FEXOFENADINE HYDROCHLORIDE AND PSEUDOEPHEDRINE HYDROCHLORIDE tablet, film coated, extended release
Fexofenadine Hydrochloride and Pseudoephedrine Hydrochloride by
Drug Labeling and Warnings
Fexofenadine Hydrochloride and Pseudoephedrine Hydrochloride by is a Otc medication manufactured, distributed, or labeled by CVS Pharmacy, Inc., Aurohealth LLC, Aurobindo Pharma Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Drug Facts
- Purpose
-
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- reduces swelling of nasal passages
- temporarily relieves sinus congestion and pressure
- temporarily restores freer breathing through the nose
-
Warnings
Do not use
- if you have ever had an allergic reaction to this product or any of its ingredients
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- if you have difficulty swallowing
- Ask a doctor before use if you have
-
When using this product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
- the tablet coating may be seen in the stool (this is normal). Continue to take as directed (see Directions).
- Stop use and ask a doctor if
- Keep out of reach of children.
-
Directions
- do not divide, crush, chew or dissolve the tablet; swallow tablet whole
adults and children 12 years of age and over
take 1 tablet with a glass of water every 12 hours on an empty stomach; do not take more than 2 tablets in 24 hours
children under 12 years of age
do not use
adults 65 years of age and older
ask a doctor
consumers with kidney disease
ask a doctor
Other information
- safety sealed: do not use if carton is opened or if individual blister units are torn or opened.
- store at 20-25°C (68-77°F).
- Meets USP dissolution test 7
-
Inactive ingredients
colloidal silicon dioxide, croscarmellose sodium, ferric oxide yellow, hydroxypropyl cellulose, hypromellose, lactose monohydrate, microcrystalline cellulose, polyethylene glycol, pregelatinized starch (maize), stearic acid
Questions or Comments?
call 1-855-274-4122
Distributed by: CVS Pharmacy, Inc.
One CVS Drive, Woonsocket, RI 02895
© 2024 CVS/pharmacy
CVS.com® 1-800-SHOP CVS
Made in India
Code: TS/DRUGS/22/2009 -
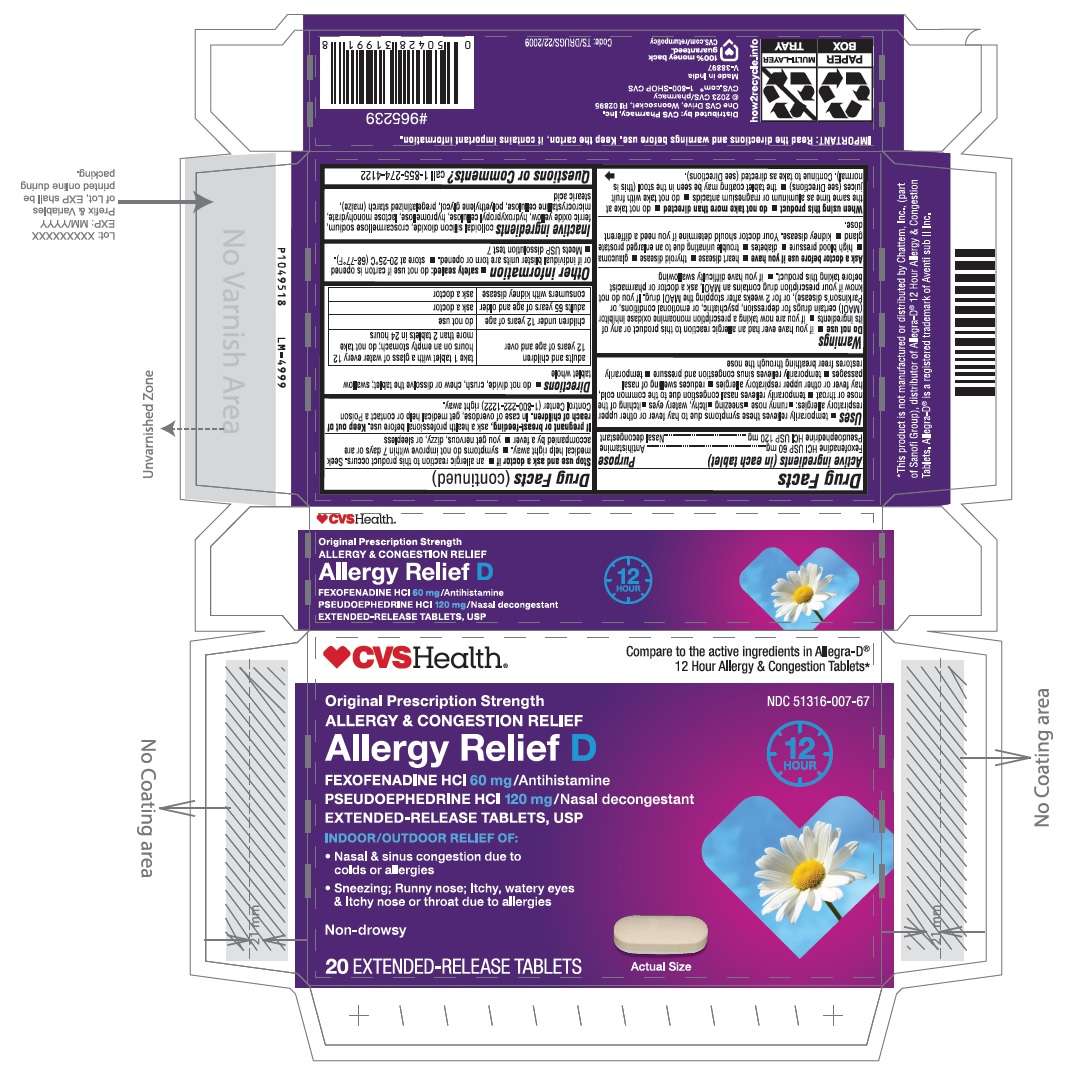
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 2 x 10 Blister Carton
CVSHealth®
Compare to the active ingredients in Allegra-D®
12 Hour Allergy & Congestion Tablets*
NDC: 51316-007-67
Non-Drowsy
ALLERGY & CONGESTION RELIRF
Allergy Relief D
Fexofenadine HCl 60 mg/Antihistamine
PSEUDOEPHEDRINE HCI 120 mg / Nasal decongestant
EXTENDED-RELEASE TABLETS, USP
12
HOUR
INDOOR/OUTDOOR RELIEF OF:
Nasal & sinus congestion due to
colds or allergiesSneezing; Runny nose; Itchy, watery eyes
& Itchy nose or throat due to allergies
20 EXTENDED-RELEASE TABLETS Actual Size
-
INGREDIENTS AND APPEARANCE
FEXOFENADINE HYDROCHLORIDE AND PSEUDOEPHEDRINE HYDROCHLORIDE
fexofenadine hydrochloride and pseudoephedrine hydrochloride tablet, film coated, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 51316-007 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FEXOFENADINE HYDROCHLORIDE (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) FEXOFENADINE HYDROCHLORIDE 60 mg PSEUDOEPHEDRINE HYDROCHLORIDE (UNII: 6V9V2RYJ8N) (PSEUDOEPHEDRINE - UNII:7CUC9DDI9F) PSEUDOEPHEDRINE HYDROCHLORIDE 120 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) HYPROMELLOSE 2208 (100000 MPA.S) (UNII: VM7F0B23ZI) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) HYDROXYPROPYL CELLULOSE (110000 WAMW) (UNII: 5Y0974F5PW) POLYETHYLENE GLYCOL 6000 (UNII: 30IQX730WE) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) STARCH, CORN (UNII: O8232NY3SJ) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color YELLOW (White to Off White Layer and Yellow to Pale Yellow) Score no score Shape CAPSULE (Bincovex) Size 19mm Flavor Imprint Code Z;79 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 51316-007-67 2 in 1 CARTON 11/17/2022 1 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC: 51316-007-84 3 in 1 CARTON 11/17/2022 2 10 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209116 11/17/2022 Labeler - CVS Pharmacy, Inc. (062312574) Registrant - Aurohealth LLC (078728447) Establishment Name Address ID/FEI Business Operations Aurobindo Pharma Limited 650381903 ANALYSIS(51316-007) , MANUFACTURE(51316-007)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.