should be based upon the severity of the patient’s underlying disease, recovery from immunosuppression, and clinical response.
2.3 Recommended Dosing in Pediatric Patients [3 months to 17 years of age]
For all indications, administer a single 70 mg/m2 loading dose on Day 1, followed by 50 mg/m2 once daily thereafter. The maximum loading dose and the daily maintenance dose should not exceed 70 mg, regardless of the patient's calculated dose. Dosing in pediatric patients (3 months to 17 years of age) should be based on the patient’s body surface area (BSA) as calculated by the Mosteller Formula [see References (15)]:
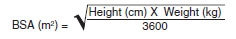
Following calculation of the patient’s BSA, the loading dose in milligrams should be calculated as BSA (m2) x 70 mg/m2. The maintenance dose in milligrams should be calculated as BSA (m2) x 50 mg/m2.
Duration of treatment should be individualized to the indication, as described for each indication in adults [see Dosage and Administration (2.2)]. If the 50 mg/m2 daily dose is well tolerated but does not provide an adequate clinical response, the daily dose can be increased to 70 mg/m2 daily (not to exceed 70 mg).
2.4 Dosage Adjustments in Patients with Hepatic Impairment
Adult patients with mild hepatic impairment (Child-Pugh score 5 to 6) do not need a dosage adjustment. For adult patients with moderate hepatic impairment (Child-Pugh score 7 to 9), caspofungin acetate for injection 35 mg once daily is recommended based upon pharmacokinetic data [see Clinical Pharmacology (12.3)] with a 70 mg loading dose administered on Day 1 where appropriate. There is no clinical experience in adult patients with severe hepatic impairment (Child-Pugh score greater than 9) and in pediatric patients with any degree of hepatic impairment.
2.5 Dosage Adjustments in Patients Receiving Concomitant Inducers of Hepatic CYP Enzymes
Adult Patients:
Adult patients on rifampin should receive 70 mg of caspofungin acetate for injection once daily. When caspofungin acetate for injection is co-administered to adult patients with other inducers of hepatic CYP enzymes such as nevirapine, efavirenz, carbamazepine, dexamethasone, or phenytoin, administration of a daily dose of 70 mg of caspofungin acetate for injection should be considered [see Drug Interactions (7)].
Pediatric Patients:
Pediatric patients on rifampin should receive 70 mg/m2 of caspofungin acetate for injection daily (not to exceed an actual daily dose of 70 mg). When caspofungin acetate for injection is co-administered to pediatric patients with other inducers of hepatic CYP enzymes, such as efavirenz, nevirapine, phenytoin, dexamethasone, or carbamazepine, a caspofungin acetate for injection dose of 70 mg/m2 once daily (not to exceed 70 mg) should be considered [see Drug Interactions (7)].
2.6 Preparation for Administration
Reconstitution of Caspofungin for Intravenous Infusion
A. Aseptically add 10.8 mL of 0.9% Sodium Chloride Injection, Sterile Water for Injection, Bacteriostatic Water for Injection with methylparaben and propylparaben, or Bacteriostatic Water for Injection with 0.9% benzyl alcohol to the vial.
B. Each vial of caspofungin acetate for injection contains an intentional overfill of caspofungin acetate for injection. Thus, the drug concentration of the resulting solution is listed in Table 1 below.
Table 1: Information for Preparation of Caspofungin Acetate for Injection
Caspofungin Acetate for Injection vial (equivalent to caspofungin)
Volume of diluent to be added*
Resulting Concentration following Reconstitution
50 mg
10.8 mL
5 mg/mL
70 mg
10.8 mL
7 mg/mL
*Reconstitution volume of diluent to be added is based on the overfill amount of caspofungin (54.6 mg and 77.2 mg, respectively).
C. The white to off-white cake will dissolve completely. Mix gently until a clear solution is obtained. Visually inspect the reconstituted solution for particulate matter or discoloration during reconstitution and prior to infusion. Do not use if the solution is cloudy or has precipitated.D. The reconstituted solution of caspofungin acetate for injection in the vial may be stored for up to one hour at ≤ 25°C (≤ 77°F) prior to the preparation of the infusion solution in the intravenous bag or bottle.
E. Caspofungin acetate for injection vials are for single-dose only. Discard unused portion.
Dilution of the Reconstituted Solution in the Intravenous Bag for InfusionA. Aseptically transfer the appropriate volume (mL) of reconstituted caspofungin acetate for injection to an intravenous (IV) bag (or bottle) containing 250 mL of 0.9%, 0.45%, or 0.225% Sodium Chloride Injection or Lactated Ringers Injection.
B. Alternatively, the volume (mL) of reconstituted caspofungin acetate for injection can be added to a reduced volume of 0.9%, 0.45%, or 0.225% Sodium Chloride Injection or Lactated Ringers Injection, not to exceed a final concentration of 0.5 mg/mL.
C. This diluted infusion solution in the intravenous bag or bottle must be used within 24 hours if stored at ≤ 25°C (≤ 77°F) or within 48 hours if stored refrigerated at 2° to 8°C (36° to 46°F).
Important Reconstitution and Dilution Instructions for Pediatric Patients 3 Months of Age and Older
Follow the reconstitution procedures described above using either the 70 mg or 50 mg vial to create the reconstituted solution [see Dosage and Administration (2.3)]. From the reconstituted solution in the vial, remove the volume of drug equal to the calculated loading dose or calculated maintenance dose based on a concentration of 7 mg/mL (if reconstituted from the 70 mg vial) or a concentration of 5 mg/mL (if reconstituted from the 50 mg vial).
The choice of vial should be based on total milligram dose of drug to be administered to the pediatric patient. To help ensure accurate dosing, it is recommended for pediatric doses less than 50 mg that 50 mg vials (with a concentration of 5 mg/mL) be used if available. The 70 mg vial should be reserved for pediatric patients requiring doses greater than 50 mg.
The maximum loading dose and the daily maintenance dose should not exceed 70 mg, regardless of the patient's calculated dose.
2.7 Drug Incompatibilities
Do not mix or co-infuse caspofungin acetate for injection with other medications, as there are no data available on the compatibility of caspofungin acetate for injection with other intravenous substances, additives, or medications.
Do not use diluents containing dextrose (α-D-glucose), as caspofungin acetate for injection is not stable in diluents containing dextrose.
-
3 DOSAGE FORMS AND STRENGTHS
Caspofungin acetate for injection, 50 mg, is a white to off-white lyophilized cake or powder for reconstitution in a single-dose glass vial, which contains 50 mg of caspofungin equivalent to 55.5 mg of caspofungin acetate.
Caspofungin acetate for injection, 70 mg, is a white to off-white lyophilized cake or powder for reconstitution in a single-dose glass vial, which contains 70 mg of caspofungin equivalent to 77.7 mg of caspofungin acetate.
-
4 CONTRAINDICATIONS
Caspofungin is contraindicated in patients with known hypersensitivity (e.g., anaphylaxis) to any component of this product [see Adverse Reactions (6)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity
Anaphylaxis has been reported during administration of caspofungin. If this occurs, discontinue caspofungin and administer appropriate treatment.
Possible histamine-mediated adverse reactions, including rash, facial swelling, angioedema, pruritus, sensation of warmth or bronchospasm have been reported and may require discontinuation and/or administration of appropriate treatment.
Cases of Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), have been reported after postmarketing use of caspofungin acetate. Caution should apply in patients with history of allergic skin reactions.5.2 Hepatic Effects
Laboratory abnormalities in liver function tests have been seen in healthy volunteers and in adult and pediatric patients treated with caspofungin. In some adult and pediatric patients with serious underlying conditions who were receiving multiple concomitant medications with caspofungin, isolated cases of clinically significant hepatic dysfunction, hepatitis, and hepatic failure have been reported; a causal relationship to caspofungin has not been established. Monitor patients who develop abnormal liver function tests during caspofungin therapy for evidence of worsening hepatic function and evaluated for risk/benefit of continuing caspofungin therapy.
5.3 Elevated Liver Enzymes During Concomitant Use with Cyclosporine
Elevated liver enzymes have occurred in patients receiving caspofungin and cyclosporine concomitantly. Only use caspofungin and cyclosporine in those patients for whom the potential benefit outweighs the potential risk. Patients who develop abnormal liver enzymes during concomitant therapy should be monitored and the risk/benefit of continuing therapy should be evaluated.
-
6 ADVERSE REACTIONS
The following serious adverse reactions are discussed in detail in another section of the labeling:
- Hypersensitivity [see Warnings and Precautions (5.1)]
- Hepatic Effects [see Warnings and Precautions (5.2)]
- Elevated Liver Enzymes During Concomitant Use with Cyclosporine [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of caspofungin cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in practice.
Clinical Trials Experience in Adults
The overall safety of caspofungin was assessed in 1,865 adult individuals who received single or multiple doses of caspofungin: 564 febrile, neutropenic patients (empirical therapy study); 382 patients with candidemia and/or intra-abdominal abscesses, peritonitis, or pleural space infections (including 4 patients with chronic disseminated candidiasis); 297 patients with esophageal and/or oropharyngeal candidiasis; 228 patients with invasive aspergillosis; and 394 individuals in phase I studies. In the empirical therapy study patients had undergone hematopoietic stem-cell transplantation or chemotherapy. In the studies involving patients with documented Candida infections, the majority of the patients had serious underlying medical conditions (e.g., hematologic or other malignancy, recent major surgery, HIV) requiring multiple concomitant medications. Patients in the noncomparative Aspergillus studies often had serious predisposing medical conditions (e.g., bone marrow or peripheral stem cell transplants, hematologic malignancy, solid tumors or organ transplants) requiring multiple concomitant medications.
Empirical Therapy for Presumed Fungal Infections in Febrile Neutropenic Patients
In the randomized, double-blinded empirical therapy study, patients received either caspofungin 50 mg/day (following a 70 mg loading dose) or AmBisome® (amphotericin B liposome for injection, 3 mg/kg/day). In this study clinical or laboratory hepatic adverse reactions were reported in 39% and 45% of patients in the caspofungin and AmBisome groups, respectively. Also reported was an isolated, serious adverse reaction of hyperbilirubinemia. Adverse reactions occurring in 7.5% or greater of the patients in either treatment group are presented in Table 2.
Table 2: Adverse Reactions Among Patients with Persistent Fever and Neutropenia Incidence 7.5% or Greater for at Least One Treatment Group
Adverse Reactions
Caspofungin*
N=564 (percent)
AmBisome†
N=547 (percent)
All Systems, Any Adverse Reaction
95
97
Investigations
58
63
Alanine Aminotransferase Increased
18
20
Blood Alkaline Phosphatase Increased
15
23
Blood Potassium Decreased
15
23
Aspartate Aminotransferase Increased
14
17
Blood Bilirubin Increased
10
14
Blood Magnesium Decreased
7
9
Blood Glucose Increased
6
9
Bilirubin Conjugated Increased
5
9
Blood Urea Increased
4
8
Blood Creatinine Increased
3
11
General Disorders and Administration Site Conditions
57
63
Pyrexia
27
29
Chills
23
31
Edema Peripheral
11
12
Mucosal Inflammation
6
8
Gastrointestinal Disorders
50
55
Diarrhea
20
16
Nausea
11
20
Abdominal Pain
9
11
Vomiting
9
17
Respiratory, Thoracic and Mediastinal Disorders
47
49
Dyspnea
9
10
Skin and Subcutaneous Tissue Disorders
42
37
Rash
16
14
Nervous System Disorders
25
27
Headache
11
12
Metabolism and Nutrition Disorders
21
24
Hypokalemia
6
8
Vascular Disorders
20
23
Hypotension
6
10
Cardiac Disorders
16
19
Tachycardia
7
9
Within any system organ class, individuals may experience more than 1 adverse reaction.
* 70 mg on Day 1, then 50 mg once daily for the remainder of treatment; daily dose was increased to 70 mg
for 73 patients.
†3 mg/kg/day; daily dose was increased to 5 mg/kg for 74 patients.
The proportion of patients who experienced an infusion-related adverse reaction (defined as a systemic event, such as pyrexia, chills, flushing, hypotension, hypertension, tachycardia, dyspnea, tachypnea, rash, or anaphylaxis, that developed during the study therapy infusion and one hour following infusion) was significantly lower in the group treated with caspofungin (35%) than in the group treated with AmBisome (52%).
To evaluate the effect of caspofungin and AmBisome on renal function, nephrotoxicity was defined as doubling of serum creatinine relative to baseline or an increase of greater than or equal to 1 mg/dL in serum creatinine if baseline serum creatinine was above the upper limit of the normal range. Among patients whose baseline creatinine clearance was greater than 30 mL/min, the incidence of nephrotoxicity was significantly lower in the group treated with caspofungin (3%) than in the group treated with AmBisome (12%).
Candidemia and Other Candida Infections
In the randomized, double-blinded invasive candidiasis study, patients received either caspofungin 50 mg/day (following a 70 mg loading dose) or amphotericin B 0.6 to 1 mg/kg/day. Adverse reactions occurring in 10% or greater of the patients in either treatment group are presented in Table 3.
Table 3: Adverse Reactions Among Patients with Candidemia or other Candida Infections* Incidence 10% or Greater for at Least One Treatment GroupAdverse Reactions
Caspofungin 50 mg†
N=114 (percent)
Amphotericin B N=125 (percent)
All Systems, Any Adverse Reaction
96
99
Investigations
67
82
Blood Potassium Decreased
23
32
Blood Alkaline Phosphatase Increased
21
32
Hemoglobin Decreased
18
23
Alanine Aminotransferase Increased
16
15
Aspartate Aminotransferase Increased
16
14
Blood Bilirubin Increased
13
17
Hematocrit Decreased
13
18
Blood Creatinine Increased
11
28
Red Blood Cells Urine Positive
10
10
Blood Urea Increased
9
23
Bilirubin Conjugated Increased
8
14
Gastrointestinal Disorders
49
53
Vomiting
17
16
Diarrhea
14
10
Nausea
9
17
General Disorders and Administration Site Conditions
47
63
Pyrexia
13
33
Edema Peripheral
11
12
Chills
9
30
Respiratory, Thoracic and Mediastinal Disorders
40
54
Tachypnea
1
11
Cardiac Disorders
26
34
Tachycardia
8
12
Skin and Subcutaneous Tissue Disorders
25
28
Rash
4
10
Vascular Disorders
25
38
Hypotension
10
16
Blood and Lymphatic System Disorders
15
13
Anemia
11
9
Within any system organ class, individuals may experience more than 1 adverse reaction.
* Intra-abdominal abscesses, peritonitis and pleural space infections.
† Patients received caspofungin 70 mg on Day 1, then 50 mg once daily for the remainder of their treatment.
The proportion of patients who experienced an infusion-related adverse reaction (defined as a systemic event, such as pyrexia, chills, flushing, hypotension, hypertension, tachycardia, dyspnea, tachypnea, rash, or anaphylaxis, that developed during the study therapy infusion and one hour following infusion) was significantly lower in the group treated with caspofungin (20%) than in the group treated with amphotericin B (49%).
To evaluate the effect of caspofungin and amphotericin B on renal function, nephrotoxicity was defined as doubling of serum creatinine relative to baseline or an increase of greater than or equal to 1 mg/dL in serum creatinine if baseline serum creatinine was above the upper limit of the normal range. In a subgroup of patients whose baseline creatinine clearance was greater than 30 mL/min, the incidence of nephrotoxicity was significantly lower in the group treated with caspofungin than in the group treated with amphotericin B.
In a second randomized, double-blinded invasive candidiasis study, patients received either caspofungin 50 mg/day (following a 70 mg loading dose) or caspofungin 150 mg/day. The proportion of patients who experienced any adverse reaction was similar in the 2 treatment groups; however, this study was not large enough to detect differences in rare or unexpected adverse reactions. Adverse reactions occurring in 5% or greater of the patients in either treatment group are presented in Table 4.
Table 4: Adverse Reactions Among Patients with Candidemia or other Candida Infections* Incidence 5% or Greater for at Least One Treatment GroupAdverse Reactions
Caspofungin 50 mg†N=104 (percent)
Caspofungin 150 mg N=100 (percent)
All Systems, Any Adverse Reaction
83
83
General Disorders and Administration Site Conditions
33
27
Pyrexia
6
6
Gastrointestinal Disorders
30
33
Vomiting
11
6
Diarrhea
6
7
Nausea
5
7
Investigations
28
35
Alkaline Phosphatase Increased
12
9
Aspartate Aminotransferase Increased
6
9
Blood Potassium Decreased
6
8
Alanine Aminotransferase Increased
4
7
Vascular Disorders
19
18
Hypotension
7
3
Hypertension
5
6
Within any system organ class, individuals may experience more than 1 adverse event.
* Intra-abdominal abscesses, peritonitis and pleural space infections.
† Patients received caspofungin 70 mg on Day 1, then 50 mg once daily for the remainder of their treatment.
Esophageal Candidiasis and Oropharyngeal Candidiasis
Adverse reactions occurring in 10% or greater of patients with esophageal and/or oropharyngeal candidiasis are presented in Table 5.
Table 5: Adverse Reactions Among Patients with Esophageal and/or Oropharyngeal Candidiasis Incidence 10% or Greater for at Least One Treatment GroupAdverse Reactions
Caspofungin 50 mg* N=83 (percent)
Fluconazole intravenous (IV) 200 mg* N=94 (percent)
All Systems, Any Adverse Reaction
90
93
Gastrointestinal Disorders
58
50
Diarrhea
27
18
Nausea
15
15
Investigations
53
61
Hemoglobin Decreased
21
16
Hematocrit Decreased
18
16
Aspartate Aminotransferase Increased
13
19
Blood Alkaline Phosphatase Increased
13
17
Alanine Aminotransferase Increased
12
17
White Blood Cell Count Decreased
12
19
General Disorders and Administration Site Conditions
31
36
Pyrexia
21
21
Vascular Disorders
19
15
Phlebitis
18
11
Nervous System Disorders
18
17
Headache
15
9
Within any system organ class, individuals may experience more than 1 adverse reaction.
*Derived from a comparator-controlled clinical study.
Invasive Aspergillosis
In an open-label, noncomparative aspergillosis study, in which 69 patients received caspofungin (70 mg loading dose on Day 1 followed by 50 mg daily), the following adverse reactions were observed with an incidence of 12.5% or greater: blood alkaline phosphatase increased (22%), hypotension (20%), respiratory failure (20%), pyrexia (17%), diarrhea (15%), nausea (15%), headache (15%), rash (13%), alanine aminotransferase increased (13%), aspartate aminotransferase increased (13%), blood bilirubin increased (13%), and blood potassium decreased (13%). Also reported in this patient population were pulmonary edema, ARDS (adult respiratory distress syndrome), and radiographic infiltrates.
Clinical Trials Experience in Pediatric Patients (3 months to 17 years of age)The overall safety of caspofungin was assessed in 171 pediatric patients who received single or multiple doses of caspofungin. The distribution among the 153 pediatric patients who were over the age of 3 months was as follows: 104 febrile, neutropenic patients; 38 patients with candidemia and/or intra-abdominal abscesses, peritonitis, or pleural space infections; 1 patient with esophageal candidiasis; and 10 patients with invasive aspergillosis. The overall safety profile of caspofungin in pediatric patients is comparable to that in adult patients. Table 6 shows the incidence of adverse reactions reported in 7.5% or greater of pediatric patients in clinical studies.
One patient (0.6%) receiving caspofungin, and three patients (12%) receiving AmBisome developed a serious drug-related adverse reaction. Two patients (1%) were discontinued from caspofungin and three patients (12%) were discontinued from AmBisome due to a drug-related adverse reaction. The proportion of patients who experienced an infusion-related adverse reaction (defined as a systemic event, such as pyrexia, chills, flushing, hypotension, hypertension, tachycardia, dyspnea, tachypnea, rash, or anaphylaxis, that developed during the study therapy infusion and one hour following infusion) was 22% in the group treated with caspofungin and 35% in the group treated with AmBisome.
Table 6: Adverse Reactions Among Pediatric Patients (0 months to 17 years of age)Incidence 7.5% or Greater for at Least One Treatment Group
Adverse Reactions
Noncomparative Clinical Studies
Comparator-Controlled Clinical Study of Empirical Therapy
Caspofungin
Any Dose
N=115 (percent)
Caspofungin
50 mg/m2*
N=56 (percent)
AmBisome
3 mg/kg
N=26 (percent)
All Systems, Any Adverse Reaction
95
96
89
Investigations
55
41
50
Blood Potassium Decreased
18
9
27
Aspartate Aminotransferase Increased
17
2
12
Alanine Aminotransferase Increased
14
5
12
Blood Potassium Increased
3
0
8
General Disorders and Administration Site Conditions
47
59
42
Pyrexia
29
30
23
Chills
10
13
8
Mucosal Inflammation
10
4
4
Edema
3
4
8
Gastrointestinal Disorders
42
41
35
Diarrhea
17
7
15
Vomiting
8
11
12
Abdominal Pain
7
4
12
Nausea
4
4
8
Infections and Infestations
40
30
35
Central Line Infection
1
9
0
Skin and Subcutaneous Tissue Disorders
33
41
39
Pruritus
7
6
8
Rash
6
23
8
Erythema
4
9
0
Vascular Disorders
24
21
19
Hypotension
12
9
8
Hypertension
10
9
4
Metabolism and Nutrition Disorders
22
11
23
Hypokalemia
8
5
4
Cardiac Disorders
17
13
19
Tachycardia
4
11
19
Nervous System Disorders
13
16
8
Headache
5
9
4
Musculoskeletal and Connective Tissue Disorders
11
14
12
Back Pain
4
0
8
Blood and Lymphatic System Disorders
10
2
15
Anemia
2
0
8
Within any system organ class, individuals may experience more than 1 adverse reaction.
* 70 mg/m2 on Day 1, then 50 mg/m2 once daily for the remainder of the treatment.
Overall Safety Experience of Caspofungin in Clinical Trials
The overall safety of caspofungin was assessed in 2,036 individuals (including 1,642 adult or pediatric patients and 394 volunteers) from 34 clinical studies. These individuals received single or multiple (once daily) doses of caspofungin, ranging from 5 mg to 210 mg. Full safety data is available from 1,951 individuals, as the safety data from 85 patients enrolled in 2 compassionate use studies was limited solely to serious adverse reactions. Adverse reactions which occurred in 5% or greater of all individuals who received caspofungin in these trials are shown in Table 7.
Overall, 1,665 of the 1,951 (85%) patients/volunteers who received caspofungin experienced an adverse reaction.
Table 7: Adverse Reactions* in Patients Who Received Caspofungin in Clinical Trials†Incidence 5% or Greater for at Least One Treatment Group
Adverse Reactions‡
Caspofungin
(N = 1,951)
N
(%)
All Systems, Any Adverse Reaction
1,665
(85)
Investigations
901
(46)
Alanine Aminotransferase Increased
258
(13)
Aspartate Aminotransferase Increased
233
(12)
Blood Alkaline Phosphatase Increased
232
(12)
Blood Potassium Decreased
220
(11)
Blood Bilirubin Increased
117
(6)
General Disorders and Administration Site Conditions
843
(43)
Pyrexia
381
(20)
Chills
192
(10)
Edema Peripheral
110
(6)
Gastrointestinal Disorders
754
(39)
Diarrhea
273
(14)
Nausea
166
(9)
Vomiting
146
(8)
Abdominal Pain
112
(6)
Infections and Infestations
730
(37)
Pneumonia
115
(6)
Respiratory, Thoracic, and Mediastinal Disorders
613
(31)
Cough
111
(6)
Skin and Subcutaneous Tissue Disorders
520
(27)
Rash
159
(8)
Erythema
98
(5)
Nervous System Disorders
412
(21)
Headache
193
(10)
Vascular Disorders
344
(18)
Hypotension
118
(6)
* Defined as an adverse reaction, regardless of causality, while on caspofungin or during the 14 day
post-caspofungin follow-up period.
† Incidence for each preferred term is 5% or greater among individuals who received at least 1 dose of
caspofungin.
‡ Within any system organ class, individuals may experience more than 1 adverse event.
Clinically significant adverse reactions, regardless of causality or incidence which occurred in less than 5% of patients are listed below.
Blood and lymphatic system disorders: anemia, coagulopathy, febrile neutropenia, neutropenia, thrombocytopenia
Cardiac disorders: arrhythmia, atrial fibrillation, bradycardia, cardiac arrest, myocardial infarction, tachycardia
Gastrointestinal disorders: abdominal distension, abdominal pain upper, constipation, dyspepsia
General disorders and administration site conditions: asthenia, fatigue, infusion site pain/pruritus/swelling, mucosal inflammation, edema
Hepatobiliary disorders: hepatic failure, hepatomegaly, hepatotoxicity, hyperbilirubinemia, jaundice
Infections and infestations: bacteremia, sepsis, urinary tract infection
Metabolic and nutrition disorders: anorexia, decreased appetite, fluid overload, hypomagnesemia, hypercalcemia, hyperglycemia, hypokalemia
Musculoskeletal, connective tissue, and bone disorders: arthralgia, back pain, pain in extremity
Nervous system disorders: convulsion, dizziness, somnolence, tremor
Psychiatric disorders: anxiety, confusional state, depression, insomnia
Renal and urinary disorders: hematuria, renal failure
Respiratory, thoracic, and mediastinal disorders: dyspnea, epistaxis, hypoxia, tachypnea
Skin and subcutaneous tissue disorders: erythema, petechiae, skin lesion, urticaria
Vascular disorders: flushing, hypertension, phlebitis
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during the post-approval use of caspofungin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal disorders: pancreatitis
Hepatobiliary disorders: hepatic necrosis
Skin and subcutaneous tissue disorders: erythema multiforme, toxic epidermal necrolysis, Stevens-Johnson syndrome, and skin exfoliation
Renal and urinary disorders: clinically significant renal dysfunction
General disorders and administration site conditions: swelling and peripheral edema
Laboratory abnormalities: gamma-glutamyltransferase increased
-
7 DRUG INTERACTIONS
Cyclosporine: In two adult clinical studies, cyclosporine (one 4 mg/kg dose or two 3 mg/kg doses) increased the AUC of caspofungin. Caspofungin did not increase the plasma levels of cyclosporine. There were transient increases in liver ALT and AST when caspofungin and cyclosporine were co-administered. Monitor patients who develop abnormal liver enzymes during concomitant therapy and evaluate the risk/benefit of continuing therapy [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)].
Tacrolimus: For patients receiving caspofungin and tacrolimus, standard monitoring of tacrolimus trough whole blood concentrations and appropriate tacrolimus dosage adjustments are recommended.
Inducers of Hepatic CYP Enzymes
Rifampin: Rifampin is a potent CYP3A4 inducer and concomitant administration with caspofungin is expected to reduce the plasma concentrations of caspofungin. Therefore, adult patients on rifampin should receive 70 mg of caspofungin daily and pediatric patients on rifampin should receive 70 mg/m2 of caspofungin daily (not to exceed an actual daily dose of 70 mg) [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3)].
Other Inducers of Hepatic CYP Enzymes
Adults: When caspofungin is co-administered to adult patients with other inducers of hepatic CYP enzymes, such as efavirenz, nevirapine, phenytoin, dexamethasone, or carbamazepine, administration of a daily dose of 70 mg of caspofungin should be considered [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3)].
Pediatric Patients: When caspofungin is co-administered to pediatric patients with other inducers of hepatic CYP enzymes, such as efavirenz, nevirapine, phenytoin, dexamethasone, or carbamazepine, administration of a daily dose of 70 mg/m2 caspofungin (not to exceed an actual daily dose of 70 mg) should be considered [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C
There are no adequate and well-controlled studies with the use of caspofungin in pregnant women. In animal studies, caspofungin caused embryofetal toxicity, including increased resorptions, increased peri-implantation loss, and incomplete ossification at multiple fetal sites. Caspofungin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
In offspring born to pregnant rats treated with caspofungin at doses comparable to the human dose based on body surface area comparisons, there was incomplete ossification of the skull and torso and increased incidences of cervical rib. There was also an increase in resorptions and peri-implantation losses. In pregnant rabbits treated with caspofungin at doses comparable to 2 times the human dose based on body surface area comparisons, there was an increased incidence of incomplete ossification of the talus/calcaneus in offspring and increases in fetal resorptions. Caspofungin crossed the placenta in rats and rabbits and was detectable in fetal plasma.
8.3 Nursing Mothers
It is not known whether caspofungin is present in human milk. Caspofungin was found in the milk of lactating, drug-treated rats. Because many drugs are excreted in human milk, caution should be exercised when caspofungin is administered to a nursing woman
8.4 Pediatric Use
The safety and effectiveness of caspofungin in pediatric patients 3 months to 17 years of age are supported by evidence from adequate and well-controlled studies in adults, pharmacokinetic data in pediatric patients, and additional data from prospective studies in pediatric patients 3 months to 17 years of age for the following indications [see Indications and Usage (1)]:
- Empirical therapy for presumed fungal infections in febrile, neutropenic patients.
- Treatment of candidemia and the following Candida infections: intra-abdominal abscesses, peritonitis, and pleural space infections.
- Treatment of esophageal candidiasis.
- Treatment of invasive aspergillosis in patients who are refractory to or intolerant of other therapies (e.g., amphotericin B, lipid formulations of amphotericin B, itraconazole).
The efficacy and safety of caspofungin has not been adequately studied in prospective clinical trials involving neonates and infants under 3 months of age. Although limited pharmacokinetic data were collected in neonates and infants below 3 months of age, these data are insufficient to establish a safe and effective dose of caspofungin in the treatment of neonatal candidiasis. Invasive candidiasis in neonates has a higher rate of CNS and multi-organ involvement than in older patients; the ability of caspofungin to penetrate the blood-brain barrier and to treat patients with meningitis and endocarditis is unknown.
Caspofungin has not been studied in pediatric patients with endocarditis, osteomyelitis, and meningitis due to Candida. Caspofungin has also not been studied as initial therapy for invasive aspergillosis in pediatric patients.
In clinical trials, 171 pediatric patients (0 months to 17 years of age), including 18 patients who were less than 3 months of age, were given intravenous caspofungin. Pharmacokinetic studies enrolled a total of 66 pediatric patients, and an additional 105 pediatric patients received caspofungin in safety and efficacy studies [see Clinical Pharmacology (12.3) and Clinical Studies (14.5)]. The majority of the pediatric patients received caspofungin at a once-daily maintenance dose of 50 mg/m2 for a mean duration of 12 days (median 9, range 1 to 87 days). In all studies, safety was assessed by the investigator throughout study therapy and for 14 days following cessation of study therapy. The most common adverse reactions in pediatric patients treated with caspofungin were pyrexia (29%), blood potassium decreased (15%), diarrhea (14%), increased aspartate aminotransferase (12%), rash (12%), increased alanine aminotransferase (11%), hypotension (11%), and chills (11%) [see Adverse Reactions (6.2)].
Postmarketing hepatobiliary adverse reactions have been reported in pediatric patients with serious underlying medical conditions [see Warnings and Precautions (5.3)].
8.5 Geriatric Use
Clinical studies of caspofungin did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients. Although the number of elderly patients was not large enough for a statistical analysis, no overall differences in safety or efficacy were observed between these and younger patients. Plasma concentrations of caspofungin in healthy older men and women (65 years of age and older) were increased slightly (approximately 28% in AUC) compared to young healthy men. A similar effect of age on pharmacokinetics was seen in patients with candidemia or other Candida infections (intra-abdominal abscesses, peritonitis, or pleural space infections). No dose adjustment is recommended for the elderly; however, greater sensitivity of some older individuals cannot be ruled out.
8.6 Patients with Hepatic Impairment
Adult patients with mild hepatic impairment (Child-Pugh score 5 to 6) do not need a dosage adjustment. For adult patients with moderate hepatic impairment (Child-Pugh score 7 to 9), caspofungin 35 mg once daily is recommended based upon pharmacokinetic data [see Clinical Pharmacology (12.3)]. However, where recommended, a 70 mg loading dose should still be administered on Day 1 [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)]. There is no clinical experience in adult patients with severe hepatic impairment (Child-Pugh score greater than 9) and in pediatric patients 3 months to 17 years of age with any degree of hepatic impairment.
8.7 Patients with Renal Impairment
No dosage adjustment is necessary for patients with renal impairment. Caspofungin is not dialyzable; thus, supplementary dosing is not required following hemodialysis [see Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
In 6 healthy subjects who received a single 210 mg dose, no significant adverse reactions were reported. Multiple doses above 150 mg daily have not been studied. Caspofungin is not dialyzable.
In clinical trials, one pediatric patient (16 years of age) unintentionally received a single dose of caspofungin of 113 mg (on Day 1), followed by 80 mg daily for an additional 7 days. No clinically significant adverse reactions were reported.
-
11 DESCRIPTION
Caspofungin acetate for injection is a sterile, lyophilized product for intravenous (IV) infusion that contains a semisynthetic lipopeptide (echinocandin) compound synthesized from a fermentation product of Glarea lozoyensis. Caspofungin acetate is an echinocandin antifungal that inhibits the synthesis of β (1,3)-D-glucan, an integral component of the fungal cell wall.
Caspofungin acetate is 1-[(4R,5S)-5-[(2-aminoethyl)amino]-N2-(10,12-dimethyl-1-oxotetradecyl)-4-hydroxy-L-ornithine]-5-[(3R)-3-hydroxy-L-ornithine] pneumocandin B0 diacetate (salt). Each vial of caspofungin acetate for injection, 50 mg, contains 50 mg of caspofungin equivalent to 55.5 mg of caspofungin acetate, arginine (100 mg), and hydrochloric acid/sodium hydroxide required for pH adjustment. Each vial of caspofungin acetate for injection, 70 mg, contains 70 mg of caspofungin equivalent to 77.7 mg of caspofungin acetate, arginine (140 mg), and hydrochloric acid/sodium hydroxide required for pH adjustment. Caspofungin acetate is a hygroscopic, white to off-white powder. It is freely soluble in water and methanol, and slightly soluble in ethanol. The pH of a saturated aqueous solution of caspofungin acetate is approximately 6.6. The structural formula is:
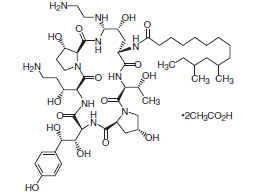
C52H88N10O152C2H4O2 M.W. 1213.42
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Caspofungin is an echinocandin antifungal drug [see Clinical Pharmacology (12.4)].
12.3 Pharmacokinetics
Adult and pediatric pharmacokinetic parameters are presented in Table 8.
Distribution
Plasma concentrations of caspofungin decline in a polyphasic manner following single 1 hour intravenous infusions. A short α-phase occurs immediately postinfusion, followed by a β-phase (half-life of 9 to 11 hours) that characterizes much of the profile and exhibits clear log-linear behavior from 6 to 48 hours postdose during which the plasma concentration decreases 10-fold. An additional, longer half-life phase, y-phase, (half-life of 40 to 50 hours), also occurs. Distribution, rather than excretion or biotransformation, is the dominant mechanism influencing plasma clearance. Caspofungin is extensively bound to albumin (approximately 97%), and distribution into red blood cells is minimal. Mass balance results showed that approximately 92% of the administered radioactivity was distributed to tissues by 36 to 48 hours after a single 70 mg dose of [3H] caspofungin acetate. There is little excretion or biotransformation of caspofungin during the first 30 hours after administration.
Metabolism
Caspofungin is slowly metabolized by hydrolysis and N-acetylation. Caspofungin also undergoes spontaneous chemical degradation to an open-ring peptide compound, L-747969. At later time points (5 or more days postdose), there is a low level (7 or less picomoles/mg protein, or 1.3% or less of the administered dose) of covalent binding of radiolabel in plasma following single-dose administration of [3H] caspofungin acetate, which may be due to two reactive intermediates formed during the chemical degradation of caspofungin to L-747969. Additional metabolism involves hydrolysis into constitutive amino acids and their degradates, including dihydroxyhomotyrosine and N-acetyl-dihydroxyhomotyrosine. These two tyrosine derivatives are found only in urine, suggesting rapid clearance of these derivatives by the kidneys.
Excretion
Two single-dose radiolabeled pharmacokinetic studies were conducted. In one study, plasma, urine, and feces were collected over 27 days, and in the second study plasma was collected over 6 months. Plasma concentrations of radioactivity and of caspofungin were similar during the first 24 to 48 hours postdose; thereafter drug levels fell more rapidly. In plasma, caspofungin concentrations fell below the limit of quantitation after 6 to 8 days postdose, while radiolabel fell below the limit of quantitation at 22.3 weeks postdose. After single intravenous administration of [3H] caspofungin acetate, excretion of caspofungin and its metabolites in humans was 35% of dose in feces and 41% of dose in urine. A small amount of caspofungin is excreted unchanged in urine (approximately 1.4% of dose). Renal clearance of parent drug is low (approximately 0.15 mL/min) and total clearance of caspofungin is 12 mL/min.
Special Populations
Renal Impairment
In a clinical study of single 70 mg doses, caspofungin pharmacokinetics were similar in healthy adult volunteers with mild renal impairment (creatinine clearance 50 to 80 mL/min) and control subjects. Moderate (creatinine clearance 31 to 49 mL/min), severe (creatinine clearance 5 to 30 mL/min), and end-stage (creatinine clearance less than 10 mL/min and dialysis dependent) renal impairment moderately increased caspofungin plasma concentrations after single-dose administration (range: 30 to 49% for AUC). However, in adult patients with invasive aspergillosis, candidemia, or other Candida infections (intra-abdominal abscesses, peritonitis, or pleural space infections) who received multiple daily doses of caspofungin 50 mg, there was no significant effect of mild to end-stage renal impairment on caspofungin concentrations. No dosage adjustment is necessary for patients with renal impairment. Caspofungin is not dialyzable, thus supplementary dosing is not required following hemodialysis.
Hepatic Impairment
Plasma concentrations of caspofungin after a single 70 mg dose in adult patients with mild hepatic impairment (Child-Pugh score 5 to 6) were increased by approximately 55% in AUC compared to healthy control subjects. In a 14 day multiple-dose study (70 mg on Day 1 followed by 50 mg daily thereafter), plasma concentrations in adult patients with mild hepatic impairment were increased modestly (19 to 25% in AUC) on Days 7 and 14 relative to healthy control subjects. No dosage adjustment is recommended for patients with mild hepatic impairment.
Adult patients with moderate hepatic impairment (Child-Pugh score 7 to 9) who received a single 70 mg dose of caspofungin had an average plasma caspofungin increase of 76% in AUC compared to control subjects. A dosage reduction is recommended for adult patients with moderate hepatic impairment based upon these pharmacokinetic data [see Dosage and Administration (2.4)].
There is no clinical experience in adult patients with severe hepatic impairment (Child-Pugh score greater than 9) or in pediatric patients with any degree of hepatic impairment.
Gender
Plasma concentrations of caspofungin in healthy adult men and women were similar following a single 70 mg dose. After 13 daily 50 mg doses, caspofungin plasma concentrations in women were elevated slightly (approximately 22% in area under the curve [AUC]) relative to men. No dosage adjustment is necessary based on gender.
Race
Regression analyses of patient pharmacokinetic data indicated that no clinically significant differences in the pharmacokinetics of caspofungin were seen among Caucasians, Blacks, and Hispanics. No dosage adjustment is necessary on the basis of race.
Geriatric Patients
Plasma concentrations of caspofungin in healthy older men and women (65 years of age and older) were increased slightly (approximately 28% AUC) compared to young healthy men after a single 70 mg dose of caspofungin. In patients who were treated empirically or who had candidemia or other Candida infections (intra-abdominal abscesses, peritonitis, or pleural space infections), a similar modest effect of age was seen in older patients relative to younger patients. No dosage adjustment is necessary for the elderly [see Use in Specific Populations (8.5)].
Pediatric Patients
Caspofungin has been studied in five prospective studies involving pediatric patients under 18 years of age, including three pediatric pharmacokinetic studies [initial study in adolescents (12 to 17 years of age) and children (2 to 11 years of age) followed by a study in younger patients (3 to 23 months of age) and then followed by a study in neonates and infants (less than 3 months of age)] [see Use in Specific Populations (8.4)].
Pharmacokinetic parameters following multiple doses of caspofungin in pediatric and adult patients are presented in Table 8.
Table 8: Pharmacokinetic Parameters Following Multiple Doses of Caspofungin in Pediatric (3 months to 17 years) and Adult PatientsPopulation
N
Daily Dose
Pharmacokinetic Parameters
(Mean ±Standard Deviation)
AUC0-24hr (mcghr/mL)
C1hr (mcg/mL)
C24hr (mcg/mL)
t1/2
(hr)*
CI
(mL/min)
PEDIATRIC PATIENTS
Adolescents,
Aged 12 to 17 years
8
50 mg/m2
124.9 ± 50.4
14.0 ± 6.9
2.4 ± 1.0
11.2 ± 1.7
12.6 ± 5.5
Children,
Aged 2 to 11 years
9
50 mg/m2
120.0 ± 33.4
16.1 ± 4.2
1.7 ± 0.8
8.2 ± 2.4
6.4 ± 2.6
Young Children,
Aged 3 to 23 months
8
50 mg/m2
131.2 ± 17.7
17.6 ± 3.9
1.7 ± 0.7
8.8 ± 2.1
3.2 ± 0.4
ADULT PATIENTS
Adults with Esophageal Candidiasis
6†
50 mg
87.3 ± 30.0
8.7 ± 2.1
1.7 ± 0.7
13.0 ± 1.9
10.6 ± 3.8
Adults receiving Empirical Therapy
119‡
50 mg§
--
8.0 ± 3.4
1.6 ± 0.7
--
--
* Harmonic Mean ± jackknife standard deviation.
† N=5 for C1hr and AUC0-24hr; N=6 for C24hr .
‡ N=117 for C24hr; N=119 for C1hr .
§ Following an initial 70 mg loading dose on day 1.
Drug Interactions [see Drug Interactions (7)]Studies in vitro show that caspofungin acetate is not an inhibitor of any enzyme in the cytochrome P (CYP) system. Caspofungin is not a substrate for P-glycoprotein and is a poor substrate for CYP enzymes.
In clinical studies, caspofungin did not induce the CYP3A4 metabolism of other drugs. Clinical studies in adult healthy volunteers also demonstrated that the pharmacokinetics of caspofungin are not altered by itraconazole, amphotericin B, mycophenolate, nelfinavir, or tacrolimus. Caspofungin has no effect on the pharmacokinetics of itraconazole, amphotericin B, or the active metabolite of mycophenolate.
Cyclosporine: In two adult clinical studies, cyclosporine (one 4 mg/kg dose or two 3 mg/kg doses) increased the AUC of caspofungin by approximately 35%. Caspofungin did not increase the plasma levels of cyclosporine. There were transient increases in liver ALT and AST when caspofungin and cyclosporine were co-administered [see Warnings and Precautions (5.2)].
Tacrolimus: Caspofungin reduced the blood AUC0-12 of tacrolimus (FK-506, Prograf ®) by approximately 20%, peak blood concentration (Cmax) by 16%, and 12 hour blood concentration (C12hr) by 26% in healthy adult subjects when tacrolimus (2 doses of 0.1 mg/kg 12 hours apart) was administered on the 10th day of caspofungin 70 mg daily, as compared to results from a control period in which tacrolimus was administered alone. For patients receiving both therapies, standard monitoring of tacrolimus whole blood trough concentrations and appropriate tacrolimus dosage adjustments are recommended.
Rifampin: A drug-drug interaction study with rifampin in adult healthy volunteers has shown a 30% decrease in caspofungin trough concentrations [see Dosage and Administration (2.5)].
Other Inducers of Hepatic CYP Enzymes
Adults: Results from regression analyses of adult patient pharmacokinetic data suggest that co-administration of other hepatic CYP enzyme inducers (e.g., efavirenz, nevirapine, phenytoin, dexamethasone, or carbamazepine) with caspofungin may result in clinically meaningful reductions in caspofungin concentrations. It is not known which drug clearance mechanism involved in caspofungin disposition may be inducible [see Dosage and Administration (2.5)].
Pediatric patients: In pediatric patients, results from regression analyses of pharmacokinetic data suggest that co-administration of dexamethasone with caspofungin may result in clinically meaningful reductions in caspofungin trough concentrations. This finding may indicate that pediatric patients will have similar reductions with inducers as seen in adults [see Dosage and Administration (2.5)].
12.4 Microbiology
Mechanism of Action
Caspofungin, an echinocandin, inhibits the synthesis of beta (1,3)-D-glucan, an essential component of the cell wall of susceptible Aspergillus species and Candida species. Beta (1,3)-D-glucan is not present in mammalian cells. Caspofungin has shown activity against Candida species and in regions of active cell growth of the hyphae of Aspergillus fumigatus.
Drug Resistance
There have been reports of clinical failures in patients receiving caspofungin therapy due to the development of drug resistance. Some of these reports have identified specific mutations in the Fks subunits of the glucan synthase enzyme. These mutations are associated with higher MICs and breakthrough infection. Candida species that exhibit reduced susceptibility to caspofungin as a result of an increase in the chitin content of the fungal cell wall have also been identified, although the significance of this phenomenon in vivo is not well known.
Drug Interactions
Studies in vitro and in vivo of caspofungin, in combination with amphotericin B, suggest no antagonism of antifungal activity against either A. fumigatus or C. albicans. The clinical significance of these results is unknown.
Activity in Vitro and in Clinical Infections
Caspofungin has been shown to be active both in vitro and in clinical infections against most strains of the following microorganisms:
Aspergillus fumigatus
Aspergillus flavus
Aspergillus terreus
Candida albicans
Candida glabrata
Candida guilliermondii
Candida krusei
Candida parapsilosis
Candida tropicalis
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC. -
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies in animals have been performed to evaluate the carcinogenic potential of caspofungin.
Caspofungin did not show evidence of mutagenic or genotoxic potential when evaluated in the following in vitro assays: bacterial (Ames) and mammalian cell (V79 Chinese hamster lung fibroblasts) mutagenesis assays, the alkaline elution/rat hepatocyte DNA strand break test, and the chromosome aberration assay in Chinese hamster ovary cells. Caspofungin was not genotoxic when assessed in the mouse bone marrow chromosomal test at doses up to 12.5 mg/kg (equivalent to a human dose of 1 mg/kg based on body surface area comparisons), administered intravenously.
Fertility and reproductive performance were not affected by the intravenous administration of caspofungin to rats at doses up to 5 mg/kg. At 5 mg/kg exposures were similar to those seen in patients treated with the 70 mg dose.
13.2 Animal Toxicology and/or Pharmacology
In one 5 week study in monkeys at doses which produced exposures approximately 4 to 6 times those seen in adult patients treated with a 70 mg dose, scattered small foci of subcapsular necrosis were observed microscopically in the livers of some animals (2/8 monkeys at 5 mg/kg and 4/8 monkeys at 8 mg/kg); however, this histopathological finding was not seen in another study of 27 weeks duration at similar doses.
No treatment-related findings were seen in a 5 week study in infant monkeys at doses which produced exposures approximately 3 times those achieved in pediatric patients receiving a maintenance dose of 50 mg/m2 daily.
-
14 CLINICAL STUDIES
14.1 Empirical Therapy in Febrile, Neutropenic Patients
A double-blind study enrolled 1,111 febrile, neutropenic (less than 500 cells/mm3) patients who were randomized to treatment with daily doses of caspofungin (50 mg/day following a 70 mg loading dose on Day 1) or AmBisome (3 mg/kg/day). Patients were stratified based on risk category (high-risk patients had undergone allogeneic stem cell transplantation or had relapsed acute leukemia) and on receipt of prior antifungal prophylaxis. Twenty-four percent of patients were high risk and 56% had received prior antifungal prophylaxis. Patients who remained febrile or clinically deteriorated following 5 days of therapy could receive 70 mg/day of caspofungin or 5 mg/kg/day of AmBisome. Treatment was continued to resolution of neutropenia (but not beyond 28 days unless a fungal infection was documented).
An overall favorable response required meeting each of the following criteria: no documented breakthrough fungal infections up to 7 days after completion of treatment, survival for 7 days after completion of study therapy, no discontinuation of the study drug because of drug-related toxicity or lack of efficacy, resolution of fever during the period of neutropenia, and successful treatment of any documented baseline fungal infection.
Based on the composite response rates, caspofungin was as effective as AmBisome in empirical therapy of persistent febrile neutropenia (see Table 11).
Table 11: Favorable Response of Patients with Persistent Fever and Neutropenia
Caspofungin*
AmBisome*
% Difference
(Confidence Interval)†
Number of Patients‡
556
539
Overall Favorable Response
190 (33.9%)
181 (33.7%)
0.2 (-5.6, 6.0)
No documented breakthrough fungal
infection
527 (94.8%)
515 (95.5%)
-0.8
Survival 7 days after end of treatment
515 (92.6%)
481 (89.2%)
3.4
No discontinuation due to toxicity or
lack of efficacy
499 (89.7%)
461 (85.5%)
4.2
Resolution of fever during neutropenia
229 (41.2%)
223 (41.4%)
-0.2
* Caspofungin: 70 mg on Day 1, then 50 mg once daily for the remainder of treatment (daily dose increased to 70 mg for 73 patients); AmBisome: 3 mg/kg/day (daily dose increased to 5 mg/kg for 74 patients).
† Overall Response: estimated % difference adjusted for strata and expressed as caspofungin – AmBisome (95.2% CI); Individual criteria presented above are not mutually exclusive. The percent difference calculated as caspofungin – AmBisome.
‡ Analysis population excluded subjects who did not have fever or neutropenia at study entry.
The rate of successful treatment of documented baseline infections, a component of the primary endpoint, was not statistically different between treatment groups.
The response rates did not differ between treatment groups based on either of the stratification variables: risk category or prior antifungal prophylaxis.
14.2 Candidemia and the Following Other Candida Infections: Intra-Abdominal Abscesses, Peritonitis and Pleural Space Infections
In a randomized, double-blind study, patients with a proven diagnosis of invasive candidiasis received daily doses of caspofungin (50 mg/day following a 70 mg loading dose on Day 1) or amphotericin B deoxycholate (0.6 to 0.7 mg/kg/day for non-neutropenic patients and 0.7 to 1 mg/kg/day for neutropenic patients). Patients were stratified by both neutropenic status and APACHE II score. Patients with Candida endocarditis, meningitis, or osteomyelitis were excluded from this study.
Patients who met the entry criteria and received one or more doses of intravenous (IV) study therapy were included in the modified intention-to-treat [MITT] analysis of response at the end of intravenous (IV) study therapy. A favorable response at this time point required both symptom/sign resolution/improvement and microbiological clearance of the Candida infection.
Two hundred thirty-nine patients were enrolled. Patient disposition is shown in Table 12.
Table 12: Disposition in Candidemia and Other Candida Infections (Intra-abdominal abscesses, peritonitis, and pleural space infections)
Caspofungin*
Amphotericin B
Randomized patients
114
125
Patients completing study†
63 (55.3%)
69 (55.2%)
DISCONTINUATIONS OF STUDY†
All Study Discontinuations
51 (44.7%)
56 (44.8%)
Study Discontinuations due to clinical adverse events
39 (34.2%)
43 (34.4%)
Study Discontinuations due to laboratory adverse events
0 (0%)
1 (0.8%)
DISCONTINUATIONS OF STUDY THERAPY
All Study Therapy Discontinuations
48 (42.1%)
58 (46.4%)
Study Therapy Discontinuations due to clinical adverse events
30 (26.3%)
37 (29.6%)
Study Therapy Discontinuations due to laboratory adverse events
1 (0.9%)
7 (5.6%)
Study Therapy Discontinuations due to all drug-related‡ adverse
events
3 (2.6%)
29 (23.2%)
* Patients received caspofungin 70 mg on Day 1, then 50 mg once daily for the remainder of their treatment.
† Study defined as study treatment period and 6 to 8 week follow-up period.
‡ Determined by the investigator to be possibly, probably, or definitely drug-related.
Of the 239 patients enrolled, 224 met the criteria for inclusion in the MITT population (109 treated with caspofungin and 115 treated with amphotericin B). Of these 224 patients, 186 patients had candidemia (92 treated with caspofungin and 94 treated with amphotericin B). The majority of the patients with candidemia were non-neutropenic (87%) and had an APACHE II score less than or equal to 20 (77%) in both arms. Most candidemia infections were caused by C. albicans (39%), followed by C. parapsilosis (20%), C. tropicalis (17%), C. glabrata (8%), and C. krusei (3%).
At the end of intravenous (IV) study therapy, caspofungin was comparable to amphotericin B in the treatment of candidemia in the MITT population. For the other efficacy time points (Day 10 of intravenous (IV) study therapy, end of all antifungal therapy, 2-week post-therapy follow-up, and 6- to 8-week post-therapy follow-up), caspofungin was as effective as amphotericin B.
Outcome, relapse and mortality data are shown in Table 13.
Table 13: Outcomes, Relapse, & Mortality in Candidemia and Other Candida Infections (Intra-abdominal abscesses, peritonitis, and pleural space infections)
Caspofungin*
Amphotericin B
% Difference† after adjusting for strata
(Confidence Interval)‡
Number of MITT§ patients
109
115
FAVORABLE OUTCOMES (MITT) AT THE END OF INTRAVENOUS (IV) STUDY THERAPY
All MITT patients
81/109 (74.3%)
78/115 (67.8%)
7.5 (-5.4, 20.3)
Candidemia
67/92 (72.8%)
63/94 (67.0%)
7.0 (-7.0, 21.1)
Neutropenic
6/14 (43%)
5/10 (50%)
Non-neutropenic
61/78 (78%)
58/84 (69%)
Endophthalmitis
0/1
2/3
Multiple Sites
4/5
4/4
Blood / Pleural
1/1
1/1
Blood / Peritoneal
1/1
1/1
Blood / Urine
-
1/1
Peritoneal / Pleural
1/2
-
Abdominal / Peritoneal
-
1/1
Subphrenic / Peritoneal
1/1
-
DISSEMINATED INFECTIONS, RELAPSES AND MORTALITY
Disseminated Infections in
neutropenic patients
4/14 (28.6%)
3/10 (30.0%)
All relapses¶
7/81 (8.6%)
8/78 (10.3%)
Culture-confirmed relapse
5/81 (6%)
2/78 (3%)
Overall study# mortality in MITT
36/109 (33.0%)
35/115 (30.4%)
Mortality during study
therapy
18/109 (17%)
13/115 (11%)
Mortality attributed to
Candida
4/109 (4%)
7/115 (6%)
*Patients received caspofungin 70 mg on Day 1, then 50 mg once daily for the remainder of their treatment.
† Calculated as caspofungin - amphotericin B.
‡ 95% CI for candidemia, 95.6% for all patients.
§ Modified intention-to-treat.
¶ Includes all patients who either developed a culture-confirmed recurrence of Candida infection or required antifungal therapy for the treatment of a proven or suspected Candida infection in the follow-up period.
# Study defined as study treatment period and 6 to 8 week follow-up period.
In this study, the efficacy of caspofungin in patients with intra-abdominal abscesses, peritonitis and pleural space Candida infections was evaluated in 19 non-neutropenic patients. Two of these patients had concurrent candidemia. Candida was part of a polymicrobial infection that required adjunctive surgical drainage in 11 of these 19 patients. A favorable response was seen in 9 of 9 patients with peritonitis, 3 of 4 with abscesses (liver, parasplenic, and urinary bladder abscesses), 2 of 2 with pleural space infections, 1 of 2 with mixed peritoneal and pleural infection, 1 of 1 with mixed abdominal abscess and peritonitis, and 0 of 1 with Candida pneumonia.
Overall, across all sites of infection included in the study, the efficacy of caspofungin was comparable to that of amphotericin B for the primary endpoint.
In this study, the efficacy data for caspofungin in neutropenic patients with candidemia were limited. In a separate compassionate use study, 4 patients with hepatosplenic candidiasis received prolonged therapy with caspofungin following other long-term antifungal therapy; three of these patients had a favorable response.
In a second randomized, double-blind study, 197 patients with proven invasive candidiasis received caspofungin 50 mg/day (following a 70 mg loading dose on Day 1) or caspofungin 150 mg/day. The diagnostic criteria, evaluation time points, and efficacy endpoints were similar to those employed in the prior study. Patients with Candida endocarditis, meningitis, or osteomyelitis were excluded. Although this study was designed to compare the safety of the two doses, it was not large enough to detect differences in rare or unexpected adverse events [see Adverse Reactions (6.1)]. The efficacy of caspofungin at the 150 mg daily dose was not significantly better than the efficacy of the 50 mg daily dose of caspofungin. The efficacy of doses higher than 50 mg daily in the other adult patients for whom caspofungin is indicated has not been evaluated.
14.3 Esophageal Candidiasis (and information on oropharyngeal candidiasis)
The safety and efficacy of caspofungin in the treatment of esophageal candidiasis was evaluated in one large, controlled, noninferiority, clinical trial and two smaller dose-response studies.
In all 3 studies, patients were required to have symptoms and microbiological documentation of esophageal candidiasis; most patients had advanced AIDS (with CD4 counts less than 50/mm3).
Of the 166 patients in the large study who had culture-confirmed esophageal candidiasis at baseline, 120 had Candida albicans and 2 had Candida tropicalis as the sole baseline pathogen whereas 44 had mixed baseline cultures containing C. albicans and one or more additional Candida species.
In the large, randomized, double-blind study comparing caspofungin 50 mg/day versus intravenous fluconazole 200 mg/day for the treatment of esophageal candidiasis, patients were treated for an average of 9 days (range 7 to 21 days). Favorable overall response at 5- to 7-days following discontinuation of study therapy required both complete resolution of symptoms and significant endoscopic improvement. The definition of endoscopic response was based on severity of disease at baseline using a 4-grade scale and required at least a two-grade reduction from baseline endoscopic score or reduction to grade 0 for patients with a baseline score of 2 or less.
The proportion of patients with a favorable overall response was comparable for caspofungin and fluconazole as shown in Table 14.
Table 14: Favorable Response Rates for Patients with Esophageal Candidiasis*
Caspofungin
Fluconazole
% Difference† (95% CI)
Day 5 to 7 post-treatment
66/81 (81.5%)
80/94 (85.1%)
-3.6 (-14.7, 7.5)
* Analysis excluded patients without documented esophageal
candidiasis or patients not receiving at least 1 day of study therapy.
† Calculated as caspofungin – fluconazole.
The proportion of patients with a favorable symptom response was also comparable (90.1% and 89.4% for caspofungin and fluconazole, respectively). In addition, the proportion of patients with a favorable endoscopic response was comparable (85.2% and 86.2% for caspofungin and fluconazole, respectively).
As shown in Table 15, the esophageal candidiasis relapse rates at the Day 14 post-treatment visit were similar for the two groups. At the Day 28 post-treatment visit, the group treated with caspofungin had a numerically higher incidence of relapse; however, the difference was not statistically significant.
Table 15: Relapse Rates at 14 and 28 Days Post-Therapy in Patients with Esophageal Candidiasis at Baseline
Caspofungin
Fluconazole
% Difference*
(95% CI)
Day 14 post-treatment
7/66 (10.6%)
6/76 (7.9%)
2.7 (-6.9, 12.3)
Day 28 post-treatment
18/64 (28.1%)
12/72 (16.7%)
11.5 (-2.5, 25.4)
* Calculated as caspofungin – fluconazole.
In this trial, which was designed to establish noninferiority of caspofungin to fluconazole for the treatment of esophageal candidiasis, 122 (70%) patients also had oropharyngeal candidiasis. A favorable response was defined as complete resolution of all symptoms of oropharyngeal disease and all visible oropharyngeal lesions. The proportion of patients with a favorable oropharyngeal response at the 5- to 7-day post-treatment visit was numerically lower for caspofungin; however, the difference was not statistically significant. Oropharyngeal candidiasis relapse rates at Day 14 and Day 28 post-treatment visits were statistically significantly higher for caspofungin than for fluconazole. The results are shown in Table 16.
Table 16: Oropharyngeal Candidiasis Response Rates at 5 to 7 Days Post-Therapy and Relapse Rates at 14 and 28 Days Post-Therapy in Patients with Oropharyngeal and Esophageal Candidiasis at Baseline
Caspofungin
Fluconazole
% Difference*
(95% CI)
Response Rate Day 5 to 7 post-treatment
40/56 (71.4%)
55/66 (83.3%)
-11.9 (-26.8, 3.0)
Relapse Rate Day 14 post-treatment
17/40 (42.5%)
7/53 (13.2%)
29.3 (11.5, 47.1)
Relapse Rate Day 28 post-treatment
23/39 (59.0%)
18/51 (35.3%)
23.7 (3.4, 43.9)
* Calculated as caspofungin – fluconazole.
The results from the two smaller dose-ranging studies corroborate the efficacy of caspofungin for esophageal candidiasis that was demonstrated in the larger study.
Caspofungin was associated with favorable outcomes in 7 of 10 esophageal C. albicans infections refractory to at least 200 mg of fluconazole given for 7 days, although the in vitro susceptibility of the infecting isolates to fluconazole was not known.
14.4 Invasive Aspergillosis
Sixty-nine patients between the ages of 18 and 80 with invasive aspergillosis were enrolled in an open-label, noncomparative study to evaluate the safety, tolerability, and efficacy of caspofungin. Enrolled patients had previously been refractory to or intolerant of other antifungal therapy(ies). Refractory patients were classified as those who had disease progression or failed to improve despite therapy for at least 7 days with amphotericin B, lipid formulations of amphotericin B, itraconazole, or an investigational azole with reported activity against Aspergillus. Intolerance to previous therapy was defined as a doubling of creatinine (or creatinine 2.5 mg/dL or greater while on therapy), other acute reactions, or infusion-related toxicity. To be included in the study, patients with pulmonary disease must have had definite (positive tissue histopathology or positive culture from tissue obtained by an invasive procedure) or probable (positive radiographic or computed tomography evidence with supporting culture from bronchoalveolar lavage or sputum, galactomannan enzyme-linked immunosorbent assay, and/or polymerase chain reaction) invasive aspergillosis. Patients with extrapulmonary disease had to have definite invasive aspergillosis. Patients were administered a single 70 mg loading dose of caspofungin and subsequently dosed with 50 mg daily. The mean duration of therapy was 33.7 days, with a range of 1 to 162 days.
An independent expert panel evaluated patient data, including diagnosis of invasive aspergillosis, response and tolerability to previous antifungal therapy, treatment course on caspofungin, and clinical outcome.
A favorable response was defined as either complete resolution (complete response) or clinically meaningful improvement (partial response) of all signs and symptoms and attributable radiographic findings. Stable, nonprogressive disease was considered to be an unfavorable response.
Among the 69 patients enrolled in the study, 63 met entry diagnostic criteria and had outcome data; and of these, 52 patients received treatment for greater than 7 days. Fifty-three (84%) were refractory to previous antifungal therapy and 10 (16%) were intolerant. Forty-five patients had pulmonary disease and 18 had extrapulmonary disease. Underlying conditions were hematologic malignancy (N=24), allogeneic bone marrow transplant or stem cell transplant (N=18), organ transplant (N=8), solid tumor (N=3), or other conditions (N=10). All patients in the study received concomitant therapies for their other underlying conditions. Eighteen patients received tacrolimus and caspofungin concomitantly, of whom 8 also received mycophenolate mofetil.
Overall, the expert panel determined that 41% (26/63) of patients receiving at least one dose of caspofungin had a favorable response. For those patients who received greater than 7 days of therapy with caspofungin, 50% (26/52) had a favorable response. The favorable response rates for patients who were either refractory to or intolerant of previous therapies were 36% (19/53) and 70% (7/10), respectively. The response rates among patients with pulmonary disease and extrapulmonary disease were 47% (21/45) and 28% (5/18), respectively. Among patients with extrapulmonary disease, 2 of 8 patients who also had definite, probable, or possible CNS involvement had a favorable response. Two of these 8 patients had progression of disease and manifested CNS involvement while on therapy.
Caspofungin is effective for the treatment of invasive aspergillosis in patients who are refractory to or intolerant of itraconazole, amphotericin B, and/or lipid formulations of amphotericin B. However, the efficacy of caspofungin for initial treatment of invasive aspergillosis has not been evaluated in comparator-controlled clinical studies.
14.5 Pediatric Patients
The safety and efficacy of caspofungin were evaluated in pediatric patients 3 months to 17 years of age in two prospective, multicenter clinical trials.
The first study, which enrolled 82 patients between 2 to 17 years of age, was a randomized, double-blind study comparing caspofungin (50 mg/m2 intravenously once daily following a 70 mg/m2 loading dose on Day 1 [not to exceed 70 mg daily]) to AmBisome (3 mg/kg intravenously daily) in a 2:1 treatment fashion (56 on caspofungin, 26 on AmBisome) as empirical therapy in pediatric patients with persistent fever and neutropenia. The study design and criteria for efficacy assessment were similar to the study in adult patients [see Clinical Studies (14.1)]. Patients were stratified based on risk category (high-risk patients had undergone allogeneic stem cell transplantation or had relapsed acute leukemia). Twenty-seven percent of patients in both treatment groups were high risk. Favorable overall response rates of pediatric patients with persistent fever and neutropenia are presented in Table 17.
Table 17: Favorable Overall Response Rates of Pediatric Patients with Persistent Fever and Neutropenia
Caspofungin
AmBisome*
Number of Patients
56
25
Overall Favorable Response
26/56 (46.4%)
8/25 (32.0%)
High risk
9/15 (60.0%)
0/7 (0.0%)
Low risk
17/41 (41.5%)
8/18 (44.4%)
* One patient excluded from analysis due to no fever at study entry.
The second study was a prospective, open-label, non-comparative study estimating the safety and efficacy of caspofungin in pediatric patients (ages 3 months to 17 years) with candidemia and other Candida infections, esophageal candidiasis, and invasive aspergillosis (as salvage therapy). The study employed diagnostic criteria which were based on established EORTC/MSG criteria of proven or probable infection; these criteria were similar to those criteria employed in the adult studies for these various indications. Similarly, the efficacy time points and endpoints used in this study were similar to those employed in the corresponding adult studies [see Clinical Studies (14.2,14.3, and 14.4)]. All patients received caspofungin at 50 mg/m2 intravenously once daily following a 70 mg/m2 loading dose on Day 1 (not to exceed 70 mg daily). Among the 49 enrolled patients who received caspofungin, 48 were included in the efficacy analysis (one patient excluded due to not having a baseline Aspergillus or Candida infection). Of these 48 patients, 37 had candidemia or other Candida infections, 10 had invasive aspergillosis, and 1 patient had esophageal candidiasis. Most candidemia and other Candida infections were caused by C. albicans (35%), followed by C. parapsilosis (22%), C. tropicalis (14%), and C. glabrata (11%). The favorable response rate, by indication, at the end of caspofungin therapy was as follows: 30/37 (81%) in candidemia or other Candida infections, 5/10 (50%) in invasive aspergillosis, and 1/1 in esophageal candidiasis.
- 15 REFERENCES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Caspofungin acetate for injection is a lyophilized white to off-white cake or powder for intravenous infusion, supplied in single-dose vials with a rubber stopper and an aluminum seal as follows:
Product Code Caspofungin
Acetate Total
Product Strength
(Concentration)Carton
NDC and
Package
ConfigurationVial NDC 356110 50 mg per vial 63323-356-10
10 Single-Dose Vials63323-356-01 358110 70 mg per vial 63323-358-10
10 Single-Dose Vials63323-358-01 Storage and Handling
The lyophilized vials should be stored at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Reconstituted ConcentrateReconstituted caspofungin acetate for injection in the vial may be stored at ≤ 25°C (≤ 77°F) for one hour prior to the preparation of the patient infusion solution.
Diluted Product
The final patient infusion solution in the intravenous bag or bottle can be stored at ≤ 25°C (≤ 77°F) for 24 hours or at 2° to 8°C (36° to 46°F) for 48 hours.
The container closure is not made with natural rubber latex.
-
17 PATIENT COUNSELING INFORMATION
Hypersensitivity
Inform patients that anaphylactic reactions have been reported during administration of caspofungin. Caspofungin can cause hypersensitivity reactions, including rash, facial swelling, angioedema, pruritus, sensation of warmth, or bronchospasm. Inform patients to report these signs or symptoms to their healthcare providers.
Hepatic Effects
Inform patients that there have been isolated reports of serious hepatic effects from caspofungin therapy.
The brand names mentioned in this document are the trademarks of their respective owners.

Lake Zurich, IL 60047
www.fresenius-kabi.com/us
451328C -
PRINCIPAL DISPLAY PANEL
PACKAGE LABEL - PRINCIPAL DISPLAY - Caspofungin Acetate- 50 mg Single-Dose Vial Label
Caspofungin Acetate for Injection
50 mg per vial*
For intravenous infusion after dilution.
Preservative free.
Discard unused portion.
Single-Dose Vial
Rx only
PACKAGE LABEL - PRINCIPAL DISPLAY - Caspofungin Acetate- 50 mg Single-Dose Vial Carton Panel
NDC 63323-356-10
Caspofungin Acetate for Injection
50 mg per vial*
For intravenous infusion after dilution.
Preservative free.
Discard unused portion.
Rx only
10 Single-Dose Vials
Do not use Diluents
containing Dextrose
(α-D-glucose)

PACKAGE LABEL - PRINCIPAL DISPLAY - Caspofungin Acetate - 70 mg Single-Dose Vial Label
Caspofungin Acetate for Injection
70 mg per vial*
For intravenous infusion after dilution.
Preservative free.
Discard unused portion.
Single-Dose Vial
Rx only

PACKAGE LABEL - PRINCIPAL DISPLAY - Caspofungin Acetate - 70 mg Single-Dose Vial Carton Panel
NDC 63323-358-10
Caspofungin Acetate for Injection
70 mg per vial*
For intravenous infusion after dilution.
Preservative free.
Discard unused portion.
Rx only
10 Single-Dose Vials
Do not use Diluents
containing Dextrose
(α-D-glucose)

-
INGREDIENTS AND APPEARANCE
CASPOFUNGIN ACETATE
caspofungin acetate injection, powder, lyophilized, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 63323-356 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CASPOFUNGIN ACETATE (UNII: VUW370O5QE) (CASPOFUNGIN - UNII:F0XDI6ZL63) CASPOFUNGIN ACETATE 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength ARGININE (UNII: 94ZLA3W45F) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 63323-356-10 10 in 1 CARTON 12/30/2016 1 NDC: 63323-356-01 10.8 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA206110 12/30/2016 CASPOFUNGIN ACETATE
caspofungin acetate injection, powder, lyophilized, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 63323-358 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CASPOFUNGIN ACETATE (UNII: VUW370O5QE) (CASPOFUNGIN - UNII:F0XDI6ZL63) CASPOFUNGIN ACETATE 7 mg in 1 mL Inactive Ingredients Ingredient Name Strength ARGININE (UNII: 94ZLA3W45F) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 63323-358-10 10 in 1 CARTON 12/30/2016 1 NDC: 63323-358-01 10.8 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA206110 12/30/2016 Labeler - Fresenius Kabi USA, LLC (608775388) Establishment Name Address ID/FEI Business Operations Fresenius Kabi USA, LLC 840771732 MANUFACTURE(63323-356, 63323-358)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.