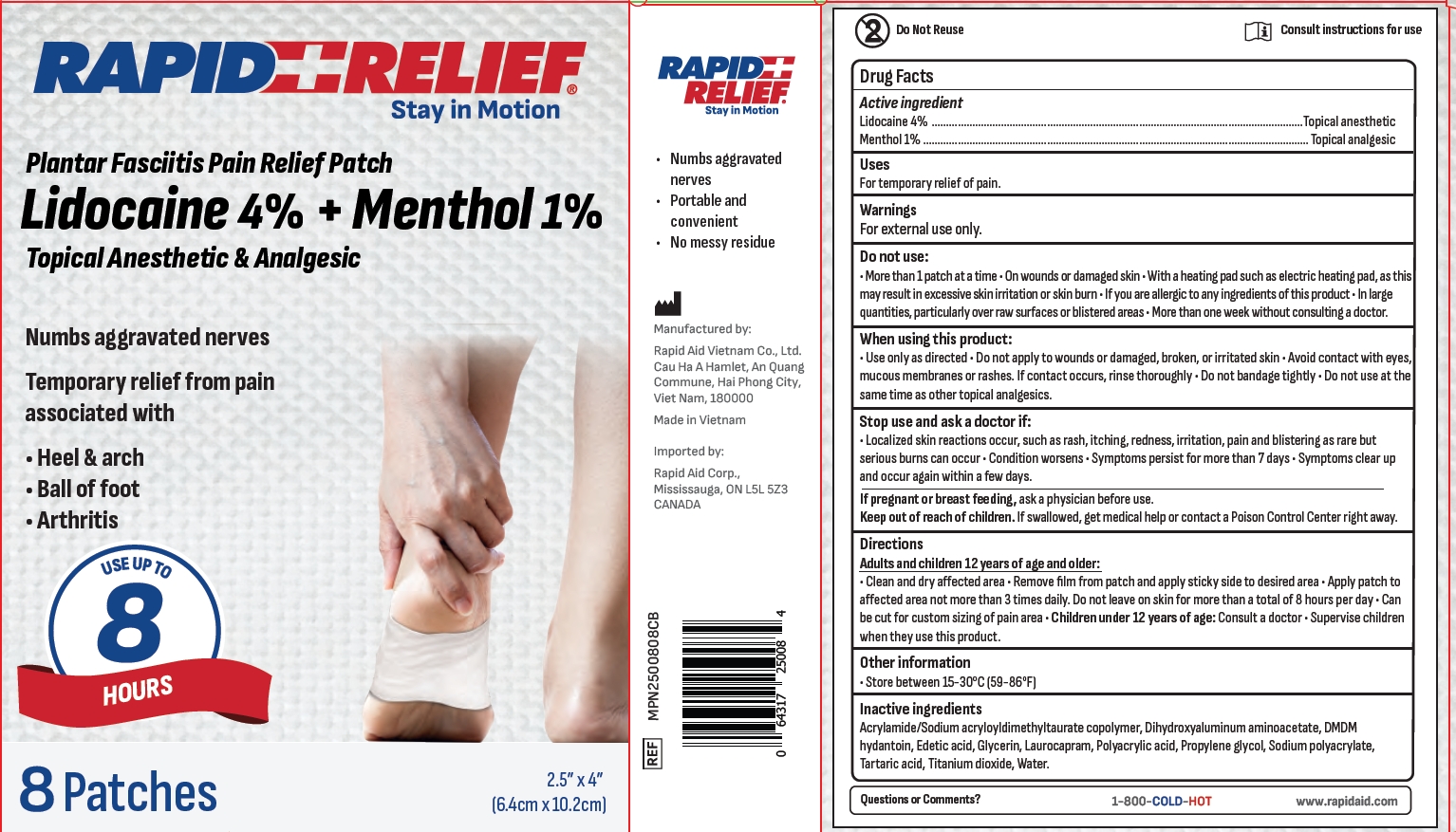
Rapid Relief Plantar Fasciitis Pain Relief Patch by RAPID AID VIET NAM CO., LTD
Rapid Relief Plantar Fasciitis Pain Relief Patch by
Drug Labeling and Warnings
Rapid Relief Plantar Fasciitis Pain Relief Patch by is a Otc medication manufactured, distributed, or labeled by RAPID AID VIET NAM CO., LTD. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
RAPID RELIEF PLANTAR FASCIITIS PAIN RELIEF PATCH- lidocaine menthol patch
RAPID AID VIET NAM CO., LTD
----------
Do not use
More than 1 patch at a time . On wounds or damaged skin . With a heating pad such as electric heating pad, as thismay result in excessive skin irritation or skin burn · lfyou are allergic to anyingredients of this product . ln largequantities, particularly over raw surfaces or blistered areas . More than one week without consulting a doctor.
When using this product:
Use only as directed . Do not apply to wounds or damaged, broken, or irritated skin · Avoid contact with eyesmucous membranes or rashes. lf contact occurs, rinse thoroughly · Do not bandage tightly. Do not use at thesame time as other topical analgesics.
Stop use and ask a doctor if:
. Localized skin reactions occur, such as rash, itching, redness, irritation, pain and blistering as rare butserious burns can occur . Condition worsens · Symptoms persist for more than 7 days · Symptoms clear upand occur again within a few days.
If pregnant or breast feeding, ask a physician before use.
KEEP OUT OF REACH OF CHILDREN
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
Adults and children 12 years of age and older:Clean and dry affected area · Remove flm from patch and apply sticky side to desired area · Apply patch toaffected area not more than 3 times daily. Do not leave on skin for more than a total of8 hours per day. Canbe cut for custom sizing of pain area . Children under 12 years of age: Consult a doctor . Supervise childrenwhen they use this product.
| RAPID RELIEF PLANTAR FASCIITIS PAIN RELIEF PATCH
lidocaine menthol patch |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - RAPID AID VIET NAM CO., LTD (673067008) |
| Registrant - RAPID AID VIET NAM CO., LTD (673067008) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| RAPID AID VIET NAM CO., LTD | 673067008 | manufacture(83569-007) | |