DEXMEDETOMIDINE injection, solution, concentrate
Dexmedetomidine Hydrochloride by
Drug Labeling and Warnings
Dexmedetomidine Hydrochloride by is a Prescription medication manufactured, distributed, or labeled by Mylan Institutional LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use DEXMEDETOMIDINE INJECTION safely and effectively. See full prescribing information for DEXMEDETOMIDINE INJECTION.
DEXMEDETOMIDINE injection for intravenous use
Initial U.S. Approval: 1999INDICATIONS AND USAGE
Dexmedetomidine injection is a relatively selective alpha2-adrenergic agonist indicated for:
- Sedation of initially intubated and mechanically ventilated patients during treatment in an intensive care setting. Administer dexmedetomidine injection by continuous infusion not to exceed 24 hours. (1.1)
- Sedation of non-intubated patients prior to and/or during surgical and other procedures. (1.2)
DOSAGE AND ADMINISTRATION
- Individualize and titrate dexmedetomidine injection dosing to desired clinical effect. (2.1)
- Administer dexmedetomidine injection using a controlled infusion device. (2.1)
- Dilute the 200 mcg/2 mL (100 mcg/mL) vial contents in 0.9% sodium chloride solution to achieve required concentration (4 mcg/mL) prior to administration. (2.4)
For Adult Intensive Care Unit Sedation: Generally initiate at one mcg/kg over 10 minutes, followed by a maintenance infusion of 0.2 to 0.7 mcg/kg/hour. (2.2)
For Adult Procedural Sedation: Generally initiate at one mcg/kg over 10 minutes, followed by a maintenance infusion initiated at 0.6 mcg/kg/hour and titrated to achieve desired clinical effect with doses ranging from 0.2 to 1 mcg/kg/hour. (2.2)
Alternative Doses: Recommended for patients over 65 years of age and awake fiberoptic intubation patients. (2.2)
DOSAGE FORMS AND STRENGTHS
Dexmedetomidine Injection, 200 mcg/2 mL (100 mcg/mL) in a glass vial. To be used after dilution. (3)
CONTRAINDICATIONS
None (4)
WARNINGS AND PRECAUTIONS
- Monitoring: Continuously monitor patients while receiving dexmedetomidine injection. (5.1)
- Bradycardia and Sinus Arrest: Have occurred in young healthy volunteers with high vagal tone or with different routes of administration, e.g., rapid intravenous or bolus administration. (5.2)
- Hypotension and Bradycardia: May necessitate medical intervention. May be more pronounced in patients with hypovolemia, diabetes mellitus, or chronic hypertension, and in the elderly. Use with caution in patients with advanced heart block or severe ventricular dysfunction. (5.2)
- Co-administration with Other Vasodilators or Negative Chronotropic Agents: Use with caution due to additive pharmacodynamic effects. (5.2)
- Transient Hypertension: Observed primarily during the loading dose. Consider reduction in loading infusion rate. (5.3)
- Arousability: Patients can become aroused/alert with stimulation; this alone should not be considered as lack of efficacy. (5.4)
- Prolonged exposure to dexmedetomidine beyond 24 hours may be associated with tolerance and tachyphylaxis and a dose-related increase in adverse events. (5.6)
ADVERSE REACTIONS
- The most common adverse reactions (incidence greater than 2%) are hypotension, bradycardia, and dry mouth. (6.1)
- Adverse reactions associated with infusions greater than 24 hours in duration include ARDS, respiratory failure, and agitation. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Mylan at 1-877-446-3679 (1-877-4-INFO-RX) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Anesthetics, Sedatives, Hypnotics, Opioids: Enhancement of pharmacodynamic effects. Reduction in dosage of dexmedetomidine injection or the concomitant medication may be required. (7.1)
USE IN SPECIFIC POPULATIONS
- Geriatric Patients: Dose reduction should be considered. (2.2, 2.3, 5.1, 8.5)
- Hepatic Impairment: Dose reduction should be considered. (2.1, 2.2, 2.3, 5.7, 8.6)
- Pregnancy: Based on animal data, may cause fetal harm. (8.1)
- Nursing Mothers: Caution should be exercised when administered to a nursing woman. (8.3)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 10/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Intensive Care Unit Sedation
1.2 Procedural Sedation
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Guidelines
2.2 Dosage Information
2.3 Dosage Adjustment
2.4 Preparation of Solution
2.5 Administration with Other Fluids
2.6 Compatibility with Natural Rubber
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Drug Administration
5.2 Hypotension, Bradycardia, and Sinus Arrest
5.3 Transient Hypertension
5.4 Arousability
5.5 Withdrawal
5.6 Tolerance and Tachyphylaxis
5.7 Hepatic Impairment
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Anesthetics, Sedatives, Hypnotics, Opioids
7.2 Neuromuscular Blockers
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Labor and Delivery
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
9.3 Dependence
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Pharmacology and/or Toxicology
14 CLINICAL STUDIES
14.1 Intensive Care Unit Sedation
14.2 Procedural Sedation
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Intensive Care Unit Sedation
Dexmedetomidine injection is indicated for sedation of initially intubated and mechanically ventilated patients during treatment in an intensive care setting. Dexmedetomidine injection should be administered by continuous infusion not to exceed 24 hours.
Dexmedetomidine injection has been continuously infused in mechanically ventilated patients prior to extubation, during extubation, and post-extubation. It is not necessary to discontinue dexmedetomidine injection prior to extubation.
-
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Guidelines
- Dexmedetomidine injection dosing should be individualized and titrated to desired clinical response.
- Dexmedetomidine injection is not indicated for infusions lasting longer than 24 hours.
- Dexmedetomidine injection should be administered using a controlled infusion device.
2.2 Dosage Information
Table 1: Dosage Information INDICATION
DOSAGE AND ADMINISTRATION
Initiation of Intensive Care Unit Sedation
For adult patients: a loading infusion of one mcg/kg over 10 minutes.
For adult patients being converted from alternate sedative therapy: a loading dose may not be required [see Dosage and Administration (2.2)].
For patients over 65 years of age: a dose reduction should be considered [see Use in Specific Populations (8.5)].
For adult patients with impaired hepatic-function: a dose reduction should be considered [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
Maintenance of Intensive Care Unit Sedation
For adult patients: a maintenance infusion of 0.2 to 0.7 mcg/kg/hour. The rate of the maintenance infusion should be adjusted to achieve the desired level of sedation.
For patients over 65 years of age: a dose reduction should be considered [see Use in Specific Populations (8.5)].
For adult patients with impaired hepatic function: a dose reduction should be considered [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
Initiation of Procedural Sedation
For adult patients: a loading infusion of one mcg/kg over 10 minutes. For less invasive procedures such as ophthalmic surgery, a loading infusion of 0.5 mcg/kg given over 10 minutes may be suitable.
For awake fiberoptic intubation in adult patients: a loading infusion of one mcg/kg over 10 minutes.
For patients over 65 years of age: a loading infusion of 0.5 mcg/ kg over 10 minutes [see Use in Specific Populations (8.5)].
For adult patients with impaired hepatic function: a dose reduction should be considered [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
Maintenance of Procedural Sedation
For adult patients: the maintenance infusion is generally initiated at 0.6 mcg/kg/hour and titrated to achieve desired clinical effect with doses ranging from 0.2 to 1 mcg/kg/hour. The rate of the maintenance infusion should be adjusted to achieve the targeted level of sedation.
For awake fiberoptic intubation in adult patients: a maintenance infusion of 0.7 mcg/kg/hour is recommended until the endotracheal tube is secured.
For patients over 65 years of age: a dose reduction should be considered [see Use in Specific Populations (8.5)].
For adult patients with impaired hepatic function: a dose reduction should be considered [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
2.3 Dosage Adjustment
Due to possible pharmacodynamic interactions, a reduction in dosage of dexmedetomidine injection or other concomitant anesthetics, sedatives, hypnotics or opioids may be required when co-administered [see Drug Interactions (7.1)].
Dosage reductions may need to be considered for adult patients with hepatic impairment, and geriatric patients [see Warnings and Precautions (5.7), Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
2.4 Preparation of Solution
Strict aseptic technique must always be maintained during handling of dexmedetomidine injection.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Dexmedetomidine Injection, 200 mcg/2 mL (100 mcg/mL)
Dexmedetomidine injection must be diluted with 0.9% sodium chloride injection to achieve required concentration (4 mcg/mL) prior to administration. Preparation of solutions is the same, whether for the loading dose or maintenance infusion.
To prepare the infusion, withdraw 2 mL of dexmedetomidine injection and add to 48 mL of 0.9% sodium chloride injection to a total of 50 mL. Shake gently to mix well.
Discard unused portion.
2.5 Administration with Other Fluids
Dexmedetomidine injection infusion should not be co-administered through the same intravenous catheter with blood or plasma because physical compatibility has not been established.
Dexmedetomidine injection has been shown to be incompatible when administered with the following drugs: amphotericin B, diazepam.
Dexmedetomidine injection has been shown to be compatible when administered with the following intravenous fluids:
- 0.9% sodium chloride in water
- 5% dextrose in water
- 20% mannitol
- Lactated Ringer's solution
- 100 mg/mL magnesium sulfate solution
- 0.3% potassium chloride solution
2.6 Compatibility with Natural Rubber
Compatibility studies have demonstrated the potential for absorption of dexmedetomidine hydrochloride to some types of natural rubber. Although dexmedetomidine injection is dosed to effect, it is advisable to use administration components made with synthetic or coated natural rubber gaskets.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Drug Administration
Dexmedetomidine injection should be administered only by persons skilled in the management of patients in the intensive care or operating room setting. Due to the known pharmacological effects of dexmedetomidine injection, patients should be continuously monitored while receiving dexmedetomidine injection.
5.2 Hypotension, Bradycardia, and Sinus Arrest
Clinically significant episodes of bradycardia and sinus arrest have been reported with dexmedetomidine injection administration in young, healthy adult volunteers with high vagal tone or with different routes of administration including rapid intravenous or bolus administration.
Reports of hypotension and bradycardia have been associated with dexmedetomidine injection infusion. Some of these cases have resulted in fatalities. If medical intervention is required, treatment may include decreasing or stopping the infusion of dexmedetomidine injection, increasing the rate of intravenous fluid administration, elevation of the lower extremities, and use of pressor agents. Because dexmedetomidine injection has the potential to augment bradycardia induced by vagal stimuli, clinicians should be prepared to intervene. The intravenous administration of anticholinergic agents (e.g., glycopyrrolate, atropine) should be considered to modify vagal tone. In clinical trials, glycopyrrolate or atropine were effective in the treatment of most episodes of dexmedetomidine hydrochloride-induced bradycardia. However, in some patients with significant cardiovascular dysfunction, more advanced resuscitative measures were required.
Caution should be exercised when administering dexmedetomidine injection to patients with advanced heart block and/or severe ventricular dysfunction. Because dexmedetomidine injection decreases sympathetic nervous system activity, hypotension and/or bradycardia may be expected to be more pronounced in patients with hypovolemia, diabetes mellitus, or chronic hypertension and in elderly patients.
In clinical trials where other vasodilators or negative chronotropic agents were co-administered with dexmedetomidine injection an additive pharmacodynamic effect was not observed. Nonetheless, caution should be used when such agents are administered concomitantly with dexmedetomidine injection.
5.3 Transient Hypertension
Transient hypertension has been observed primarily during the loading dose in association with the initial peripheral vasoconstrictive effects of dexmedetomidine injection. Treatment of the transient hypertension has generally not been necessary, although reduction of the loading infusion rate may be desirable.
5.4 Arousability
Some patients receiving dexmedetomidine injection have been observed to be arousable and alert when stimulated. This alone should not be considered as evidence of lack of efficacy in the absence of other clinical signs and symptoms.
5.5 Withdrawal
Intensive Care Unit Sedation
With administration up to 7 days, regardless of dose, 12 (5%) dexmedetomidine injection adult subjects experienced at least 1 event related to withdrawal within the first 24 hours after discontinuing study drug and 7 (3%) dexmedetomidine injection adult subjects experienced at least 1 event 24 to 48 hours after end of study drug. The most common events were nausea, vomiting, and agitation.
In adult subjects, tachycardia and hypertension requiring intervention in the 48 hours following study drug discontinuation occurred at frequencies of < 5%. If tachycardia and/or hypertension occurs after discontinuation of dexmedetomidine injection supportive therapy is indicated.
5.6 Tolerance and Tachyphylaxis
Use of dexmedetomidine beyond 24 hours has been associated with tolerance and tachyphylaxis and a dose-related increase in adverse reactions [see Adverse Reactions (6.1)].
5.7 Hepatic Impairment
Since dexmedetomidine clearance decreases with severity of hepatic impairment, dose reduction should be considered in patients with impaired hepatic function [see Dosage and Administration (2.2)].
-
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reactions rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in practice.
Use of dexmedetomidine injection has been associated with the following serious adverse reactions:
- Hypotension, bradycardia and sinus arrest [see Warnings and Precautions (5.2)]
- Transient hypertension [see Warnings and Precautions (5.3)]
Most common treatment-emergent adverse reactions, occurring in greater than 2% of patients in both Intensive Care Unit and procedural sedation studies include hypotension, bradycardia and dry mouth.
Intensive Care Unit Sedation
Adverse reaction information is derived from the continuous infusion trials of dexmedetomidine injection for sedation in the Intensive Care Unit setting in which 1007 adult patients received dexmedetomidine injection. The mean total dose was 7.4 mcg/kg (range: 0.8 to 84.1), mean dose per hour was 0.5 mcg/kg/hr (range: 0.1 to 6.0) and the mean duration of infusion of 15.9 hours (range: 0.2 to 157.2). The population was between 17 to 88 years of age, 43% ≥ 65 years of age, 77% male and 93% Caucasian. Treatment-emergent adverse reactions occurring at an incidence of > 2% are provided in Table 2. The most frequent adverse reactions were hypotension, bradycardia and dry mouth [see Warnings and Precautions (5.2)].
Table 2: Adverse Reactions with an Incidence > 2% – Adult Intensive Care Unit Sedation Population < 24 hours* - * 26 subjects in the all dexmedetomidine injection group and 10 subjects in the randomized dexmedetomidine injection group had exposure for greater than 24 hours.
Adverse Event
All Dexmedetomidine Injection
(N = 1007)
(%)Randomized
Dexmedetomidine Injection
(N = 798)
(%)
Placebo
(N = 400)
(%)
Propofol
(N = 188)
(%)
Hypotension
25%
24%
12%
13%
Hypertension
12%
13%
19%
4%
Nausea
9%
9%
9%
11%
Bradycardia
5%
5%
3%
0
Atrial Fibrillation
4%
5%
3%
7%
Pyrexia
4%
4%
4%
4%
Dry Mouth
4%
3%
1%
1%
Vomiting
3%
3%
5%
3%
Hypovolemia
3%
3%
2%
5%
Atelectasis
3%
3%
3%
6%
Pleural Effusion
2%
2%
1%
6%
Agitation
2%
2%
3%
1%
Tachycardia
2%
2%
4%
1%
Anemia
2%
2%
2%
2%
Hyperthermia
2%
2%
3%
0
Chills
2%
2%
3%
2%
Hyperglycemia
2%
2%
2%
3%
Hypoxia
2%
2%
2%
3%
Post-procedural Hemorrhage
2%
2%
3%
4%
Pulmonary Edema
1%
1%
1%
3%
Hypocalcemia
1%
1%
0
2%
Acidosis
1%
1%
1%
2%
Urine Output Decreased
1%
1%
0
2%
Sinus Tachycardia
1%
1%
1%
2%
Ventricular Tachycardia
< 1%
1%
1%
5%
Wheezing
< 1%
1%
0
2%
Edema Peripheral
< 1%
0%
1%
2%
Adverse reaction information was also derived from the placebo-controlled, continuous infusion trials of dexmedetomidine injection for sedation in the surgical intensive care unit setting in which 387 adult patients received dexmedetomidine injection for less than 24 hours. The most frequently observed treatment-emergent adverse events included hypotension, hypertension, nausea, bradycardia, fever, vomiting, hypoxia, tachycardia and anemia (see Table 3).
Table 3: Treatment-Emergent Adverse Events Occurring in > 1% Of All Dexmedetomidine-Treated Adult Patients in the Randomized Placebo-Controlled Continuous Infusion < 24 Hours ICU Sedation Studies Adverse Event
Randomized Dexmedetomidine
(N = 387)
Placebo
(N = 379)
Hypotension
28%
13%
Hypertension
16%
18%
Nausea
11%
9%
Bradycardia
7%
3%
Fever
5%
4%
Vomiting
4%
6%
Atrial Fibrillation
4%
3%
Hypoxia
4%
4%
Tachycardia
3%
5%
Hemorrhage
3%
4%
Anemia
3%
2%
Dry Mouth
3%
1%
Rigors
2%
3%
Agitation
2%
3%
Hyperpyrexia
2%
3%
Pain
2%
2%
Hyperglycemia
2%
2%
Acidosis
2%
2%
Pleural Effusion
2%
1%
Oliguria
2%
< 1%
Thirst
2%
< 1%
In a controlled clinical trial, dexmedetomidine injection was compared to midazolam for ICU sedation exceeding 24 hours duration in adult patients. Key treatment emergent adverse events occurring in dexmedetomidine or midazolam treated patients in the randomized active comparator continuous infusion long-term intensive care unit sedation study are provided in Table 4. The number (%) of subjects who had a dose-related increase in treatment-emergent adverse events by maintenance adjusted dose rate range in the dexmedetomidine injection group is provided in Table 5.
Table 4: Key Treatment-Emergent Adverse Events Occurring in Dexmedetomidine- or Midazolam-Treated Adult Patients in the Randomized Active Comparator Continuous Infusion Long-Term Intensive Care Unit Sedation Study - * Hypotension was defined in absolute terms as Systolic blood pressure of < 80 mmHg or Diastolic blood pressure of < 50 mmHg or in relative terms as ≤ 30% lower than pre-study drug infusion value.
- † Bradycardia was defined in absolute terms as < 40 bpm or in relative terms as ≤ 30% lower than pre-study drug infusion value.
- ‡ Hypertension was defined in absolute terms as Systolic blood pressure > 180 mmHg or Diastolic blood pressure of > 100 mmHg or in relative terms as ≥ 30% higher than pre-study drug infusion value.
- § Tachycardia was defined in absolute terms as > 120 bpm or in relative terms as ≥ 30% greater than pre-study drug infusion value.
- ¶ Includes any type of hypertension.
Adverse Event
Dexmedetomidine
(N = 244)
Midazolam
(N = 122)
Hypotension*
56%
56%
Hypotension Requiring Intervention
28%
27%
Bradycardia†
42%
19%
Bradycardia Requiring Intervention
5%
1%
Systolic Hypertension‡
28%
42%
Tachycardia§
25%
44%
Tachycardia Requiring Intervention
10%
10%
Diastolic Hypertension‡
12%
15%
Hypertension
11%
15%
Hypertension Requiring Intervention¶
19%
30%
Hypokalemia
9%
13%
Pyrexia
7%
2%
Agitation
7%
6%
Hyperglycemia
7%
2%
Constipation
6%
6%
Hypoglycemia
5%
6%
Respiratory Failure
5%
3%
Renal Failure Acute
2%
1%
Acute Respiratory Distress Syndrome
2%
1%
Generalized Edema
2%
6%
Hypomagnesemia
1%
7%
The following adverse events occurred between 2 and 5% for dexmedetomidine injection and Midazolam, respectively: renal failure acute (2.5%, 0.8%), acute respiratory distress syndrome (2.5%, 0.8%), and respiratory failure (4.5%, 3.3%).
Table 5: Number (%) of Adult Subjects Who Had a Dose-Related Increase in Treatment Emergent Adverse Events by Maintenance Adjusted Dose Rate Range in the Dexmedetomidine Injection Group - * Average maintenance dose over the entire study drug administration
Dexmedetomidine Injection mcg/kg/hr
Adverse Event
≤ 0.7*
(N = 95)
> 0.7 to ≤ 1.1*
(N = 78)
> 1.1*
(N = 71)
Constipation
6%
5%
14%
Agitation
5%
8%
14%
Anxiety
5%
5%
9%
Edema Peripheral
3%
5%
7%
Atrial Fibrillation
2%
4%
9%
Respiratory Failure
2%
6%
10%
Acute Respiratory Distress Syndrome
1%
3%
9%
Procedural Sedation
Adverse reaction information is derived from the two trials for procedural sedation [see Clinical Studies (14.2)] in which 318 adult patients received dexmedetomidine injection. The mean total dose was 1.6 mcg/kg (range: 0.5 to 6.7), mean dose per hour was 1.3 mcg/kg/hr (range: 0.3 to 6.1) and the mean duration of infusion of 1.5 hours (range: 0.1 to 6.2). The population was between 18 to 93 years of age, ASA I-IV, 30% ≥ 65 years of age, 52% male and 61% Caucasian.
Treatment-emergent adverse reactions occurring at an incidence of > 2% are provided in Table 6. The most frequent adverse reactions were hypotension, bradycardia, and dry mouth [see Warnings and Precautions (5.2)]. Pre-specified criteria for the vital signs to be reported as adverse reactions are footnoted below the table. The decrease in respiratory rate and hypoxia was similar between dexmedetomidine injection and comparator groups in both studies.
Table 6: Adverse Reactions With an Incidence > 2% – Procedural Sedation Population - * Hypotension was defined in absolute and relative terms as Systolic blood pressure of < 80 mmHg or ≤ 30% lower than pre-study drug infusion value, or Diastolic blood pressure of < 50 mmHg.
- † Respiratory depression was defined in absolute and relative terms as respiratory rate (RR) < 8 beats per minute or > 25% decrease from baseline.
- ‡ Bradycardia was defined in absolute and relative terms as < 40 beats per minute or ≤ 30% lower than pre-study drug infusion value.
- § Hypertension was defined in absolute and relative terms as Systolic blood pressure > 180 mmHg or ≥ 30% higher than pre-study drug infusion value or Diastolic blood pressure of > 100 mmHg.
- ¶ Tachycardia was defined in absolute and relative terms as > 120 beats per minute or ≥ 30% greater than pre-study drug infusion value.
- # Hypoxia was defined in absolute and relative terms as SpO2 < 90% or 10% decrease from baseline.
Adverse Event
Dexmedetomidine Injection
N = 318
(%)Placebo
N = 113
(%)Hypotension*
54%
30%
Respiratory Depression†
37%
32%
Bradycardia‡
14%
4%
Hypertension§
13%
24%
Tachycardia¶
5%
17%
Nausea
3%
2%
Dry Mouth
3%
1%
Hypoxia#
2%
3%
Bradypnea
2%
4%
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of dexmedetomidine injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypotension and bradycardia were the most common adverse reactions associated with the use of dexmedetomidine injection during post approval use of the drug.
Table 7: Adverse Reactions Experienced During Post-Approval Use of Dexmedetomidine Injection System Organ Class
Preferred Term
Blood and Lymphatic System Disorders
Anemia
Cardiac Disorders
Arrhythmia, atrial fibrillation, atrioventricular block, bradycardia, cardiac arrest, cardiac disorder, extrasystoles, myocardial infarction, supraventricular tachycardia, tachycardia, ventricular arrhythmia, ventricular tachycardia
Eye Disorders
Photopsia, visual impairment
Gastrointestinal Disorders
Abdominal pain, diarrhea, nausea, vomiting
General Disorders and Administration Site Conditions
Chills, hyperpyrexia, pain, pyrexia, thirst
Hepatobiliary Disorders
Hepatic function abnormal, hyperbilirubinemia
Investigations
Alanine aminotransferase increased, aspartate aminotransferase increased, blood alkaline phosphatase increased, blood urea increased, electrocardiogram T wave inversion, gammaglutamyltransferase increased, electrocardiogram QT prolonged
Metabolism and Nutrition Disorders
Acidosis, hyperkalemia, hypoglycemia, hypovolemia, hypernatremia
Nervous System Disorders
Convulsion, dizziness, headache, neuralgia, neuritis, speech disorder
Psychiatric Disorders
Agitation, confusional state, delirium, hallucination, illusion
Renal and Urinary Disorders
Oliguria, polyuria
Respiratory, Thoracic and Mediastinal Disorders
Apnea, bronchospasm, dyspnea, hypercapnia, hypoventilation, hypoxia, pulmonary congestion, respiratory acidosis
Skin and Subcutaneous Tissue Disorders
Hyperhidrosis
Surgical and Medical Procedures
Light anesthesia
Vascular Disorders
Blood pressure fluctuation, hemorrhage, hypertension, hypotension
-
7 DRUG INTERACTIONS
7.1 Anesthetics, Sedatives, Hypnotics, Opioids
Co-administration of dexmedetomidine injection with anesthetics, sedatives, hypnotics, and opioids is likely to lead to an enhancement of effects. Specific studies have confirmed these effects with sevoflurane, isoflurane, propofol, alfentanil, and midazolam. No pharmacokinetic interactions between dexmedetomidine injection and isoflurane, propofol, alfentanil and midazolam have been demonstrated. However, due to possible pharmacodynamic interactions, when co-administered with dexmedetomidine injection, a reduction in dosage of dexmedetomidine injection or the concomitant anesthetic, sedative, hypnotic or opioid may be required.
7.2 Neuromuscular Blockers
In one study of 10 healthy adult volunteers, administration of dexmedetomidine injection for 45 minutes at a plasma concentration of one ng/mL resulted in no clinically meaningful increases in the magnitude of neuromuscular blockade associated with rocuronium administration.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C
There are no adequate and well-controlled studies of dexmedetomidine injection use in pregnant women. In an in vitro human placenta study, placental transfer of dexmedetomidine occurred. In a study in the pregnant rat, placental transfer of dexmedetomidine was observed when radiolabeled dexmedetomidine was administered subcutaneously. Thus, fetal exposure should be expected in humans, and dexmedetomidine injection should be used during pregnancy only if the potential benefits justify the potential risk to the fetus.
Teratogenic effects were not observed in rats following subcutaneous administration of dexmedetomidine during the period of fetal organogenesis (from gestation day 5 to 16) with doses up to 200 mcg/kg (representing a dose approximately equal to the maximum recommended human intravenous dose based on body surface area) or in rabbits following intravenous administration of dexmedetomidine during the period of fetal organogenesis (from gestation day 6 to 18) with doses up to 96 mcg/kg (representing approximately half the human exposure at the maximum recommended dose based on plasma area under the time-curve comparison). However, fetal toxicity, as evidenced by increased post-implantation losses and reduced live pups, was observed in rats at a subcutaneous dose of 200 mcg/kg. The no-effect dose in rats was 20 mcg/kg (representing a dose less than the maximum recommended human intravenous dose based on a body surface area comparison). In another reproductive toxicity study when dexmedetomidine was administered subcutaneously to pregnant rats at 8 and 32 mcg/kg (representing a dose less than the maximum recommended human intravenous dose based on a body surface area comparison) from gestation day 16 through weaning, lower offspring weights were observed. Additionally, when offspring of the 32 mcg/kg group were allowed to mate, elevated fetal and embryocidal toxicity and delayed motor development was observed in second generation offspring.
8.2 Labor and Delivery
The safety of dexmedetomidine injection during labor and delivery has not been studied.
8.3 Nursing Mothers
It is not known whether dexmedetomidine hydrochloride is excreted in human milk. Radio-labeled dexmedetomidine administered subcutaneously to lactating female rats was excreted in milk. Because many drugs are excreted in human milk, caution should be exercised when dexmedetomidine injection is administered to a nursing woman.
8.4 Pediatric Use
Safety and efficacy have not been established for Procedural or ICU Sedation in pediatric patients. One assessor-blinded trial in pediatric patients and two open label studies in neonates were conducted to assess efficacy for ICU sedation. These studies did not meet their primary efficacy endpoints and the safety data submitted were insufficient to fully characterize the safety profile of dexmedetomidine injection for this patient population. The use of dexmedetomidine for procedural sedation in pediatric patients has not been evaluated.
8.5 Geriatric Use
Intensive Care Unit Sedation
A total of 729 patients in the clinical studies were 65 years of age and over. A total of 200 patients were 75 years of age and over. In patients greater than 65 years of age, a higher incidence of bradycardia and hypotension was observed following administration of dexmedetomidine injection [see Warnings and Precautions (5.2)]. Therefore a dose reduction may be considered in patients over 65 years of age [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
Procedural Sedation
A total of 131 patients in the clinical studies were 65 years of age and over. A total of 47 patients were 75 years of age and over. Hypotension occurred in a higher incidence in dexmedetomidine injection-treated patients 65 years or older (72%) and 75 years or older (74%) as compared to patients < 65 years (47%). A reduced loading dose of 0.5 mcg/kg given over 10 minutes is recommended and a reduction in the maintenance infusion should be considered for patients greater than 65 years of age.
8.6 Hepatic Impairment
Since dexmedetomidine clearance decreases with increasing severity of hepatic impairment, dose reduction should be considered in patients with impaired hepatic function [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
-
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
Dexmedetomidine injection (dexmedetomidine hydrochloride) is not a controlled substance.
9.3 Dependence
The dependence potential of dexmedetomidine injection has not been studied in humans. However, since studies in rodents and primates have demonstrated that dexmedetomidine injection exhibits pharmacologic actions similar to those of clonidine, it is possible that dexmedetomidine injection may produce a clonidine-like withdrawal syndrome upon abrupt discontinuation [see Warnings and Precautions (5.5)].
-
10 OVERDOSAGE
The tolerability of dexmedetomidine injection was studied in one study in which healthy adult subjects were administered doses at and above the recommended dose of 0.2 to 0.7 mcg/kg/hr. The maximum blood concentration achieved in this study was approximately 13 times the upper boundary of the therapeutic range. The most notable effects observed in two subjects who achieved the highest doses were first degree atrioventricular block and second degree heart block. No hemodynamic compromise was noted with the atrioventricular block and the heart block resolved spontaneously within one minute.
Five adult patients received an overdose of dexmedetomidine injection in the intensive care unit sedation studies. Two of these patients had no symptoms reported; one patient received a 2 mcg/kg loading dose over 10 minutes (twice the recommended loading dose) and one patient received a maintenance infusion of 0.8 mcg/kg/hr. Two other patients who received a 2 mcg/kg loading dose over 10 minutes, experienced bradycardia and/or hypotension. One patient who received a loading bolus dose of undiluted dexmedetomidine injection (19.4 mcg/kg), had cardiac arrest from which he was successfully resuscitated.
-
11 DESCRIPTION
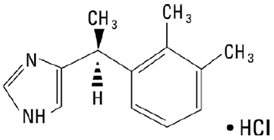
Dexmedetomidine injection, USP is a sterile, nonpyrogenic solution suitable for intravenous infusion following dilution. Dexmedetomidine hydrochloride is the S-enantiomer of medetomidine and is chemically described as (+)-4-(S)-[1-(2,3-dimethylphenyl)ethyl]-1H-imidazole monohydrochloride. Dexmedetomidine hydrochloride has a molecular weight of 236.7 and the empirical formula is C13H16N2 HCl and the structural formula is:
Dexmedetomidine hydrochloride, USP is a white or almost white powder that is freely soluble in water and has a pKa of 7.1. Its partition coefficient in-octanol: water at pH 7.4 is 2.89.
Dexmedetomidine injection is supplied as a clear, colorless, isotonic solution with a pH of 4.5 to 7.0. Each mL contains 118 mcg of dexmedetomidine hydrochloride equivalent to 100 mcg (0.1 mg) of dexmedetomidine and 9 mg of sodium chloride in water and is to be used after dilution. The solution is preservative-free and contains no additives or chemical stabilizers.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Dexmedetomidine injection is a relatively selective alpha2-adrenergic agonist with sedative properties. Alpha2 selectivity is observed in animals following slow intravenous infusion of low and medium doses (10-300 mcg/kg). Both alpha1 and alpha2 activity is observed following slow intravenous infusion of high doses (≥ 1000 mcg/kg) or with rapid intravenous administration.
12.2 Pharmacodynamics
In a study in healthy volunteers (N = 10), respiratory rate and oxygen saturation remained within normal limits and there was no evidence of respiratory depression when dexmedetomidine injection was administered by intravenous infusion at doses within the recommended dose range (0.2–0.7 mcg/kg/hr).
12.3 Pharmacokinetics
Following intravenous administration, dexmedetomidine exhibits the following pharmacokinetic parameters: a rapid distribution phase with a distribution half-life (t1/2) of approximately 6 minutes; a terminal elimination half-life (t1/2) of approximately 2 hours; and steady-state volume of distribution (Vss) of approximately 118 liters. Clearance is estimated to be approximately 39 L/h. The mean body weight associated with this clearance estimate was 72 kg.
Dexmedetomidine exhibits linear pharmacokinetics in the dosage range of 0.2 to 0.7 mcg/kg/hr when administered by intravenous infusion for up to 24 hours. Table 8 shows the main pharmacokinetic parameters when dexmedetomidine injection was infused (after appropriate loading doses) at maintenance infusion rates of 0.17 mcg/kg/hr (target plasma concentration of 0.3 ng/mL) for 12 and 24 hours, 0.33 mcg/kg/hr (target plasma concentration of 0.6 ng/mL) for 24 hours, and 0.70 mcg/kg/hr (target plasma concentration of 1.25 ng/mL) for 24 hours.
Table 8: Mean ± SD Pharmacokinetic Parameters - * Presented as harmonic mean and pseudo standard deviation.
- † Mean Css = Average steady-state concentration of dexmedetomidine. The mean Css was calculated based on post-dose sampling from 2.5 to 9 hours samples for 12 hour infusion and post-dose sampling from 2.5 to 18 hours for 24 hour infusions.
Parameter
Loading Infusion (min)/Total Infusion Duration (hrs)
10 min/12 hrs
10 min/24 hrs
10 min/24 hrs
35 min/24 hrs
Dexmedetomidine Target Plasma Concentration (ng/mL)
and Dose (mcg/kg/hr)0.3/0.17
0.3/0.17
0.6/0.33
1.25/0.70
t1/2*, hour
1.78 ± 0.30
2.22 ± 0.59
2.23 ± 0.21
2.50 ± 0.61
CL, liter/hour
46.3 ± 8.3
43.1 ± 6.5
35.3 ± 6.8
36.5 ± 7.5
Vss, liter
88.7 ± 22.9
102.4 ± 20.3
93.6 ± 17.0
99.6 ± 17.8
Avg Css†, ng/mL
0.27 ± 0.05
0.27 ± 0.05
0.67 ± 0.10
1.37 ± 0.20
The loading doses for each of the above indicated groups were 0.5, 0.5, 1 and 2.2 mcg/kg, respectively.
Dexmedetomidine pharmacokinetic parameters after dexmedetomidine injection maintenance doses of 0.2 to 1.4 mcg/kg/hr for > 24 hours were similar to the PK parameters after dexmedetomidine injection maintenance dosing for < 24 hours in other studies. The values for clearance (CL), volume of distribution (V), and t1/2 were 39.4 L/hr, 152 L, and 2.67 hours, respectively.
Distribution
The steady-state volume of distribution (Vss) of dexmedetomidine was approximately 118 liters. Dexmedetomidine protein binding was assessed in the plasma of normal healthy male and female subjects. The average protein binding was 94% and was constant across the different plasma concentrations tested. Protein binding was similar in males and females. The fraction of dexmedetomidine that was bound to plasma proteins was significantly decreased in subjects with hepatic impairment compared to healthy subjects.
The potential for protein binding displacement of dexmedetomidine by fentanyl, ketorolac, theophylline, digoxin and lidocaine was explored in vitro, and negligible changes in the plasma protein binding of dexmedetomidine were observed. The potential for protein binding displacement of phenytoin, warfarin, ibuprofen, propranolol, theophylline and digoxin by dexmedetomidine was explored in vitro and none of these compounds appeared to be significantly displaced by dexmedetomidine.
Metabolism
Dexmedetomidine undergoes almost complete biotransformation with very little unchanged dexmedetomidine excreted in urine and feces. Biotransformation involves both direct glucuronidation as well as cytochrome P450 mediated metabolism. The major metabolic pathways of dexmedetomidine are: direct N-glucuronidation to inactive metabolites; aliphatic hydroxylation (mediated primarily by CYP2A6 with a minor role of CYP1A2, CYP2E1, CYP2D6 and CYP2C19) of dexmedetomidine to generate 3-hydroxy-dexmedetomidine, the glucuronide of 3-hydroxy-dexmedetomidine, and 3-carboxy-dexmedetomidine; and N-methylation of dexmedetomidine to generate 3-hydroxy N-methyl-dexmedetomidine, 3-carboxy N-methyl-dexmedetomidine, and dexmedetomidine-N-methyl O-glucuronide.
Elimination
The terminal elimination half-life (t1/2) of dexmedetomidine is approximately 2 hours and clearance is estimated to be approximately 39 L/h. A mass balance study demonstrated that after nine days an average of 95% of the radioactivity, following intravenous administration of radiolabeled dexmedetomidine, was recovered in the urine and 4% in the feces. No unchanged dexmedetomidine was detected in the urine. Approximately 85% of the radioactivity recovered in the urine was excreted within 24 hours after the infusion. Fractionation of the radioactivity excreted in urine demonstrated that products of N-glucuronidation accounted for approximately 34% of the cumulative urinary excretion. In addition, aliphatic hydroxylation of parent drug to form 3-hydroxy-dexmedetomidine, the glucuronide of 3-hydroxy-dexmedetomidine, and 3-carboxylic acid-dexmedetomidine together represented approximately 14% of the dose in urine. N-methylation of dexmedetomidine to form 3-hydroxy N-methyl dexmedetomidine, 3-carboxy N-methyl dexmedetomidine, and N-methyl O-glucuronide dexmedetomidine accounted for approximately 18% of the dose in urine. The N-Methyl metabolite itself was a minor circulating component and was undetected in urine. Approximately 28% of the urinary metabolites have not been identified.
Gender
There was no observed difference in dexmedetomidine injection pharmacokinetics due to gender.
Geriatrics
The pharmacokinetic profile of dexmedetomidine injection was not altered by age. There were no differences in the pharmacokinetics of dexmedetomidine injection in young (18–40 years), middle age (41–65 years), and elderly (> 65 years) subjects.
Hepatic Impairment
In subjects with varying degrees of hepatic impairment (Child-Pugh Class A, B, or C), clearance values for dexmedetomidine were lower than in healthy subjects. The mean clearance values for patients with mild, moderate, and severe hepatic impairment were 74%, 64% and 53% of those observed in the normal healthy subjects, respectively. Mean clearances for free drug were 59%, 51% and 32% of those observed in the normal healthy subjects, respectively.
Although dexmedetomidine injection is dosed to effect, it may be necessary to consider dose reduction in subjects with hepatic impairment [see Dosage and Administration (2.2), Warnings and Precautions (5.7)].
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal carcinogenicity studies have not been performed with dexmedetomidine.
Dexmedetomidine was not mutagenic in vitro, in either the bacterial reverse mutation assay (E. coli and Salmonella typhimurium) or the mammalian cell forward mutation assay (mouse lymphoma). Dexmedetomidine was clastogenic in the in vitro human lymphocyte chromosome aberration test with, but not without, rat S9 metabolic activation. In contrast, dexmedetomidine was not clastogenic in the in vitro human lymphocyte chromosome aberration test with or without human S9 metabolic activation. Although dexmedetomidine was clastogenic in an in vivo mouse micronucleus test in NMRI mice, there was no evidence of clastogenicity in CD-1 mice.
Fertility in male or female rats was not affected after daily subcutaneous injections of dexmedetomidine at doses up to 54 mcg/kg (less than the maximum recommended human intravenous dose on a mcg/m2 basis) administered from 10 weeks prior to mating in males, and 3 weeks prior to mating and during mating in females.
13.2 Animal Pharmacology and/or Toxicology
There were no differences in the adrenocorticotropic hormone (ACTH)-stimulated cortisol response in dogs following a single dose of dexmedetomidine compared to saline control. However, after continuous subcutaneous infusions of dexmedetomidine at 3 mcg/kg/hr and 10 mcg/kg/hr for one week in dogs (exposures estimated to be within the clinical range), the ACTH-stimulated cortisol response was diminished by approximately 27% and 40%, respectively, compared to saline-treated control animals indicating a dose-dependent adrenal suppression.
-
14 CLINICAL STUDIES
The safety and efficacy of dexmedetomidine injection has been evaluated in four randomized, double-blind, placebo-controlled multicenter clinical trials in 1185 adult patients.
14.1 Intensive Care Unit Sedation
Two randomized, double-blind, parallel-group, placebo-controlled multicenter clinical trials included 754 adult patients being treated in a surgical intensive care unit. All patients were initially intubated and received mechanical ventilation. These trials evaluated the sedative properties of dexmedetomidine injection by comparing the amount of rescue medication (midazolam in one trial and propofol in the second) required to achieve a specified level of sedation (using the standardized Ramsay Sedation Scale) between dexmedetomidine injection and placebo from onset of treatment to extubation or to a total treatment duration of 24 hours. The Ramsay Level of Sedation Scale is displayed in Table 9.
Table 9: Ramsay Level of Sedation Scale Clinical Score
Level of Sedation Achieved
6
Asleep, no response
5
Asleep, sluggish response to light glabellar tap or loud auditory stimulus
4
Asleep, but with brisk response to light glabellar tap or loud auditory stimulus
3
Patient responds to commands
2
Patient cooperative, oriented, and tranquil
1
Patient anxious, agitated, or restless
In the first study, 175 adult patients were randomized to receive placebo and 178 to receive dexmedetomidine injection by intravenous infusion at a dose of 0.4 mcg/kg/hr (with allowed adjustment between 0.2 and 0.7 mcg/kg/hr) following an initial loading infusion of one mcg/kg intravenous over 10 minutes. The study drug infusion rate was adjusted to maintain a Ramsay sedation of ≥ 3. Patients were allowed to receive “rescue” midazolam as needed to augment the study drug infusion. In addition, morphine sulfate was administered for pain as needed. The primary outcome measure for this study was the total amount of rescue medication (midazolam) needed to maintain sedation as specified while intubated. Patients randomized to placebo received significantly more midazolam than patients randomized to dexmedetomidine injection (see Table 10).
A second prospective primary analysis assessed the sedative effects of dexmedetomidine injection by comparing the percentage of patients who achieved a Ramsay sedation score of ≥ 3 during intubation without the use of additional rescue medication. A significantly greater percentage of patients in the dexmedetomidine injection group maintained Ramsay score of ≥ 3 without receiving any midazolam rescue compared to the placebo group (see Table 10).
Table 10: Midazolam Use as Rescue Medication During Intubation (ITT) Study One ITT (intent-to-treat) population includes all randomized patients. - * ANOVA model with treatment center.
- † Chi-square.
Placebo
(N = 175)
Dexmedetomidine Injection
(N = 178)
p-value
Mean Total Dose (mg) of Midazolam
Standard deviation
19 mg
53 mg
5 mg
19 mg
0.0011*
Categorized Midazolam Use
0 mg
43 (25%)
108 (61%)
< 0.001†
0–4 mg
34 (19%)
36 (20%)
> 4 mg
98 (56%)
34 (19%)
A prospective secondary analysis assessed the dose of morphine sulfate administered to patients in the dexmedetomidine injection and placebo groups. On average, dexmedetomidine injection-treated patients received less morphine sulfate for pain than placebo-treated patients (0.47 versus 0.83 mg/h). In addition, 44% (79 of 178 patients) of dexmedetomidine injection patients received no morphine sulfate for pain versus 19% (33 of 175 patients) in the placebo group.
In a second study, 198 adult patients were randomized to receive placebo and 203 to receive dexmedetomidine injection by intravenous infusion at a dose of 0.4 mcg/kg/hr (with allowed adjustment between 0.2 and 0.7 mcg/kg/hr) following an initial loading infusion of one mcg/kg intravenous over 10 minutes. The study drug infusion was adjusted to maintain a Ramsay score of ≥ 3. Patients were allowed to receive “rescue” propofol as needed to augment the study drug infusion. In addition, morphine sulfate was administered as needed for pain. The primary outcome measure for this study was the total amount of rescue medication (propofol) needed to maintain sedation as specified while intubated.
Patients randomized to placebo received significantly more propofol than patients randomized to dexmedetomidine injection (see Table 11).
A significantly greater percentage of patients in the dexmedetomidine injection group compared to the placebo group maintained a Ramsay score of ≥ 3 without receiving any propofol rescue (see Table 11).
Table 11: Propofol Use as Rescue Medication During Intubation (ITT) Study Two - * ANOVA model with treatment center.
- † Chi-square
Placebo
(N = 198)
Dexmedetomidine Injection
(N = 203)
p-value
Mean Total Dose (mg) of Propofol
Standard deviation
513 mg
782 mg
72 mg
249 mg
< 0.0001*
Categorized Propofol Use
0 mg
47 (24%)
122 (60%)
< 0.001†
0–50 mg
30 (15%)
43 (21%)
> 50 mg
121 (61%)
38 (19%)
A prospective secondary analysis assessed the dose of morphine sulfate administered to patients in the dexmedetomidine injection and placebo groups. On average, dexmedetomidine injection-treated patients received less morphine sulfate for pain than placebo-treated patients (0.43 versus 0.89 mg/h). In addition, 41% (83 of 203 patients) of dexmedetomidine injection patients received no morphine sulfate for pain versus 15% (30 of 198 patients) in the placebo group.
In a controlled clinical trial, dexmedetomidine injection was compared to midazolam for ICU sedation exceeding 24 hours duration. Dexmedetomidine injection was not shown to be superior to midazolam for the primary efficacy endpoint, the percent of time patients were adequately sedated (81% versus 81%). In addition, administration of dexmedetomidine injection for longer than 24 hours was associated with tolerance, tachyphylaxis, and a dose-related increase in adverse events [see Adverse Reactions (6.1)].
14.2 Procedural Sedation
The safety and efficacy of dexmedetomidine injection for sedation of non-intubated patients prior to and/or during surgical and other procedures was evaluated in two randomized, double-blind, placebo-controlled multicenter clinical trials. Study 1 evaluated the sedative properties of dexmedetomidine injection in patients having a variety of elective surgeries/procedures performed under monitored anesthesia care. Study 2 evaluated dexmedetomidine injection in patients undergoing awake fiberoptic intubation prior to a surgical or diagnostic procedure.
In Study 1, the sedative properties of dexmedetomidine injection were evaluated by comparing the percent of patients not requiring rescue midazolam to achieve a specified level of sedation using the standardized Observer’s Assessment of Alertness/Sedation Scale (see Table 12).
Table 12: Observer’s Assessment of Alertness/Sedation Assessment Categories
Responsiveness
Speech
Facial Expression
Eyes
Composite Score
Responds readily to name spoken in normal tone
Normal
Normal
Clear, no ptosis
5 (alert)
Lethargic response to name spoken in normal tone
Mild slowing or thickening
Mild relaxation
Glazed or mild ptosis (less than half the eye)
4
Responds only after name is called loudly and/or repeatedly
Slurring or prominent slowing
Marked relaxation (slack jaw)
Glazed and marked ptosis (half the eye or more)
3
Responds only after mild prodding or shaking
Few recognizable words
–
–
2
Does not respond to mild prodding or shaking
–
–
–
1 (deep sleep)
Patients were randomized to receive a loading infusion of either dexmedetomidine injection 1 mcg/kg, dexmedetomidine injection 0.5 mcg/kg, or placebo (normal saline) given over 10 minutes and followed by a maintenance infusion started at 0.6 mcg/kg/hr. The maintenance infusion of study drug could be titrated from 0.2 mcg/kg/hr to 1 mcg/kg/hr to achieve the targeted sedation score (Observer’s Assessment of Alertness/Sedation Scale ≤ 4). Patients were allowed to receive rescue midazolam as needed to achieve and/or maintain an Observer’s Assessment of Alertness/Sedation Scale ≤ 4. After achieving the desired level of sedation, a local or regional anesthetic block was performed. Demographic characteristics were similar between the dexmedetomidine injection and comparator groups. Efficacy results showed that dexmedetomidine injection was more effective than the comparator group when used to sedate non-intubated patients requiring monitored anesthesia care during surgical and other procedures (see Table 13).
In Study 2, the sedative properties of dexmedetomidine injection were evaluated by comparing the percent of patients requiring rescue midazolam to achieve or maintain a specified level of sedation using the Ramsay Sedation Scale score ≥ 2 (see Table 9). Patients were randomized to receive a loading infusion of dexmedetomidine injection 1 mcg/kg or placebo (normal saline) given over 10 minutes and followed by a fixed maintenance infusion of 0.7 mcg/kg/hr. After achieving the desired level of sedation, topicalization of the airway occurred. Patients were allowed to receive rescue midazolam as needed to achieve and/or maintain a Ramsay Sedation Scale ≥ 2. Demographic characteristics were similar between the dexmedetomidine injection and comparator groups. For efficacy results see Table 13.
Table 13: Key Efficacy Results of Procedural Sedation Studies - * Based on ITT population defined as all randomized and treated patients.
- † Normal approximation to the binomial with continuity correction.
Study
Loading
Infusion
Treatment
ArmNumber of
Patients
Enrolled*% Not
Requiring
Midazolam
RescueConfidence†
Interval on the
Difference vs.
PlaceboMean (SD)
Total Dose
(mg) of Rescue
Midazolam
RequiredConfidence†
Intervals of the
Mean Rescue
DoseStudy 1
Dexmedetomidine
0.5 mcg/kg134
40
37 (27, 48)
1.4 (1.7)
-2.7 (-3.4, -2.0)
Dexmedetomidine
1 mcg/kg129
54
51 (40, 62)
0.9 (1.5)
-3.1 (-3.8, -2.5)
placebo
63
3
—
4.1 (3.0)
—
Study 2
Dexmedetomidine
1 mcg/kg55
53
39 (20, 57)
1.1 (1.5)
-1.8 (-2.7, -0.9)
placebo
50
14
—
2.9 (3.0)
—
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Dexmedetomidine Injection USP, 200 mcg/2 mL (100 mcg/mL), for intravenous infusion, is available in:
NDC: 67457-251-02
2 mL in a 3 mL single-dose vial, cartons of 25The strength is based on the dexmedetomidine base. Vials are intended for single-dose only.
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Discard unused portion.
-
17 PATIENT COUNSELING INFORMATION
Dexmedetomidine injection is indicated for short-term intravenous sedation. Dosage must be individualized and titrated to the desired clinical effect. Blood pressure, heart rate and oxygen levels will be monitored both continuously during the infusion of dexmedetomidine injection and as clinically appropriate after discontinuation.
- When dexmedetomidine injection is infused for more than 6 hours, patients should be informed to report nervousness, agitation, and headaches that may occur for up to 48 hours.
- Additionally, patients should be informed to report symptoms that may occur within 48 hours after the administration of dexmedetomidine injection such as: weakness, confusion, excessive sweating, weight loss, abdominal pain, salt cravings, diarrhea, constipation, dizziness or light-headedness.
Sterile, Non-pyrogenic
Preservative Free
Manufactured for:
Mylan Institutional LLC
Rockford, IL 61103 U.S.A.Manufactured by:
Mylan Institutional
Galway, IrelandRevised: 10/2019
0940L101
MI:DEXDIJ:R9 -
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 200 mcg/2 mL
NDC: 67457-251-02
Dexmedetomidine
Injection, USP
200 mcg/2 mL
(100 mcg/mL) DexmedetomidineFor Intravenous Infusion Only
MUST BE DILUTED
Rx only 25 x 2 mL Single-Dose Vials
Sterile, Nonpyrogenic
Preservative free
Each mL contains: 118 mcg of dexmedetomidine hydrochloride equivalent to
100 mcg of dexmedetomidine; sodium chloride, 9 mg; water for injection, q.s. The
solution is preservative free and contains no additives or chemical stabilizers. pH is
4.5 to 7.0.Usual Dosage: See accompanying prescribing information.
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Must be diluted with 0.9% Sodium Chloride Injection USP for intravenous use.
Discard unused portion.
MI:251:25C:R7
Manufactured for:
Mylan Institutional LLC
Rockford, IL 61103 U.S.A.Manufactured by:
Mylan Institutional
Galway, IrelandMylan.com
-
INGREDIENTS AND APPEARANCE
DEXMEDETOMIDINE
dexmedetomidine injection, solution, concentrateProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 67457-251 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXMEDETOMIDINE HYDROCHLORIDE (UNII: 1018WH7F9I) (DEXMEDETOMIDINE - UNII:67VB76HONO) DEXMEDETOMIDINE 100 ug in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 67457-251-02 25 in 1 CARTON 08/18/2014 1 NDC: 67457-251-00 2 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA202881 08/18/2014 Labeler - Mylan Institutional LLC (790384502)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.