ZONTIVITY- vorapaxar tablet, film coated
ZONTIVITY by
Drug Labeling and Warnings
ZONTIVITY by is a Prescription medication manufactured, distributed, or labeled by Aralez Pharmaceuticals Us Inc., ARALEZ PHARMACEUTICALS TRADING DESIGNATED ACTIVITY COMPANY. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ZONTIVITY safely and effectively. See full prescribing information for ZONTIVITY.
ZONTIVITY® (vorapaxar) Tablets 2.08 mg*, for oral use
*Equivalent to 2.5 mg vorapaxar sulfate
Initial U.S. Approval: 2014WARNING: BLEEDING RISK
See full prescribing information for complete boxed warning.
INDICATIONS AND USAGE
ZONTIVITY is a protease-activated receptor-1 (PAR-1) antagonist indicated for the reduction of thrombotic cardiovascular events in patients with a history of myocardial infarction (MI) or with peripheral arterial disease (PAD). ZONTIVITY has been shown to reduce the rate of a combined endpoint of cardiovascular death, MI, stroke, and urgent coronary revascularization. (1.1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Tablets: 2.08 mg vorapaxar. (3)
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
- Bleeding, including life-threatening and fatal bleeding, is the most commonly reported adverse reaction. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Aralez Pharmaceuticals at 1-833-694-8235 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 11/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: BLEEDING RISK
1 INDICATIONS AND USAGE
1.1 Patients with History of Myocardial Infarction (MI) or with Peripheral Arterial Disease (PAD)
2 DOSAGE AND ADMINISTRATION
2.1 General Dosing Information
2.2 Coadministration with Other Antiplatelet Drugs
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
4.1 History of Stroke, Transient Ischemic Attack (TIA), or Intracranial Hemorrhage (ICH)
4.2 Active Pathologic Bleeding
5 WARNINGS AND PRECAUTIONS
5.1 General Risk of Bleeding
5.2 Strong CYP3A Inhibitors or Inducers
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Effects of Other Drugs on ZONTIVITY
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Pharmacology
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: BLEEDING RISK
- Do not use ZONTIVITY in patients with a history of stroke, transient ischemic attack (TIA), or intracranial hemorrhage (ICH); or active pathological bleeding [see CONTRAINDICATIONS (4.1, 4.2)].
- Antiplatelet agents, including ZONTIVITY, increase the risk of bleeding, including ICH and fatal bleeding [see Warnings and Precautions (5.1)].
-
1 INDICATIONS AND USAGE
1.1 Patients with History of Myocardial Infarction (MI) or with Peripheral Arterial Disease (PAD)
ZONTIVITY® is indicated for the reduction of thrombotic cardiovascular events in patients with a history of myocardial infarction (MI) or with peripheral arterial disease (PAD). ZONTIVITY has been shown to reduce the rate of a combined endpoint of cardiovascular death, MI, stroke, and urgent coronary revascularization (UCR).
-
2 DOSAGE AND ADMINISTRATION
2.1 General Dosing Information
Take one tablet of ZONTIVITY 2.08 mg orally once daily, with or without food.
2.2 Coadministration with Other Antiplatelet Drugs
There is no experience with use of ZONTIVITY alone as the only administered antiplatelet agent. ZONTIVITY has been studied only as an addition to aspirin and/or clopidogrel. Use ZONTIVITY with aspirin and/or clopidogrel according to their indications or standard of care [see Clinical Studies (14)]. There is limited clinical experience with other antiplatelet drugs.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
4.1 History of Stroke, Transient Ischemic Attack (TIA), or Intracranial Hemorrhage (ICH)
ZONTIVITY is contraindicated in patients with a history of stroke, TIA, or ICH because of an increased risk of ICH in this population [see Adverse Reactions (6)].
Discontinue ZONTIVITY in patients who experience a stroke, TIA, or ICH [see Adverse Reactions (6.1) and Clinical Studies (14)].
4.2 Active Pathologic Bleeding
ZONTIVITY is contraindicated in patients with active pathological bleeding such as ICH or peptic ulcer [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
-
5 WARNINGS AND PRECAUTIONS
5.1 General Risk of Bleeding
Antiplatelet agents, including ZONTIVITY, increase the risk of bleeding, including ICH and fatal bleeding [see Adverse Reactions (6.1)].
ZONTIVITY increases the risk of bleeding in proportion to the patient's underlying bleeding risk. Consider the underlying risk of bleeding before initiating ZONTIVITY. General risk factors for bleeding include older age, low body weight, reduced renal or hepatic function, history of bleeding disorders, and use of certain concomitant medications (e.g., anticoagulants, fibrinolytic therapy, chronic nonsteroidal anti-inflammatory drugs [NSAIDS], selective serotonin reuptake inhibitors, serotonin norepinephrine reuptake inhibitors) increases the risk of bleeding [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)]. Avoid concomitant use of warfarin or other anticoagulants.
Suspect bleeding in any patient who is hypotensive and has recently undergone coronary angiography, percutaneous coronary intervention (PCI), coronary artery bypass graft surgery (CABG), or other surgical procedures.
Withholding ZONTIVITY for a brief period will not be useful in managing an acute bleeding event because of its long half-life. There is no known treatment to reverse the antiplatelet effect of ZONTIVITY. Significant inhibition of platelet aggregation remains 4 weeks after discontinuation [see Overdosage (10) and Clinical Pharmacology (12.2, 12.3)].
5.2 Strong CYP3A Inhibitors or Inducers
Strong CYP3A inhibitors increase and inducers decrease ZONTIVITY exposure. Avoid concomitant use of ZONTIVITY with strong CYP3A inhibitors or inducers [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
-
6 ADVERSE REACTIONS
The following serious adverse reaction is also discussed elsewhere in the labeling:
- Bleeding [see Boxed Warning and Warnings and Precautions (5.1)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
ZONTIVITY was evaluated for safety in 13,186 patients, including 2,187 patients treated for more than 3 years, in the Phase 3 study TRA 2°P TIMI 50 (Thrombin Receptor Antagonist in Secondary Prevention of Atherothrombotic Ischemic Events). The overall study population, patients who had evidence or a history of atherosclerosis involving the coronary (post-MI), cerebral (ischemic stroke), or peripheral vascular (documented history of PAD) systems, was treated once a day with ZONTIVITY (n=13,186) or placebo (n=13,166). Patients randomized to ZONTIVITY received treatment for a median of 2.3 years.
The adverse events in the ZONTIVITY-treated (n=10,059) and placebo-treated (n=10,049) post-MI or PAD patients with no history of stroke or TIA are shown below [see Contraindications (4)].
Bleeding
GUSTO severe bleeding was defined as fatal, intracranial, or bleeding with hemodynamic compromise requiring intervention; GUSTO moderate bleeding was defined as bleeding requiring transfusion of whole blood or packed red blood cells without hemodynamic compromise. (GUSTO: Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Arteries.)The results for the bleeding endpoints in the post-MI or PAD patients without a history of stroke or TIA are shown in Table 1. ZONTIVITY increased GUSTO moderate or severe bleeding by 55%.
Table 1: Non-CABG-Related Bleeds in Post-MI or PAD Patients without a History of Stroke or TIA (First Dose to Last Dose + 30 Days) in the TRA 2°P Study Placebo
(n=10,049)ZONTIVITY
(n=10,059)Endpoints Patients with events
(%)K-M %* Patients with events
(%)K-M %* Hazard Ratio†,‡
(95% CI)- * K-M estimate at 1,080 days.
- † Clinically significant bleeding includes any bleeding requiring medical attention including ICH, or clinically significant overt signs of hemorrhage associated with a drop in hemoglobin (Hgb) of ≥3 g/dL (or, when Hgb is not available, an absolute drop in hematocrit (Hct) of ≥9%).
- ‡ Hazard ratio is ZONTIVITY group vs. placebo group.
GUSTO Bleeding Categories Severe 82 (0.8%) 1.0% 100 (1.0%) 1.3% 1.24 (0.92 - 1.66) Moderate or Severe 199 (2.0%) 2.4% 303 (3.0%) 3.7% 1.55 (1.30 - 1.86) Any GUSTO Bleeding (Severe/Moderate/Mild) 1769 (17.6%) 19.8% 2518 (25.0%) 27.7% 1.52 (1.43 - 1.61) Fatal Bleeding 14 (0.1%) 0.2% 16 (0.2%) 0.2% 1.15 (0.56 - 2.36) Intracranial Hemorrhage (ICH) 31 (0.3%) 0.4% 45 (0.4%) 0.6% 1.46 (0.92-2.31) Clinically Significant Bleeding† 950 (9.5%) 10.9% 1349 (13.4%) 15.5% 1.47 (1.35 - 1.60) Gastrointestinal Bleeding 297 (3.0%) 3.5% 400 (4.0%) 4.7% 1.37 (1.18-1.59) The effects of ZONTIVITY on bleeding were examined in a number of subsets based on demographic and other baseline characteristics. Many of these are shown in Figure 1. Such analyses must be interpreted cautiously, as differences can reflect the play of chance among a large number of analyses.
Figure 1: Subgroup Analyses (GUSTO Moderate or Severe Bleeding) in Post-MI or PAD Patients without a History of Stroke or TIA in the TRA 2°P Study (First Dose to Last Dose + 30 Days) 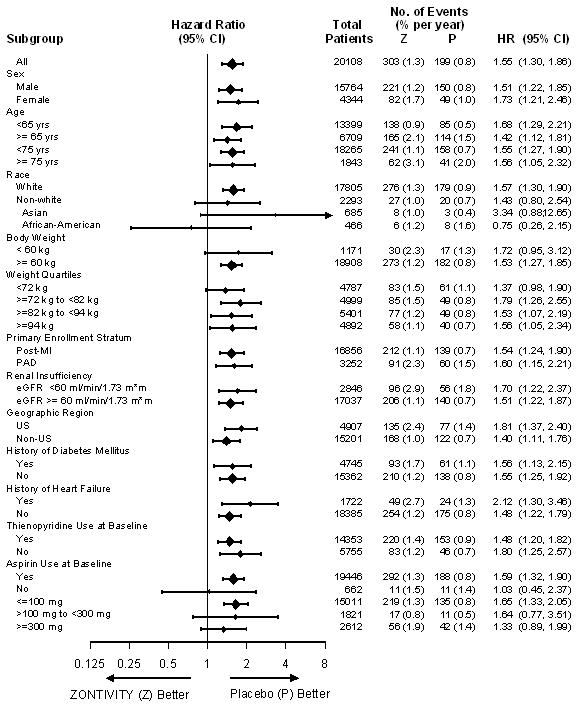
In TRA 2°P, 367 post-MI or PAD patients without a history of stroke or TIA underwent CABG surgery. Study investigators were encouraged not to discontinue treatment with study drug (i.e., ZONTIVITY or placebo) prior to surgery. Approximately 12.3% of patients discontinued ZONTIVITY more than 30 days prior to CABG. The relative risk for GUSTO moderate or severe bleeding was approximately 1.2 on ZONTIVITY vs. placebo.
Bleeding events that occurred on ZONTIVITY were treated in the same manner as for other antiplatelet agents.
Use in Patients with History of Stroke, TIA, or ICH
In the TRA 2°P study, patients with a history of ischemic stroke had a higher rate for ICH on ZONTIVITY than on placebo. ZONTIVITY is contraindicated in patients with a history of stroke, TIA, or ICH [see Contraindications (4)].Other Adverse Reactions
Adverse reactions other than bleeding were evaluated in 19,632 patients treated with ZONTIVITY [13,186 patients in the TRA 2°P study and 6,446 patients in the TRACER (Thrombin Receptor Antagonist for Clinical Event Reduction in Acute Coronary Syndrome) study]. Adverse events other than bleeding that occurred at a rate that was at least 2% in the ZONTIVITY group and also 10% greater than the rate in the placebo group are shown in Table 2.Table 2: TRA 2°P / TRACER - Percentage of Patients Reporting Non-hemorrhagic Adverse Reactions at a Rate at Least 2% in the ZONTIVITY Group and at Least 10% Greater than Placebo ZONTIVITY
N=19,632Placebo
N=19,607n (%) n (%) Anemia 982 (5.0) 783 (4.0) Depression 477 (2.4) 405 (2.1) Rashes, Eruptions, and Exanthemas 439 (2.2) 395 (2.0) The following adverse reactions occurred at a rate less than 2% in the ZONTIVITY group but at least 40% greater than placebo. In descending order of rate in the ZONTIVITY group: iron deficiency, retinopathy or retinal disorder, and diplopia/oculomotor disturbances.
An increased rate of diplopia and related oculomotor disturbances was observed with ZONTIVITY treatment (30 subjects, 0.2%) vs. placebo (10 subjects, 0.06%). While some cases resolved during continued treatment, information on resolution of symptoms was not available for some cases.
-
7 DRUG INTERACTIONS
7.1 Effects of Other Drugs on ZONTIVITY
Vorapaxar is eliminated primarily by metabolism, with contributions from CYP3A4 and CYP2J2.
Strong CYP3A Inhibitors
Avoid concomitant use of ZONTIVITY with strong inhibitors of CYP3A (e.g., ketoconazole, itraconazole, posaconazole, clarithromycin, nefazodone, ritonavir, saquinavir, nelfinavir, indinavir, boceprevir, telaprevir, telithromycin and conivaptan) [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)].Strong CYP3A Inducers
Avoid concomitant use of ZONTIVITY with strong inducers of CYP3A (e.g., rifampin, carbamazepine, St. John's Wort and phenytoin) [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)]. -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on the potential for serious adverse reactions (such as maternal bleeding/hemorrhage) and the long half-life which makes it effectively irreversible, discontinue vorapaxar when pregnancy is detected and initiate alternative therapy with a shorter duration of action [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.1, 12.2)]. Available data from postmarketing experience with ZONTIVITY use in pregnant women are insufficient to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. In animal reproduction studies, no embryo/fetal toxicities, malformations or maternal toxicities were observed in rats and rabbits exposed during the period of organogenesis at exposures 56 times and 26 times, respectively, the human systemic exposure at the recommended human dose (RHD) (see Data).The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In the rat embryo/fetal developmental toxicity study, pregnant rats received daily oral doses of vorapaxar at 0, 5, 25, and 75 mg/kg from implantation to closure of the fetal hard palate (6th to 17th day of gestation). Maternal systemic exposures were approximately 0, 7, 56, and 285 times greater than exposures in women treated at the RHD based on AUC. No embryo/fetal toxicities, malformations, or maternal toxicities were observed in rats receiving exposures up to 56 times the human systemic exposure at the RHD.In the rabbit embryo/fetal developmental toxicity study, pregnant rabbits received daily oral doses of vorapaxar at 0, 2, 10, or 20 mg/kg from implantation to closure of the fetal hard palate (7th to 19th day of gestation). The NOAEL for maternal and fetal toxicity was equal to or above the highest dose tested. However, an overall increase in the number of litters with any malformation was observed at the highest dose, where systemic exposures were 89-fold higher than the human exposure at RHD.
The effects of vorapaxar on prenatal and postnatal development were assessed in pregnant rats dosed at 0, 5, 25, or 50 mg/kg/day from implantation through the end of lactation. Rat pups had decreased survival and body weight gain from birth to postnatal day 4 and decreased body weight gain for the overall pre-weaning period at exposures 67 times the human exposure at the RHD. Both male and female pups displayed effects on sensory function (acoustic startle) and neurobehavioral (locomotor assay) development on post-natal day (PND) 20 and 21, but not later (PND 60, 61) in development, whereas decreased memory was observed in female pups on PND 27 at 31 times the human exposure at the RHD. In utero and lactational exposure did not affect fertility or reproductive behavior of offspring at exposures up to 67 times the RHD.
8.2 Lactation
Risk Summary
There are no data on the presence of vorapaxar or its metabolites in human milk, the effects on the breastfed infant, or the effects on milk production. When vorapaxar was administered to lactating rats, vorapaxar was actively secreted in the milk of rats. When a drug is present in animal milk, it is likely the drug will be present in human milk. Because of the potential for serious adverse reactions in the breastfed infant, such as bleeding, advise patients that breastfeeding is not recommended during treatment with ZONTIVITY.8.4 Pediatric Use
The safety and effectiveness of ZONTIVITY in pediatric patients have not been established.
8.5 Geriatric Use
In TRA 2°P, in post-MI or PAD patients without a history of stroke or TIA, 33% of patients were ≥65 years of age and 9% were ≥75 years of age. The relative risk of bleeding (ZONTIVITY compared with placebo) was similar across age groups. No overall differences in safety or effectiveness were observed between these patients and younger patients. ZONTIVITY increases the risk of bleeding in proportion to a patient's underlying risk. Because older patients are generally at a higher risk of bleeding, consider patient age before initiating ZONTIVITY [see Adverse Reactions (6.1)].
8.6 Renal Impairment
No dose adjustment is required in patients with renal impairment [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
No dose adjustment is required in patients with mild and moderate hepatic impairment. Based on the increased inherent risk of bleeding in patients with severe hepatic impairment, ZONTIVITY is not recommended in such patients [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
There is no known treatment to reverse the antiplatelet effect of ZONTIVITY, and neither dialysis nor platelet transfusion can be expected to be beneficial if bleeding occurs after overdose. Inhibition of platelet aggregation can be expected for weeks after discontinuation of normal dosing [see Clinical Pharmacology (12.2)]. There is no standard test available to assess the risk of bleeding in an overdose situation.
-
11 DESCRIPTION
ZONTIVITY contains vorapaxar sulfate, a tricyclic himbacine-derived selective inhibitor of platelet aggregation mediated by PAR-1.
The chemical name of vorapaxar sulfate is ethyl [(1R,3aR,4aR,6R,8aR,9S,9aS)-9-{(1E)-2-[5-(3-fluorophenyl)pyridin-2-yl]ethen-1-yl}-1-methyl-3-oxododecahydronaphtho[2,3-c]furan-6-yl]carbamate sulfate. The empirical formula is C29H33FN2O4∙H2SO4, and its molecular weight is 590.7. The structural formula is:
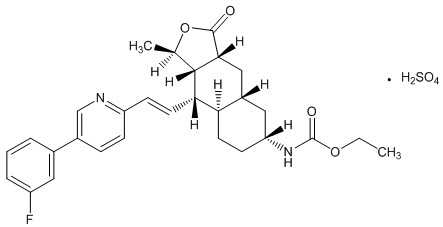
Vorapaxar sulfate is a white to off-white solid. Vorapaxar sulfate is freely soluble in methanol and slightly soluble in ethanol, acetone, 2-propanol, and acetonitrile. In aqueous solution, it is slightly soluble in pH 1; its solubility decreases with increasing pH. ZONTIVITY tablets are formulated with vorapaxar sulfate, but during manufacture and storage, partial conversion from vorapaxar sulfate to vorapaxar free base may occur.
ZONTIVITY is available for oral use as tablets containing 2.08 mg of vorapaxar, which is equivalent to 2.5 mg of vorapaxar sulfate.
Each film-coated tablet of ZONTIVITY contains the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, croscarmellose sodium, povidone, and magnesium stearate. In addition, the film coating contains the following inactive ingredients: lactose monohydrate, hypromellose, titanium dioxide, triacetin (glycerol triacetate), and iron oxide yellow.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Vorapaxar is a reversible antagonist of the protease-activated receptor-1 (PAR-1) expressed on platelets, but its long half-life makes it effectively irreversible. Vorapaxar inhibits thrombin-induced and thrombin receptor agonist peptide (TRAP)-induced platelet aggregation in in vitro studies. Vorapaxar does not inhibit platelet aggregation induced by adenosine diphosphate (ADP), collagen or a thromboxane mimetic and does not affect coagulation parameters ex vivo. PAR-1 receptors are also expressed in a wide variety of cell types, including endothelial cells, neurons, and smooth muscle cells, but the pharmacodynamic effects of vorapaxar in these cell types have not been assessed.
12.2 Pharmacodynamics
At the recommended dose, ZONTIVITY achieves ≥80% inhibition of TRAP-induced platelet aggregation within one week of initiation of treatment. The duration of platelet inhibition is dose- and concentration-dependent. Inhibition of TRAP-induced platelet aggregation at a level of 50% can be expected at 4 weeks after discontinuation of daily doses of ZONTIVITY 2.08 mg, consistent with the terminal elimination half-life of vorapaxar [see Clinical Pharmacology (12.3)].
In healthy volunteer studies, no changes in platelet P-selectin and soluble CD40 ligand (sCD40L) expression or coagulation test parameters (TT, PT, aPTT, ACT, ECT) occurred after single- or multiple- dose (28 days) administration of vorapaxar. No meaningful changes in P-selectin, sCD40L, or hs-CRP concentrations were observed in patients treated with vorapaxar in the phase 2/3 clinical trials.
12.3 Pharmacokinetics
Vorapaxar exposure increases in an approximately dose-proportional manner following single doses up to 16 times the recommended dose. Vorapaxar pharmacokinetics are similar in healthy subjects and patients.
Absorption
After oral administration of a single ZONTIVITY 2.08 mg dose under fasted conditions, peak concentrations (Cmax) occur at 1 hour post-dose (range: 1 to 2 h). The mean absolute bioavailability as determined from a microdosing study is approximately 100%.Ingestion of vorapaxar with a high-fat meal resulted in no meaningful change in AUC with a small (21%) decrease in Cmax and delayed time to peak concentration (45 minutes). ZONTIVITY may be taken with or without food.
Distribution
The mean volume of distribution of vorapaxar is approximately 424 liters (95% CI: 351-512). Vorapaxar and the major circulating active metabolite, M20, are extensively bound (≥99%) to human plasma proteins. Vorapaxar is highly bound to human serum albumin and does not preferentially distribute into red blood cells.Metabolism
Vorapaxar is eliminated by metabolism via CYP3A4 and CYP2J2. The major active circulating metabolite is M20 (monohydroxy metabolite) and the predominant metabolite identified in excreta is M19 (amine metabolite). The systemic exposure of M20 is ~20% of the exposure to vorapaxar.Excretion
The primary route of elimination is through the feces. In a 6-week study, 84% of the administered radiolabeled dose was recovered as total radioactivity with 58% collected in feces and 25% in urine. Vorapaxar is eliminated primarily in the form of metabolites, with no unchanged vorapaxar detected in urine.Vorapaxar exhibits multi-exponential disposition with an effective half-life of 3-4 days and an apparent terminal elimination half-life of 8 days. Steady-state is achieved by 21 days following once-daily dosing with an accumulation of 5- to 6-fold. The apparent terminal elimination half-life for vorapaxar is approximately 8 days (range 5-13 days) and is similar for the active metabolite. The terminal elimination half-life is important to determine the time to offset the pharmacodynamic effect [see Clinical Pharmacology (12.2)].
Specific Populations
The effects of intrinsic factors on the pharmacokinetics of vorapaxar are presented in Figure 2 [see Use in Specific Populations (8.5, 8.6, 8.7)].In general, effects on the exposure of vorapaxar based on age, race, gender, weight, and moderate renal insufficiency were modest (20-40%; see Figure 2). No dose adjustments are necessary based upon these factors. Because of the inherent bleeding risks in patients with severe hepatic impairment, ZONTIVITY is not recommended in such patients [see Warnings and Precautions (5.1) and Use in Specific Populations (8.7)].
Figure 2: Effect of Intrinsic Factors on the Pharmacokinetics of Vorapaxar † See Warnings and Precautions. (5.1) and Use in Specific Populations. (8.7) 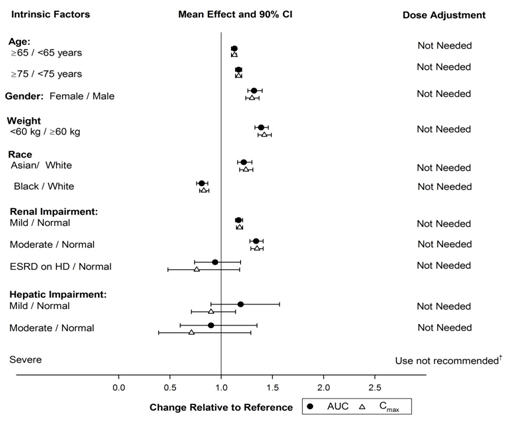
Drug Interactions [see also Drug Interactions (7)]
Anticoagulants and Antiplatelet Agents
An interaction study with vorapaxar and warfarin in healthy subjects did not demonstrate a clinically significant pharmacokinetic or pharmacodynamic interaction [see Warnings and Precautions (5.1) and Figure 4].Vorapaxar did not affect prasugrel pharmacokinetics and prasugrel did not affect vorapaxar pharmacokinetics following multiple-dose administration at steady-state [see Warnings and Precautions (5.1) and Figures 3 and 4]. The pharmacokinetic interaction between vorapaxar and clopidogrel has not been evaluated. However, the use of vorapaxar on a background of clopidogrel is supported by the clinical data from TRA 2°P and TRACER [see Adverse Reactions (6.1) and Clinical Studies (14)].
Effects of Other Drugs on Vorapaxar
The effects of other drugs on the pharmacokinetics of vorapaxar are presented in Figure 3 as change relative to vorapaxar administered alone (test/reference). Phase 3 data suggest that coadministration of a weak or moderate CYP3A inhibitor with vorapaxar does not increase bleeding risk or alter the efficacy of vorapaxar. No dose adjustment for ZONTIVITY is required in patients taking weak to moderate inhibitors of CYP3A.Figure 3: Effects of Other Drugs on the Pharmacokinetics of Vorapaxar † See Warnings and Precautions. (5.2) and Drug Interactions. (7.1) ‡ See Dosage and Administration. (2.2) 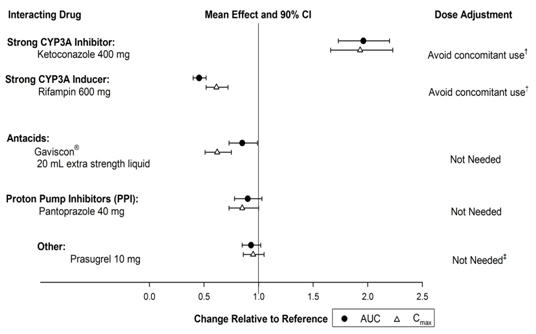
Effects of Vorapaxar on Other Drugs
In vitro metabolism studies demonstrate that vorapaxar or M20 is unlikely to cause clinically significant inhibition or induction of major CYP isoforms or inhibition of OATP1B1, OATP1B3, BCRP, OAT1, OAT3, and OCT2 transporters.Specific in vivo effects on the pharmacokinetics of digoxin, warfarin, rosiglitazone and prasugrel are presented in Figure 4 as a change relative to the interacting drug administered alone (test/reference). Vorapaxar is a weak inhibitor of the intestinal P-glycoprotein (P-gp) transporter. No dosage adjustment of digoxin or ZONTIVITY is required.
Figure 4: Effects of Vorapaxar on the Pharmacokinetics of Other Drugs † See Warnings and Precautions. (5.1) ‡ See Dosage and Administration. (2.2) 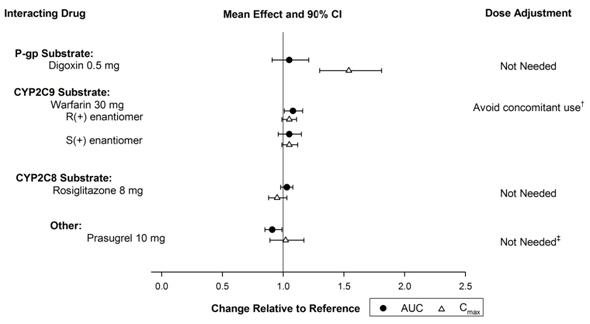
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Carcinogenicity studies were conducted in rats and mice dosed orally with vorapaxar for two years. Male and female rats dosed at 0, 3, 10 or 30 mg/kg/day showed no carcinogenic potential at systemic exposures (AUC) in males and females that were 9- and 29-fold, respectively, the human systemic exposure at the RHD. In male and female mice dosed at 0, 1, 5, and 15 mg/kg/day, vorapaxar showed no carcinogenic potential at systemic exposures (AUC) that were up to 30-fold the human systemic exposure.13.2 Animal Pharmacology
Vorapaxar did not increase bleeding time in non-human primates when administered alone. Bleeding time was prolonged slightly with administration of aspirin or aspirin plus vorapaxar. The combination of aspirin, vorapaxar, and clopidogrel produced significant prolongation of bleeding time. Transfusion of human platelet rich plasma normalized bleeding times with partial recovery of ex vivo platelet aggregation induced with arachidonic acid, but not induced with ADP or TRAP. Platelet poor plasma had no effect on bleeding times or platelet aggregation [see Warnings and Precautions (5.1)].
-
14 CLINICAL STUDIES
The clinical evidence for the effectiveness of ZONTIVITY is supported by TRA 2°P - TIMI 50. TRA 2°P was a multicenter, randomized, double-blind, placebo-controlled study conducted in patients who had evidence or a history of atherosclerosis involving the coronary (spontaneous MI ≥2 weeks but ≤12 months prior), cerebral (ischemic stroke), or peripheral vascular (documented peripheral arterial disease [PAD]) systems. Patients were randomized to receive daily treatment with ZONTIVITY (n=13,225) or placebo (n=13,224) in addition to standard of care. The study's primary endpoint was the composite of cardiovascular death, MI, stroke, and urgent coronary revascularization (UCR). The composite of cardiovascular death, MI, and stroke was assessed as key secondary endpoint. The median follow-up was 2.5 years (up to 4 years).
The findings in all randomized patients for the primary efficacy composite endpoint show a 3-year K-M event rate of 11.2% in the ZONTIVITY group compared to 12.4% in the placebo group (hazard ratio [HR]: 0.88; 95% confidence interval [CI], 0.82 to 0.95; p=0.001).
The findings for the key secondary efficacy endpoint show a 3-year Kaplan-Meier (K-M) event rate of 9.3% in the ZONTIVITY group compared to 10.5% in placebo group (HR 0.87; 95% CI, 0.80 to 0.94; p<0.001).
Although TRA 2°P was not designed to evaluate the relative benefits and risks of ZONTIVITY in individual patient subgroups, patients with a history of stroke or TIA showed an increased risk of ICH. Of the patients who comprised the post-MI and PAD strata and had no baseline history of stroke or TIA,10,080 were randomized to treatment with ZONTIVITY and 10,090 to placebo. These patients were 89% Caucasian, 22% female, and 33% ≥65 years of age, with a median age of 60 years. The population included patients with diabetes (24%) and patients with hypertension (65%). Of the patients who qualified for the trial with MI without a history of stroke or TIA, 98% were receiving aspirin, 78% were receiving a thienopyridine, and 77% were receiving both aspirin and a thienopyridine when they enrolled in the trial. Of the patients who qualified for the trial with PAD without a history of stroke or TIA, 88% were receiving aspirin, 35% were receiving a thienopyridine, and 27% were receiving both aspirin and a thienopyridine when they enrolled.
In post-MI or PAD patients without a history of stroke or TIA the 3-year K-M event rate for the primary efficacy endpoint (composite of time to first CV death, MI, stroke, or UCR) was 10.1% in the ZONTIVITY group compared to 11.8% in the placebo group (HR 0.83; 95% CI, 0.76 to 0.90; p<0.001) (see Figure 5 and Table 3).
The results for the key secondary efficacy endpoint (composite of time to first CV death, MI, or stroke) show a 3-year K-M event rate of 7.9% in the ZONTIVITY group compared to 9.5% in the placebo group (HR 0.80; 95% CI, 0.73 to 0.89; p<0.001) (see Table 3).
The effect of chronic dosing with ZONTIVITY on the primary and key secondary endpoints was maintained for the duration of the trial (median follow up 2.5 years, up to 4 years).
Figure 5: Time to First Occurrence of the Composite Endpoint of CV Death, MI, Stroke or UCR in Post-MI or PAD Patients without a History of Stroke or TIA in TRA 2°P 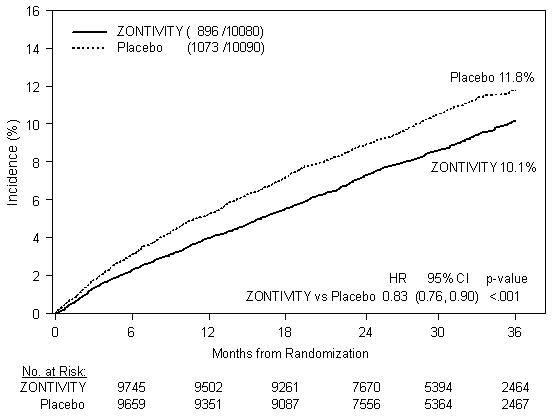
Table 3: TRA 2°P: Time to First Event in Post-MI or PAD Patients without a History of Stroke or TIA Placebo
(n=10,090)ZONTIVITY
(n=10,080)Endpoints Patients with events*
(%)K-M %† Patients with events*
(%)K-M %† Hazard Ratio ‡,§
(95% CI)p-value§ - * Each patient was counted only once (first component event) in the component summary that contributed to the primary efficacy endpoint.
- † K-M estimate at 1,080 days.
- ‡ Hazard ratio is ZONTIVITY group versus placebo group.
- § Cox proportional hazard model with covariates treatment and stratification factors (qualifying atherosclerotic disease and planned thienopyridine use).
- ¶ Including patients who could have had other non-fatal events or subsequently died.
Primary Composite Efficacy Endpoint
(CV death/MI/stroke/UCR)*,§1073 (10.6%) 11.8% 896 (8.9%) 10.1% 0.83 (0.76-0.90) <0.001 Secondary Composite Efficacy Endpoint
(CV death/MI/stroke)*,§851 (8.4%) 9.5% 688 (6.8%) 7.9% 0.80 (0.73-0.89) <0.001 Other Secondary Efficacy Endpoints (first occurrences of specified event at any time) ¶ CV Death 239 (2.4%) 2.8% 205 (2.0%) 2.4% 0.86 (0.71-1.03) MI 569 (5.6%) 6.4% 470 (4.7%) 5.4% 0.82 (0.73-0.93) Stroke 145 (1.4%) 1.6% 98 (1.0%) 1.2% 0.67 (0.52-0.87) UCR 283 (2.8%) 3.0% 249 (2.5%) 2.8% 0.88 (0.74-1.04) In post-MI or PAD patients who survived an on-study efficacy event, the incidence of subsequent events was lower with ZONTIVITY.
The time from the prior MI to randomization had no relationship to the treatment benefit for the primary study outcome.
A range of demographic, concurrent baseline medications, and other treatment differences were examined for their influence on outcomes as shown in Figure 6. Such analyses must be interpreted cautiously, as differences can reflect the play of chance among a large number of analyses.
Figure 6: Subgroup Analyses (Primary Endpoints) of the TRA 2°P Post-MI or PAD Patients without a History of Stroke or TIA 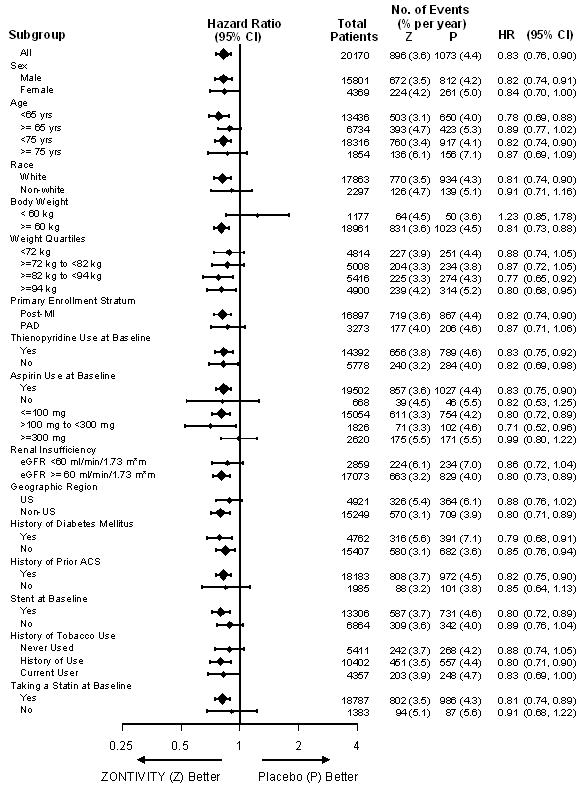
-
16 HOW SUPPLIED/STORAGE AND HANDLING
ZONTIVITY tablets, 2.08 mg vorapaxar, are yellow, oval-shaped, film-coated tablets with "351" on one side and the Merck logo on the other side.
They are supplied as follows:
- NDC: 70347-208-30 bottles of 30 tablets
- NDC: 70347-208-90 bottles of 90 tablets
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved Patient Labeling ((Medication Guide).
Benefits and Risks
- Summarize the benefits and potential side effects of ZONTIVITY.
- Tell patients to take ZONTIVITY exactly as prescribed.
- Inform patients not to discontinue ZONTIVITY without discussing it with the prescribing physician.
- Tell patients to read the Medication Guide.
Bleeding
Inform patients that they:- May bleed and bruise more easily.
- Should report any unanticipated, prolonged or excessive bleeding, or blood in their stool or urine.
Invasive Procedures
Instruct patients to:- Inform physicians and dentists that they are taking ZONTIVITY before any surgery or dental procedure.
- Tell the doctor performing any surgery or dental procedure to talk to the prescribing physician before stopping ZONTIVITY.
Concomitant Medications
Tell patients to list all prescription medications, over-the-counter medications, or dietary supplements they are taking or plan to take so that the physician knows about other treatments that may affect bleeding risk.Lactation
Advise patients that breastfeeding is not recommended during treatment with Zontivity - SPL UNCLASSIFIED SECTION
-
MEDICATION GUIDE
Medication Guide
ZONTIVITY® (zon-TIV-iti)
(vorapaxar)
TabletsRead this Medication Guide before you start taking ZONTIVITY and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment.
What is the most important information I should know about ZONTIVITY?
ZONTIVITY is used to lower your chance of having another serious problem with your heart or blood vessels, but ZONTIVITY (and similar drugs) can cause bleeding that can be serious and lead to death.
Call your doctor right away if you have any of these signs or symptoms of bleeding while taking ZONTIVITY:
- bleeding that is severe or that you cannot control
- pink, red, or brown urine
- vomiting blood or your vomit looks like "coffee grounds"
- red or black stools (looks like tar)
- coughing up blood or blood clots.
While you take ZONTIVITY and for about 4 weeks after your treatment with ZONTIVITY is stopped:
- you may bruise and bleed more easily (nose bleeds may be common)
- it will take longer than usual for any bleeding to stop.
Do not take ZONTIVITY if you:
- have had a stroke or "mini stroke" (also known as transient ischemic attack or TIA)
- have had bleeding in your brain
- currently have unusual bleeding, such as bleeding in your head, stomach or intestines (an ulcer).
If you have a stroke, TIA, or bleeding in your brain while taking ZONTIVITY your doctor should stop your treatment with ZONTIVITY. Follow your doctor's instructions about stopping ZONTIVITY.
Do not stop taking ZONTIVITY without talking to the doctor who prescribed it for you.
What is ZONTIVITY?
ZONTIVITY is a prescription medicine used to treat people who have
- had a heart attack or
- reduced blood flow in their legs (peripheral arterial disease).
ZONTIVITY is used with aspirin and/or clopidogrel to lower your chance of having another serious problem with your heart or blood vessels, such as heart attack, stroke, or death.
It is not known if ZONTIVITY is safe and effective in children.
What should I tell my doctor before taking ZONTIVITY?
Before you take ZONTIVITY, tell your doctor if you:
- have had bleeding problems or history of stomach ulcers
- have had a stroke or "mini-stroke" (also known as transient ischemic attack or TIA)
- have had any recent serious injury or surgery
- plan to have surgery or a dental procedure
- have kidney problems or severe liver problems
- are pregnant or plan to become pregnant. It is not known if ZONTIVITY will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if ZONTIVITY passes into your breast milk. You and your doctor should decide if you will take ZONTIVITY or breastfeed. You should not do both.
Tell all of your doctors and dentists that you are taking ZONTIVITY. They should talk to the doctor who prescribed ZONTIVITY for you before you have any surgery or dental procedure.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, dietary or herbal supplements. Taking ZONTIVITY with certain other medicines may increase your risk of bleeding and may affect how ZONTIVITY works.
Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine.
How should I take ZONTIVITY?
Take ZONTIVITY exactly as prescribed by your doctor.
- Take ZONTIVITY 1 time each day.
- Take ZONTIVITY with or without food.
- Take ZONTIVITY with aspirin and/or clopidogrel as prescribed by your doctor.
- Do not stop taking ZONTIVITY without first talking to the doctor who prescribed it for you.
- If you take too much ZONTIVITY, call your doctor, or go to the nearest emergency room right away.
What are the possible side effects of ZONTIVITY?
- See "What is the most important information I should know about ZONTIVITY?"
- Anemia (low level of red blood cells)
- Depression
- Rash
These are not all the possible side effects of ZONTIVITY.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store ZONTIVITY?
- Store ZONTIVITY at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep ZONTIVITY in the bottle it comes in.
- Keep the bottle tightly closed.
- The ZONTIVITY bottle contains a desiccant packet to help keep your medicine dry (protect it from moisture). Keep the desiccant packet in the bottle. Do not throw away the desiccant packet.
- Store blister packs of ZONTIVITY in the original package it comes in.
Keep ZONTIVITY and all medicines out of the reach of children.
You can ask your doctor or pharmacist for information about ZONTIVITY that is written for health professionals.
For more information, go to www.zontivity.com or call 1-833-694-8235.
What are the ingredients in ZONTIVITY?
Active ingredient: vorapaxar sulfateInactive ingredients:
Tablet: lactose monohydrate, microcrystalline cellulose, croscarmellose sodium, povidone, and magnesium stearateFilm coating: lactose monohydrate, hypromellose, titanium dioxide, triacetin (glycerol triacetate), and iron oxide yellow
This Medication Guide has been approved by the U.S. Food and Drug Administration.
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 30 Tablet Bottle Label
NDC: 70347-208-30
Zontivity®
(vorapaxar) tablets2.08 mg*
Dispense the accompanying
Medication Guide to each patient.*Each tablet contains 2.08 mg
vorapaxar, equivalent to 2.5 mg
vorapaxar sulfate.Rx only
30 Tablets
Aralez Pharmaceuticals

-
INGREDIENTS AND APPEARANCE
ZONTIVITY
vorapaxar tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 70347-208 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength vorapaxar sulfate (UNII: IN66038E6C) (vorapaxar - UNII:ZCE93644N2) vorapaxar 2.08 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) POVIDONE (UNII: FZ989GH94E) MAGNESIUM STEARATE (UNII: 70097M6I30) HYPROMELLOSE 2910 (15 MPA.S) (UNII: 36SFW2JZ0W) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color YELLOW Score no score Shape OVAL Size 9mm Flavor Imprint Code 351 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 70347-208-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2017 2 NDC: 70347-208-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2017 3 NDC: 70347-208-05 5 in 1 BLISTER PACK; Type 0: Not a Combination Product 07/06/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204886 05/08/2014 Labeler - Aralez Pharmaceuticals Us Inc. (046550531) Registrant - ARALEZ PHARMACEUTICALS TRADING DESIGNATED ACTIVITY COMPANY (985620239)
Trademark Results [ZONTIVITY]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ZONTIVITY 86164928 not registered Dead/Abandoned |
Merck Sharp & Dohme Corp. 2014-01-14 |
 ZONTIVITY 85307417 4628973 Live/Registered |
ARALEZ PHARMACEUTICALS TRADING DESIGNATED ACTIVITY COMPANY 2011-04-28 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.