BLISTEX MEDICATED (dimethicone, camphor- synthetic, menthol, and phenol ointment
Blistex by
Drug Labeling and Warnings
Blistex by is a Otc medication manufactured, distributed, or labeled by Blistex Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
- Warnings
- Directions
-
Inactive ingredients
allantoin, ammonium hydroxide, beeswax, calcium disodium EDTA, calcium hydroxide, cetyl alcohol, flavors, glycerin, hydrated silica, lanolin, lauric acid, mineral oil, myristic acid, oleic acid, palmitic acid, paraffin, petrolatum, polyglyceryl-3 diisostearate, potassium hydroxide, purified water, SD alcohol 36, sodium hydroxide, sodium saccharin, stearyl alcohol
-
PRINCIPAL DISPLAY PANEL - 6 g Tube Carton
NDC: 10157-9951-2
Blistex®
MEDICATED
LIP OINTMENTLIP PROTECTANT/
EXTERNAL ANALGESICNET WT..21oz(6g)
Helps Heal Severe Dryness And Relieve Cold Sores
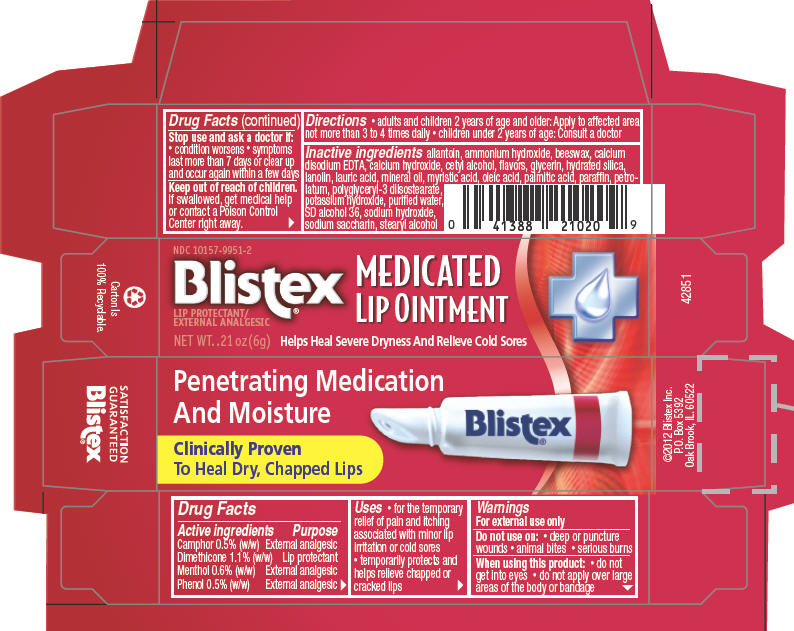
-
INGREDIENTS AND APPEARANCE
BLISTEX MEDICATED
dimethicone, camphor (synthetic), menthol, and phenol ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 10157-9951 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Dimethicone (UNII: 92RU3N3Y1O) (Dimethicone - UNII:92RU3N3Y1O) Dimethicone 1.1 g in 100 g Camphor (synthetic) (UNII: 5TJD82A1ET) (Camphor (synthetic) - UNII:5TJD82A1ET) Camphor (synthetic) 0.5 g in 100 g Menthol, unspecified form (UNII: L7T10EIP3A) (Menthol, unspecified form - UNII:L7T10EIP3A) Menthol, unspecified form 0.625 g in 100 g Phenol (UNII: 339NCG44TV) (Phenol - UNII:339NCG44TV) Phenol 0.5 g in 100 g Inactive Ingredients Ingredient Name Strength allantoin (UNII: 344S277G0Z) ammonia (UNII: 5138Q19F1X) yellow wax (UNII: 2ZA36H0S2V) edetate calcium disodium anhydrous (UNII: 8U5D034955) calcium hydroxide (UNII: PF5DZW74VN) cetyl alcohol (UNII: 936JST6JCN) glycerin (UNII: PDC6A3C0OX) hydrated silica (UNII: Y6O7T4G8P9) lanolin (UNII: 7EV65EAW6H) lauric acid (UNII: 1160N9NU9U) mineral oil (UNII: T5L8T28FGP) myristic acid (UNII: 0I3V7S25AW) oleic acid (UNII: 2UMI9U37CP) palmitic acid (UNII: 2V16EO95H1) paraffin (UNII: I9O0E3H2ZE) petrolatum (UNII: 4T6H12BN9U) polyglyceryl-3 diisostearate (UNII: 46P231IQV8) potassium hydroxide (UNII: WZH3C48M4T) water (UNII: 059QF0KO0R) sodium hydroxide (UNII: 55X04QC32I) saccharin sodium (UNII: SB8ZUX40TY) stearyl alcohol (UNII: 2KR89I4H1Y) Product Characteristics Color WHITE Score Shape Size Flavor MINT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 10157-9951-1 6 g in 1 TUBE; Type 0: Not a Combination Product 12/01/2000 2 NDC: 10157-9951-3 10 g in 1 TUBE; Type 0: Not a Combination Product 12/01/2000 3 NDC: 10157-9951-4 2.25 g in 1 TUBE; Type 0: Not a Combination Product 12/01/2000 4 NDC: 10157-9951-5 8 g in 1 TUBE; Type 0: Not a Combination Product 12/01/2000 5 NDC: 10157-9951-2 1 in 1 CARTON 12/01/2000 5 NDC: 10157-9951-6 6 g in 1 TUBE; Type 0: Not a Combination Product 6 NDC: 10157-9951-7 3 in 1 CARTON 12/01/2000 6 6 g in 1 TUBE; Type 0: Not a Combination Product 7 NDC: 10157-9951-8 4 in 1 CARTON 12/01/2000 7 6 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part348 12/01/2000 Labeler - Blistex Inc. (005126354) Establishment Name Address ID/FEI Business Operations Blistex Inc. 005126354 MANUFACTURE(10157-9951)
Trademark Results [Blistex]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 BLISTEX 88143446 5924118 Live/Registered |
Blistex Inc. 2018-10-04 |
 BLISTEX 78233453 2817472 Live/Registered |
BLISTEX INC. 2003-04-03 |
 BLISTEX 74421493 1897266 Dead/Cancelled |
Blistex Bracken Limited Partnership 1993-08-04 |
 BLISTEX 73541344 1373396 Live/Registered |
BLISTEX BRACKEN LIMITED PARTNERSHIP 1985-06-05 |
 BLISTEX 73136745 1094204 Live/Registered |
BRACKEN, JIM L. 1977-08-08 |
 BLISTEX 72226892 0804558 Live/Registered |
BLISTEX COMPANY 1965-09-01 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.