GUAIFENESIN tablet, extended release
Guaifenesin by
Drug Labeling and Warnings
Guaifenesin by is a Otc medication manufactured, distributed, or labeled by BluePoint Laboratories, APL HEALTHCARE LIMITED. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredient
- Purpose
- Uses
- Warnings
- Ask a doctor before use if you have
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children
- Directions
- Other information
- Inactive ingredients
- Questions?
-
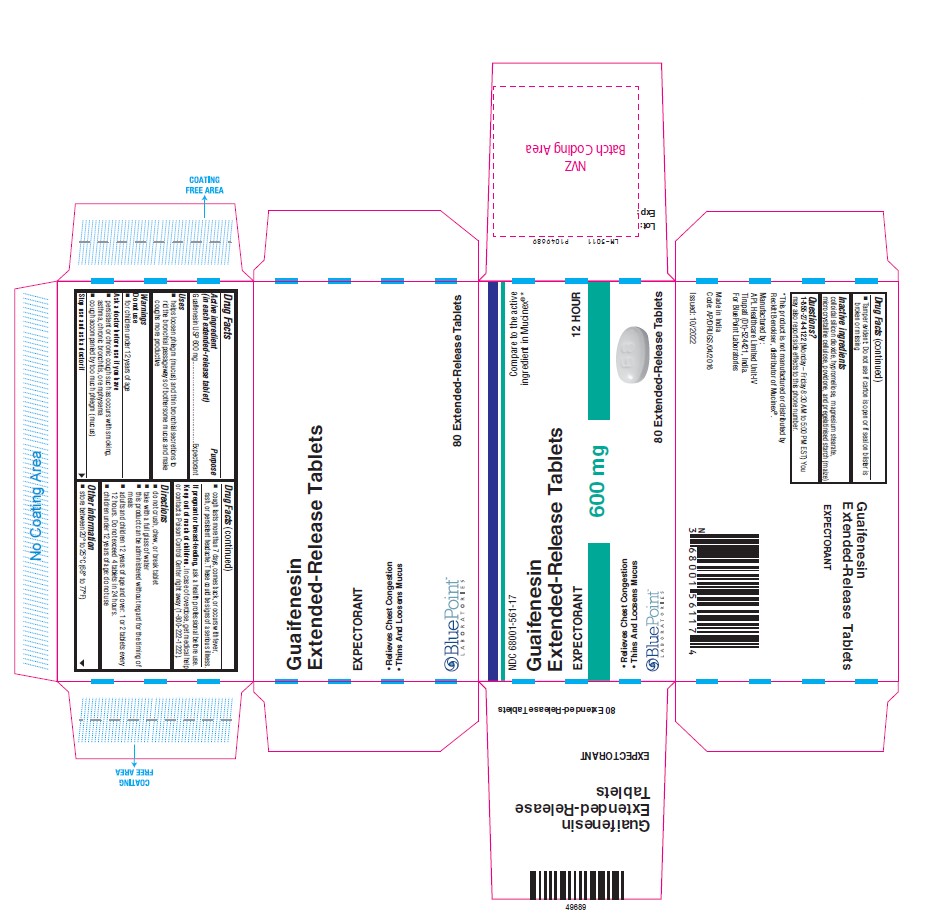
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 600 mg Blister Foil and 600 mg Carton (80 Extended-Release Tablets, 1x10's).
Blister Pack : NDC: 68001-561-16
Carton Pack : NDC: 68001-561-17
Guaifenesin Extended-Release Tablets 600 mg
EXPECTORANT
12 HOUR- Relieves Chest Congestion
- Thins And Loosens Mucus
80 Extended-Release Tablets
-
INGREDIENTS AND APPEARANCE
GUAIFENESIN
guaifenesin tablet, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 68001-561 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 600 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) HYPROMELLOSE 2208 (15000 MPA.S) (UNII: Z78RG6M2N2) HYPROMELLOSE 2910 (10000 MPA.S) (UNII: 0HO1H52958) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE 101 (UNII: 7T9FYH5QMK) MICROCRYSTALLINE CELLULOSE 102 (UNII: PNR0YF693Y) POVIDONE K90 (UNII: RDH86HJV5Z) POVIDONE K25 (UNII: K0KQV10C35) STARCH, CORN (UNII: O8232NY3SJ) Product Characteristics Color white (white to off-white) Score no score Shape OVAL Size 16mm Flavor Imprint Code L;68 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68001-561-17 8 in 1 CARTON 03/15/2023 1 NDC: 68001-561-16 10 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210453 03/15/2023 Labeler - BluePoint Laboratories (985523874) Establishment Name Address ID/FEI Business Operations APL HEALTHCARE LIMITED 650918514 analysis(68001-561) , manufacture(68001-561)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.