FIRAZYR- icatibant acetate injection, solution
Firazyr by
Drug Labeling and Warnings
Firazyr by is a Prescription medication manufactured, distributed, or labeled by Shire US Manufacturing Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use FIRAZYR® (icatibant) safely and effectively. See full prescribing information for FIRAZYR.
FIRAZYR (icatibant) Injection, for subcutaneous use
Initial U.S. Approval: 2011INDICATIONS AND USAGE
FIRAZYR is a bradykinin B2 receptor antagonist indicated for treatment of acute attacks of hereditary angioedema (HAE) in adults 18 years of age and older. (1)
DOSAGE AND ADMINISTRATION
- 30 mg injected subcutaneously in the abdominal area. (2.1)
- If response is inadequate or symptoms recur, additional injections of 30 mg may be administered at intervals of at least 6 hours. (2.1)
- Do not administer more than 3 injections in 24 hours. (2.1)
- Patients may self-administer upon recognition of an HAE attack. (2.2)
DOSAGE FORMS AND STRENGTHS
Injection: 10 mg per mL (3)
CONTRAINDICATIONS
None (4)
WARNINGS AND PRECAUTIONS
- Laryngeal attacks: Following treatment of laryngeal attacks with FIRAZYR, advise patients to seek immediate medical attention. (5.1)
ADVERSE REACTIONS
The most commonly reported adverse reactions were injection site reactions, which occurred in almost all patients (97%) in clinical trials. Other common adverse reactions occurring in greater than 1% of patients included pyrexia, transaminase increase, dizziness, and rash. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Shire Human Genetic Therapies at 1-800-828-2088 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
USE IN SPECIFIC POPULATIONS
- Elderly patients demonstrate increased systemic exposure to icatibant. Differences in efficacy and safety between elderly and younger patients have not been identified. (8.5)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 12/2015
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosing
2.2 Administration Instructions
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Laryngeal Attacks
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Immunogenicity
6.3 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 ACE Inhibitors
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Labor and Delivery
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
8.7 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
17.1 Information for Patients
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosing
The recommended dose of FIRAZYR is 30 mg administered by subcutaneous (SC) injection in the abdominal area. Additional doses may be administered at intervals of at least 6 hours if response is inadequate or if symptoms recur. No more than 3 doses may be administered in any 24 hour period.
2.2 Administration Instructions
FIRAZYR should be inspected visually for particulate matter and discoloration prior to administration. The drug solution should be clear and colorless. Do not administer if the product contains particulates or is discolored.
Attach the provided 25 gauge needle to the syringe hub and screw on securely. Do not use a different needle. Disinfect the injection site and administer FIRAZYR by subcutaneous injection over at least 30 seconds.
Patients may self-administer FIRAZYR upon recognition of symptoms of an HAE attack after training under the guidance of a healthcare professional [see Patient Counseling Information (17)].
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
- 5 WARNINGS AND PRECAUTIONS
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
The safety of icatibant was evaluated in three controlled trials that included 223 patients who received FIRAZYR 30 mg (n=113), placebo (n=75), or comparator (n=38). The mean age at study entry was 38 years (range 18 to 83 years), 64% were female, and 95% were white. The data described below represent adverse reactions observed from the two placebo-controlled trials, consisting of 77 patients who received FIRAZYR at a dose of 30 mg SC, and 75 who received placebo.
The most frequently reported adverse reactions (occurring in greater than 1% of patients and at a higher rate with FIRAZYR versus placebo) are shown in Table 1.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Table 1 Adverse reactions observed in >1% of patients with acute attacks of HAE and at a higher rate with FIRAZYR versus placebo in the placebo-controlled trials* FIRAZYR
(N =77)Placebo
(N = 75)System Organ Class
Preferred TermSubjects (%) Subjects (%) - * Events occurring within 14 days of study drug administration
- † Injection site bruising, Injection site hematoma, Injection site burning, Injection site erythema, Injection site hypoesthesia, Injection site irritation, Injection site numbness, Injection site edema, Injection site pain, Injection site pressure sensation, Injection site pruritus, Injection site swelling, Injection site urticaria, and Injection site warmth
General disorders and administration site conditions Injection site reaction † 75 (97) 25 (33) Pyrexia 3 (4) 0 Investigations Transaminase increased 3 (4) 0 Nervous system disorders Dizziness 2 (3) 1 (1) The third trial was active-controlled and was comprised of 35 patients who received FIRAZYR 30 mg and 38 patients who received the comparator. Adverse reactions for FIRAZYR were similar in nature and frequency to those reported in Table 1.
In all three controlled trials, patients were eligible for treatment of subsequent attacks in an open-label extension. Patients were treated with FIRAZYR 30 mg and could receive up to 3 doses of FIRAZYR 30 mg administered at least 6 hours apart for each attack. A total of 225 patients were treated with 1,076 doses of 30 mg FIRAZYR for 987 attacks of acute HAE. Adverse reactions similar in nature and frequency were observed to those seen in the controlled phase of the trials. Other adverse reactions reported included rash, nausea, and headache in patients exposed to FIRAZYR.
The safety of self-administration was evaluated in a separate, open-label trial in 56 patients with HAE. In this trial, the safety profile of FIRAZYR in patients who self-administered FIRAZYR was similar in nature and frequency to that of patients whose therapy was administered by healthcare professionals.
6.2 Immunogenicity
Across repeated treatment in the controlled trials, 4 patients tested positive for anti-icatibant antibodies. Three of these patients had subsequent tests which were negative. No hypersensitivity or anaphylactic reactions were reported with FIRAZYR. No association between anti-icatibant antibodies and efficacy was observed.
6.3 Postmarketing Experience
Similar adverse reactions have been observed in postmarketing use as compared to the clinical trials. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C. There are no adequate and well-controlled studies in pregnant women. Icatibant was not teratogenic in rats or rabbits; however, it caused delayed parturition, fetal death, and pre-implantation loss in rats and premature birth, abortion, fetal death, and pre-implantation loss in rabbits. FIRAZYR should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Delayed parturition and fetal death in rats occurred at 0.5 and 2-fold, respectively, the maximum recommended human dose (MRHD) (on an AUC basis at maternal doses of 1 and 3 mg/kg, respectively). Increased pre-implantation loss in rats occurred at 7-fold the MRHD (on an AUC basis at a maternal dose of 10 mg/kg). In rabbits, premature birth and abortion rates increased at a dose that was less than 1/40th the MRHD (on a mg/m2 basis at a maternal dose of 0.1 mg/kg). Studies in rabbits also indicated that pre-implantation loss and increased fetal deaths occurred at 13-fold greater than the MRHD (on an AUC basis at a maternal dose of 10 mg/kg).
8.2 Labor and Delivery
There are no human studies that have investigated the effects of FIRAZYR on preterm labor or labor at term; however, animal studies showed that icatibant causes delayed parturition and associated fetal death in rats and premature birth and abortion in rabbits. Delayed parturition occurred in rats at 0.5-fold times the MRHD (on an AUC basis at a maternal dose of 1 mg/kg).
8.3 Nursing Mothers
Because many drugs are excreted in human milk, caution should be exercised when FIRAZYR is administered to a nursing woman. Icatibant is excreted into the milk of lactating rats.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients below the age of 18 years have not been established.
Juvenile Toxicity Data
Subcutaneous daily administration of icatibant to young rats during the juvenile period of development (postnatal days 22-70) delayed the sexual maturation of male reproductive tissues (atrophy of testes and epididymides) at exposures approximating one-third or greater the MRHD on a mg/m2 basis. Impaired fertility and reproductive performance were also observed in male rats at the end of the postnatal treatment period at exposures approximating the MRHD or greater on a mg/m2 basis. No effects were observed in females at exposures approximating 3-fold the MRHD on a mg/m2 basis. The observed tissue findings in males were consistent with those seen in sexually mature rats and dogs and are attributed to antagonism of the bradykinin B2 receptor and subsequent effects on gonadotropins. The observed effects may be a consequence of daily icatibant administration. Toxicity to the testis did not occur in dogs treated twice a week for 9 months [see Carcinogenesis, Mutagenesis, Impairment of Fertility (13.1)].
8.5 Geriatric Use
Clinical studies of FIRAZYR did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Elderly patients are likely to have increased systemic exposure to FIRAZYR compared to younger (18-45 years) patients [see Clinical Pharmacology (12.3)]. Since other reported clinical experience has not identified differences in efficacy and safety between elderly and younger patients, no dose adjustment is recommended.
8.6 Hepatic Impairment
FIRAZYR was studied in patients with mild to moderate (Child Pugh scores of 5 to 8) hepatic impairment. No change in systemic exposure is noted in these patient populations. No dose adjustment is required in patients with hepatic impairment [see Clinical Pharmacology (12.3)].
8.7 Renal Impairment
Although a formal renal impairment study has not been conducted, 10 of 37 patients treated with FIRAZYR had hepatorenal syndrome with glomerular filtration rate (GFR) below 60 mL/min. FIRAZYR is cleared non-renally and hence it is not expected to show any change in systemic exposure in patients with impaired renal function. No dose adjustment is required in patients with renal impairment [see Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
In a clinical study evaluating a 90 mg dose (30 mg in each of 3 subcutaneous sites), the adverse event profile was similar to that seen with 30 mg administered in a single subcutaneous site.
In another clinical study, a dose of 3.2 mg/kg administered intravenously (approximately 8 times the therapeutic dose for HAE) caused erythema, itching and hypotension in healthy subjects. No therapeutic intervention was necessary.
-
11 DESCRIPTION
FIRAZYR (icatibant) is a synthetic decapeptide with five non-proteinogenic amino acids. The chemical structure of icatibant acetate is presented in Figure 1.
Figure 1 Chemical Structure
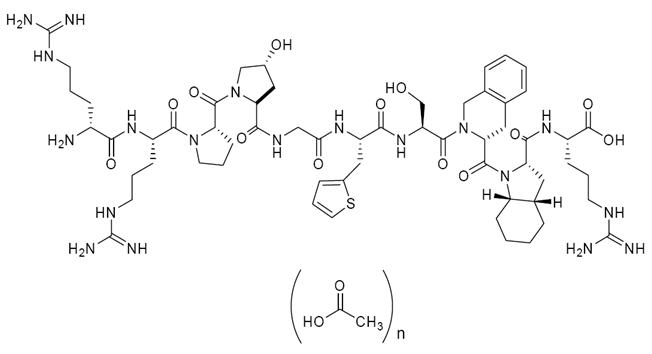
Chemical name: D-Arginyl-L-arginyl-L-prolyl-L[(4R)-4-hydroxyprolyl]-glycyl-L[3-(2-thienyl)alanyl]-L-seryl-D-(1,2,3,4-tetrahydroisoquinolin-3-ylcarbonyl)-L[(3aS,7aS)-octahydroindol-2-ylcarbonyl]-L-arginine, acetate salt
FIRAZYR is provided as a sterile, isotonic, and buffered solution of icatibant acetate in a single-use, prefilled syringe for subcutaneous administration. Each mL of the solution contains 10 mg of icatibant (free base). Each prefilled syringe delivers 3 mL of solution equivalent to a 30 mg icatibant dose. The solution is clear and colorless.
The solution also contains sodium chloride, glacial acetic acid, sodium hydroxide and water for injection with a pH of approximately 5.5. The solution does not contain preservatives.
Pharmacological class: Icatibant is a bradykinin B2 receptor antagonist.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Icatibant is a competitive antagonist selective for the bradykinin B2 receptor, with an affinity similar to bradykinin. Hereditary angioedema is caused by an absence or dysfunction of C1-esterase-inhibitor, a key regulator of the Factor XII/kallikrein proteolytic cascade that leads to bradykinin production. Bradykinin is a vasodilator which is thought to be responsible for the characteristic HAE symptoms of localized swelling, inflammation, and pain. Icatibant inhibits bradykinin from binding the B2 receptor and thereby treats the clinical symptoms of an acute, episodic attack of HAE.
12.2 Pharmacodynamics
Following bradykinin challenge, intravenous administration of FIRAZYR caused dose and time-dependent inhibition of development of bradykinin-induced hypotension, vasodilation, and reflex tachycardia in healthy young subjects. FIRAZYR intravenous doses of 0.4 and 0.8 mg/kg infused over 4 hours inhibited response to bradykinin challenge for 6 to 8 hours following completion of the infusion. Based on exposure-response analysis, a subcutaneous dose of 30 mg FIRAZYR is predicted to be effective against bradykinin challenge for at least 6 hours. The clinical significance of these findings is unknown.
The effect of FIRAZYR 30 and 90 mg following a single subcutaneous injection on QTc interval was evaluated in a randomized, placebo-, and active-controlled (moxifloxacin 400 mg) four-period crossover thorough QT study in 72 healthy subjects. In a study with demonstrated ability to detect small effects, the upper bound of the one-sided 95% confidence interval for the largest placebo adjusted, baseline-corrected QTc based on individual correction method (QTcI) was below 10 ms, the threshold for regulatory concern. The dose of 90 mg is adequate to represent the high exposure clinical scenario.
12.3 Pharmacokinetics
The pharmacokinetics of FIRAZYR has been characterized in studies using both intravenous and subcutaneous administration to healthy subjects and patients. The pharmacokinetic profile of FIRAZYR in patients with HAE is similar to that in healthy subjects.
The absolute bioavailability of FIRAZYR following a 30 mg subcutaneous dose is approximately 97%. Following subcutaneous administration of a single 30 mg dose of FIRAZYR to healthy subjects (N=96), a mean (± standard deviation) maximum plasma concentration (Cmax) of 974 ± 280 ng/mL was observed after approximately 0.75 hours. The mean area under the concentration-time curve (AUC0-∞) after a single 30 mg dose was 2165 ± 568 ng∙hr/mL, with no evidence of accumulation of icatibant following three 30 mg doses administered 6 hours apart. Following subcutaneous administration, plasma clearance was 245 ± 58 mL/min with a mean elimination half-life of 1.4 ± 0.4 hours and volume of distribution at steady state (Vss) of 29.0 ± 8.7 L.
Icatibant is extensively metabolized by proteolytic enzymes to inactive metabolites that are primarily excreted in the urine, with less than 10% of the dose eliminated as unchanged drug. Icatibant is not degraded by oxidative metabolic pathways, is not an inhibitor of major cytochrome P450 (CYP) isoenzymes (CYP 1A2, 2A6, 2B6, 2C8, 2C9, 2C19, 2D6, 2E1, and 3A4) and is not an inducer of CYP 1A2 and 3A4.
Special populations
Hepatic Impairment
The pharmacokinetic parameters of FIRAZYR were found to be generally comparable between healthy subjects (n=8) and mild to moderate (Child Pugh scores of 5 to 8) hepatic impaired patients (n=8) following a dose of 0.15 mg/kg/day as continuous intravenous infusion over 3 days. In a separate study, FIRAZYR clearance in subjects with a wide range of hepatic impairment (Child-Pugh scores of 7 to 15) was similar to that in healthy subjects. No dose adjustment is necessary for patients with impairment of hepatic function [see Use in Specific Populations (8.6)].
Renal Impairment
Since renal clearance of icatibant is a minor eliminating pathway, renal impairment is not expected to affect the pharmacokinetics of FIRAZYR and hence a formal renal impairment study was not conducted for FIRAZYR. In 10 patients with hepatorenal syndrome (GFR 30-60 mL/min), clearance of FIRAZYR was not dependent on renal function and therefore, did not show any observable differences in the plasma levels of icatibant or its metabolites compared to subjects with normal renal function. No dose adjustment is necessary for patients with impairment of renal function [see Use in Specific Populations (8.7)].
Age and Gender
Three 30 mg subcutaneous doses of FIRAZYR administered every 6 hours were studied in young (18 to 45 years of age) and elderly (over 65 years of age) healthy male and female subjects. Following single-dose administration of 30 mg subcutaneous FIRAZYR, elderly males and females showed approximately 2-fold higher AUC compared to young males and females, respectively. However, only minor differences (~12-14%) between Cmax of gender–matched elderly and young subjects were observed. Older subjects tend to exhibit lower clearance compared to younger subjects and therefore higher systemic exposure. Gender effect on FIRAZYR pharmacokinetics was also observed in addition to age effect. Clearance of FIRAZYR is significantly correlated with bodyweight with lower clearance values noted for lower bodyweights. Hence, females with typically lower body weights compared to males exhibit lower clearance values, resulting in approximately 2-fold higher systemic exposure (both AUC and Cmax) compared to males. Differences in efficacy and safety between elderly and younger patients and male and female patients have not been identified. Dose adjustment based on age and gender is not warranted.
Drug Interactions
Formal drug-drug interaction studies were not conducted with FIRAZYR. Icatibant metabolism is not mediated by CYP450 enzymes. In vitro study did not show any significant inhibition and/or induction of drug metabolizing CYP450 enzymes; therefore, metabolic drug interactions between FIRAZYR and CYP450 substrates, inhibitors and inducers are not expected.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year studies were conducted in CD1 mice and Wistar rats to assess the carcinogenic potential of FIRAZYR. No evidence of tumorigenicity was observed in mice and rats at icatibant subcutaneous doses up to 15 mg/kg/day (twice per week) and 6 mg/kg/day (daily), respectively (approximately 10-fold and 6-fold greater than the Maximum Recommended Human Dose on an AUC basis, respectively).
Icatibant tested negative for genotoxicity in the in vitro Ames bacterial reverse mutation test, in vitro Chinese hamster bone marrow chromosome aberration assay, and in vivo mouse micronucleus test.
Daily subcutaneous administration of icatibant to rats and dogs caused ovarian, uterine, and testicular atrophy/degeneration and adverse effects on the mammary and prostate glands. In rats, testicular atrophy, reduced prostate gland secretion, decreased testosterone levels and degenenerate corpora lutea occurred at doses greater than or equal to 3 mg/kg (approximately 5-fold greater than the MRHD in males and 2-fold greater than the MRHD in females on an AUC basis) and a decrease in developing ovarian follicles, mammary gland masculinization, and uterine atrophy occurred at doses greater than or equal to 10 mg/kg (approximately 6-fold greater than MRHD in females on an AUC basis). In dogs, reduced sperm counts and uterine atrophy occurred at doses greater than or equal to 1 mg/kg (approximately 2-fold greater than the MRHD on an AUC basis). Atrophy of the testes and prostate with decreased testosterone levels, decreased ovary size and decreased number of developing follicles occurred at a dose of 10 mg/kg (approximately 30-fold greater than the MRHD in males and 15-fold greater than at the MRHD in females on an AUC basis).
In contrast to the effects of daily icatibant administration, toxicity to the ovary, uterus, testis, mammary gland, and prostate did not occur in dogs treated twice a week for 9 months. AUC exposures from a dose of 3 mg/kg in these dogs were 5- and 3-fold the MRHD exposures in men and women, respectively. Sperm counts and testosterone remained unaffected over the course of the study in male dogs dosed twice a week.
Reproduction studies in male mice and rats with daily administration of icatibant found no effects on fertility or reproductive performance with intravenous doses up to 81 mg/kg (approximately 5-fold greater than the MRHD on a mg/m2 basis) or subcutaneous doses up to 10 mg/kg (approximately 11-fold greater than the MRHD on an AUC basis), respectively.
13.2 Animal Toxicology and/or Pharmacology
The B2 receptor has been implicated in the cardioprotective effects of bradykinin and antagonism of this receptor could potentially have negative cardiovascular effects during reperfusion after acute ischemia. Icatibant decreased coronary blood flow in the isolated guinea pig heart and aggravated the duration of post-ischemic reperfusion arrhythmias in the isolated rat heart. Intracoronary infusion of icatibant in an anesthetized myocardial infarction dog model increased mortality rate 2-fold over saline ischemia. There is limited human experience in acute ischemia. FIRAZYR should be used during acute coronary ischemia, unstable angina pectoris, or in the weeks following a stroke only if the benefit exceeds the theoretical risk to the patient.
-
14 CLINICAL STUDIES
The efficacy and safety of FIRAZYR for the treatment of acute attacks of HAE in adults were studied in three controlled clinical trials. Among the 223 patients in these studies, the mean age was 38 years, 64% were female, and 95% were white. Approximately 57% of patients reported use of attenuated androgens, antifibrinolytic agents, or C1 inhibitors. Response to therapy was primarily assessed using visual analog scores on a 100 mm scale and patient- and physician-reported symptom scores for abdominal and cutaneous pain and swelling.
Trial 1 was a randomized, placebo-controlled, double-blind, parallel-group study of 98 adult patients with a median age of 36 years. Patients who had developed moderate to severe cutaneous or abdominal or mild to moderate laryngeal attacks of HAE were randomized to receive either FIRAZYR 30 mg or placebo by subcutaneous injection. Patients with severe laryngeal attacks of HAE received open-label FIRAZYR 30 mg. The primary endpoint was assessed using a 3-item composite visual analog score (VAS), comprised of averaged assessments of skin swelling, skin pain, and abdominal pain. Response was defined as at least a 50% reduction from the pretreatment composite 3-item VAS score (Figure 2). The median time to 50% reduction in symptoms for patients with cutaneous or abdominal attacks treated with FIRAZYR (n=43) compared to placebo (n=45) was 2.0 hours [95% CI 1.5, 3.0] versus 19.8 hours [95% CI 6.1, 26.3], respectively (p<0.001).
Figure 2 Time to 50% reduction from baseline in 3-item VAS score.
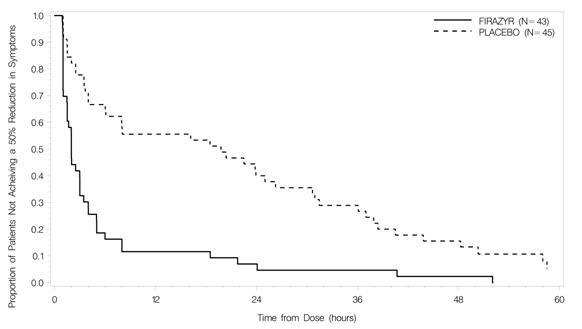
Other evaluated endpoints included time to almost complete symptom relief (VAS<10 mm) and rescue medication use. In Trial 1, the median times to almost complete symptom relief were 8.0 versus 36.0 hours for FIRAZYR and placebo, respectively. In terms of rescue medication use, 3/43 (7%) patients treated with FIRAZYR used additional rescue medication in comparison to 18/45 (40%) patients treated with placebo.
In a second placebo-controlled trial and an active-controlled trial, a total of 26 and 35 patients, respectively, received FIRAZYR 30 mg for the treatment of an acute HAE attack. Across the three trials, FIRAZYR had a median time to 50% reduction from baseline symptoms ranging from 2.0 to 2.3 hours.
Recurrent attacks
In all three controlled trials, patients were eligible for treatment of subsequent attacks in an open-label extension. Patients were treated with FIRAZYR 30 mg and could receive up to 3 doses of FIRAZYR 30 mg administered at least 6 hours apart for each attack. A total of 225 patients were treated with 1,076 doses of 30 mg FIRAZYR for 987 attacks of acute HAE in these trials. In an assessment of the first 5 FIRAZYR-treated attacks (621 doses for 582 attacks), the median times to a 50% reduction from the pretreatment composite 3-itemVAS score were similar across attacks (2.0, 2.0, 2.4, 2.0, 1.5 hours). The majority (93%) of these attacks of HAE were treated with a single dose of FIRAZYR.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
FIRAZYR is supplied as a single-use, prefilled syringe for subcutaneous administration. Each syringe delivers 3 mL of a sterile solution of icatibant 30 mg (as icatibant acetate). Each glass syringe has a bromobutyl plunger stopper, which is not made of latex natural rubber.
FIRAZYR is available in cartons containing one single-use, prefilled syringe and one 25 G Luer lock needle. NDC: 54092-702-02.
FIRAZYR is also available in a pack containing 3 cartons; each carton contains one single-use, prefilled syringe and one 25 G Luer lock needle. NDC: 54092-702-03.
-
17 PATIENT COUNSELING INFORMATION
See FDA-approved patient labeling (Patient Information and Instructions for Use).
17.1 Information for Patients
Patients may self-administer FIRAZYR upon recognition of an HAE attack after training under the guidance of a healthcare professional.
Patients with laryngeal symptoms should seek medical attention immediately in an appropriate healthcare facility after administration of FIRAZYR [see Warnings and Precautions (5.1)].
Injection site reactions are reported in most patients after administration of FIRAZYR. Other adverse reactions reported after administration of FIRAZYR include pyrexia, increase in transaminases, dizziness, and rash [see Adverse Reactions (6.1)].
Tiredness, drowsiness, and dizziness have been reported following the use of FIRAZYR. Patients should be advised not to drive or use machinery if they feel tired or dizzy.
- SPL UNCLASSIFIED SECTION
-
Patient InformationFIRAZYR® (FIR-a-zeer)(icatibant) Injection
Please read this Patient Information before you use FIRAZYR and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment.
What is FIRAZYR?
FIRAZYR is a medicine used to treat acute attacks of hereditary angioedema (HAE) in adults 18 years and older. It is not known if FIRAZYR is safe or effective for children under 18 years of age.
What should I tell my healthcare provider before taking FIRAZYR?
Before you use FIRAZYR, tell your healthcare provider if you:
- have any other medical conditions.
- are breastfeeding or plan to breastfeed. It is not known if FIRAZYR passes into your breast milk. Talk to your healthcare provider about the best way to feed your baby if you use FIRAZYR.
- are pregnant or plan to become pregnant. It is not known if FIRAZYR will harm your unborn baby. You and your healthcare provider will decide if FIRAZYR is right for you.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
How should I use FIRAZYR?
- Use FIRAZYR exactly as your healthcare provider tells you to use it.
- Your healthcare provider will prescribe the right dose of FIRAZYR for you and tell you when to use it.
- Your healthcare provider will teach you or a caregiver how to give FIRAZYR injections
- Read the Instructions for Use at the end of the Patient Information for information about the right way to use FIRAZYR.
- If your symptoms continue or come back, you may repeat your FIRAZYR injection at least six hours apart.
- Do not use more than 3 doses in 24 hours.
- If you have a laryngeal attack, inject FIRAZYR and then go to the nearest hospital emergency room right away.
What should I avoid while using FIRAZYR?
Tiredness, drowsiness, and dizziness can occur in people who take FIRAZYR. If this occurs, do not drive a car, use machinery, or do anything that needs you to be alert.
What are the possible side effects of FIRAZYR?
The most common side effects of FIRAZYR include:
- redness, bruising, swelling, warmth, burning, itching, irritation, hives, numbness, pressure, or pain at the injection site
- fever
- too much of an enzyme called transaminase in your blood
- dizziness
- nausea
- headache
- rash
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all of the possible side effects of FIRAZYR. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store FIRAZYR?
- Store FIRAZYR between 36°F to 77°F (2°C to 25°C).
- Do not freeze.
- Store FIRAZYR in the original carton until you are ready to use it.
Keep FIRAZYR and all medicines out of the reach of children.
General information about the safe and effective use of FIRAZYR
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use FIRAZYR for a condition for which it was not prescribed. Do not give FIRAZYR to other people, even if they have the same symptoms that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about FIRAZYR. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about FIRAZYR that is written for health professionals.
For more information, go to www.FIRAZYR.com or call 1-800-828-2088.
What are the ingredients in FIRAZYR?
Active ingredient: icatibant acetate
Inactive Ingredients: sodium chloride, glacial acetic acid, sodium hydroxide, and water
-
Step-by-Step Instructions for your FIRAZYR injection
Step 1. Preparing your dose of FIRAZYR
- Wash your hands with soap and water.
- You will need the following supplies:
- Your FIRAZYR carton that includes 1 single-use FIRAZYR prefilled syringe and 1 needle.
- An alcohol wipe
- The medicine inside your FIRAZYR prefilled syringe should be clear and colorless. Do not use your FIRAZYR prefilled syringe if the solution contains particles, is cloudy, or an unusual color.
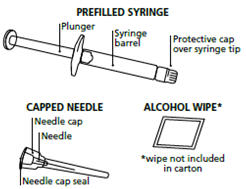
Figure A
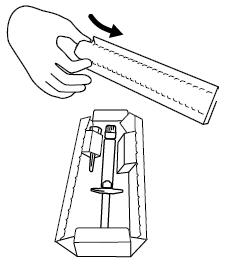
Step 2. Remove the prefilled syringe and needle from the carton. See Figure B.
Step 3. Remove the seal from the needle cap (the needle should remain inside the protective needle cap until ready to use). See Figure C.
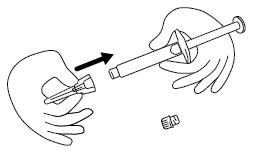
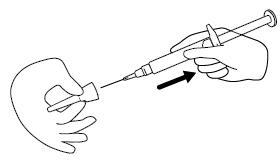
Step 4. Remove the protective cap from the end of the pre-filled syringe by unscrewing the cap. Hold the syringe firmly. Carefully attach the needle to the prefilled syringe containing the colorless FIRAZYR solution. See Figure D.
Step 5. Firmly screw the needle on the prefilled syringe. Be careful not to remove the needle from the needle cap. See Figure E.
Preparing the Injection Site
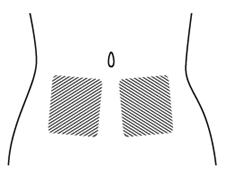
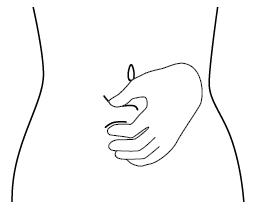
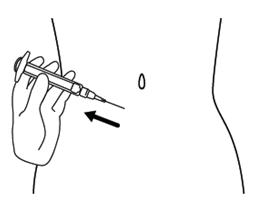
Step 6. Choose the injection site. The injection site should be a fold of skin on your stomach, about 2 to 4 inches (5 to 10 cm) below your belly button on either side. See Figure F.
The area you choose for injection should be at least 2 inches (5 cm) away from any scars. Do not choose an area that is bruised, swollen, or painful.
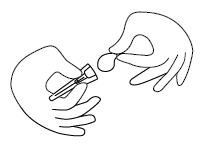
Step 7. Clean your FIRAZYR injection site with an alcohol wipe and allow it to dry. See Figure G.
Injecting your FIRAZYR
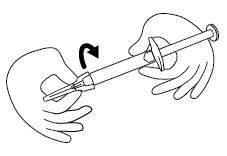
Step 8. Remove the needle from the needle cap by holding the needle cap and carefully pulling the syringe. Do not pull up on the plunger. See Figure H.
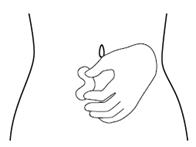
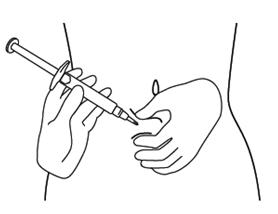
Step 9. Hold the FIRAZYR prefilled syringe in 1 hand, between your fingers and thumb. See Figure I.
Step 10. Use your other hand to gently pinch the fold of skin you cleaned with the alcohol wipe between your thumb and fingers for your injection. See Figure J.
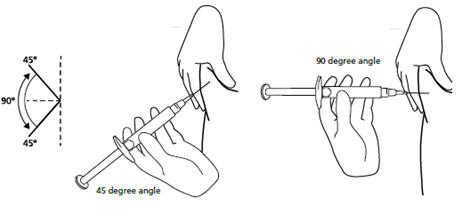
Step 11. Hold the syringe between a 45 to 90 degree angle to your skin with the needle facing the fold of skin you are holding. See Figure K.
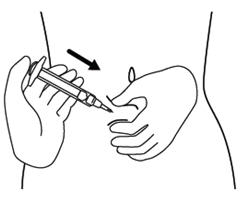
Step 12. Hold the fold of skin. Bring the syringe to the skin and quickly insert the needle into the skin fold. See Figure L.
Step 13. Push the plunger, at the top of the syringe, over at least 30 seconds until no FIRAZYR is in the syringe. See Figure M.
Step 14. Release the skin fold and gently pull the needle out. See Figure N.
Disposal of your used FIRAZYR prefilled syringe
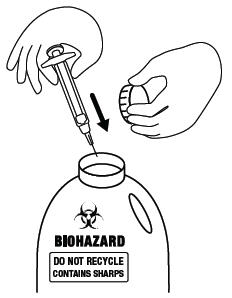
Step 15. Place the used FIRAZYR syringe, with the needle attached, in a sharps container (such as a red biohazard container), a hard plastic container, (such as a detergent bottle), or a metal container (such as an empty coffee can). Seal the container and throw it away the right way. There may be state and local laws about the right way to throw away used syringes and needles. Ask your healthcare provider or pharmacist how to throw away used syringes and needles. See Figure O.
This Patient Package Insert and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Manufactured for:
Shire Orphan Therapies LLC
300 Shire Way
Lexington, MA 02421December2015
-
PRINCIPAL DISPLAY PANEL - 10 mg/mL Syringe Box
Shire
Prefilled Syringe
Rx ONLY
NDC: 54092-702-02Carton contains:
One single-dose, single-use,
prefilled syringe and one 25G
hypodermic needle.
Full prescribing information with
patient injection instructions.
The syringe is closed with a protective cap.www.firazyr.com
www.shire.com
1-800-828-2088firazyr®
(icatibant injection)
30 mg/3 mL
(10 mg/mL)
FOR SUBCUTANEOUS USE ONLY
-
PRINCIPAL DISPLAY PANEL - 10 mg/mL Carton
Shire
Prefilled SyringeRx ONLY
NDC: 54092-702-03Carton contains:
Three cartons, each with one single-dose, single-use, prefilled syringe
and one 25G hypodermic needle.
Full prescribing information with patient injection instructions.
The syringe is closed with a protective cap.www.firazyr.com
www.shire.com
1-800-828-2088firazyr®
(icatibant injection)
30 mg/3 mL
(10 mg/mL)FOR SUBCUTANEOUS USE ONLY
-
PRINCIPAL DISPLAY PANEL - 30 mg/3 mL Syringe Carton
Shire
Prefilled Syringe
Rx ONLY
NDC: 54092-702-01Sample - Not For Sale
Carton contains:
One single-dose, single-use,
prefilled syringe and one 25G
hypodermic needle.
Full prescribing information with
patient injection instructions.
The syringe is closed with a protective cap.www.firazyr.com
www.shire.com
1-800-828-2088firazyr®
(icatibant injection)
30 mg/3 mL
(10 mg/mL)
FOR SUBCUTANEOUS USE ONLY
-
INGREDIENTS AND APPEARANCE
FIRAZYR
icatibant acetate injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 54092-702 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength icatibant acetate (UNII: 325O8467XK) (icatibant - UNII:7PG89G35Q7) icatibant 30.0 mg in 3 mL Inactive Ingredients Ingredient Name Strength sodium hydroxide (UNII: 55X04QC32I) 1.95 mg in 3 mL acetic acid (UNII: Q40Q9N063P) 4.03 mg in 3 mL sodium chloride (UNII: 451W47IQ8X) 22.72 mg in 3 mL water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 54092-702-02 1 in 1 CARTON 08/25/2011 1 3 mL in 1 SYRINGE, GLASS; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 2 NDC: 54092-702-03 3 in 1 CARTON 08/25/2011 2 3 mL in 1 SYRINGE, GLASS; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 3 NDC: 54092-702-01 1 in 1 CARTON 08/25/2011 3 3 mL in 1 SYRINGE, GLASS; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022150 08/25/2011 Labeler - Shire US Manufacturing Inc. (964907406)
Trademark Results [Firazyr]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 FIRAZYR 78876453 3329286 Live/Registered |
Shire Orphan Therapies GmbH 2006-05-04 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.