MAXIMUM-H- hydrocortisone cream
Maximum-H by
Drug Labeling and Warnings
Maximum-H by is a Otc medication manufactured, distributed, or labeled by New GPC, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active Ingredient
- Purpose
- Uses
-
Inactive ingredients
Water, Petrolatum, Hydrogenated Didecene Cetearyl Alcohol, Ceteareth-20, Lanolin, Mineral Oil, Glycerin, Sorbitan Stearate, Dimethicone, Methylpropanediol, PEG-40 Stearate, Lanolin Alcohol, Esculin, Disodium EDTA, Magnesium Aluminum Silicate, Acrylates/10-30 Alkyl Acrylate, Grosspolymer, 1,2-Hexanediol, Caprylyl Glycol, BHT, Soy, Isoflavones, Sodium Hydroxide, Phenoxyethanol, Sorbic Acid, Menthol, Tricaprylin, Polymethyl Methacrylate, Polyacrylamide, C13-14 Isoparafin, Laureth-7, Citric Acid.
- Questions or Comments?
- WARNINGS
-
Directions
Adults and Children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily.
Children under 2 years of age: Do not use, consult a Doctor.
For hemorrhoids - clean affected area with a cleansing wipe, then dry with a soft cloth. Apply externally to lower part of anal canal only. Apply up to 3 times daily at night and after every bowel movement.
- PRINCIPAL DISPLAY PANEL - 20g Tube Carton
-
INGREDIENTS AND APPEARANCE
MAXIMUM-H
hydrocortisone creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 65588-2210 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE (UNII: WI4X0X7BPJ) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE 1 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) EDETATE DISODIUM (UNII: 7FLD91C86K) MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) METHYLPROPANEDIOL (UNII: N8F53B3R4R) GLYCERIN (UNII: PDC6A3C0OX) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) PETROLATUM (UNII: 4T6H12BN9U) CAPRYLYL GLYCOL (UNII: 00YIU5438U) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) MINERAL OIL (UNII: T5L8T28FGP) LANOLIN ALCOHOLS (UNII: 884C3FA9HE) LANOLIN (UNII: 7EV65EAW6H) SORBITAN MONOSTEARATE (UNII: NVZ4I0H58X) POLYOXYL 40 STEARATE (UNII: 13A4J4NH9I) DIMETHICONE (UNII: 92RU3N3Y1O) TROLAMINE (UNII: 9O3K93S3TK) SOY ISOFLAVONES (UNII: 71B37NR06D) PHENOXYETHANOL (UNII: HIE492ZZ3T) SORBIC ACID (UNII: X045WJ989B) MENTHOL (UNII: L7T10EIP3A) TRICAPRYLIN (UNII: 6P92858988) ESCULIN (UNII: 1Y1L18LQAF) C13-14 ISOPARAFFIN (UNII: E4F12ROE70) LAURETH-7 (UNII: Z95S6G8201) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 65588-2210-1 1 in 1 CARTON 1 20 g in 1 TUBE 2 NDC: 65588-2210-2 1 in 1 CARTON 2 40 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part348 12/01/2009 Labeler - New GPC, Inc. (895025443) Establishment Name Address ID/FEI Business Operations New GPC, Inc. 895025443 MANUFACTURE
Trademark Results [Maximum-H]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
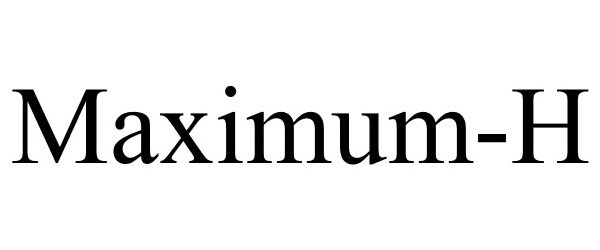 MAXIMUM-H 78829827 3747845 Dead/Cancelled |
Moss Pharmaceuticals LLC 2006-03-06 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
