MIDAZOLAM IN SODIUM CHLORIDE- midazolam injection, solution
Midazolam in Sodium Chloride by
Drug Labeling and Warnings
Midazolam in Sodium Chloride by is a Prescription medication manufactured, distributed, or labeled by WG Critical Care, LLC., InfoRLife SA. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use Midazolam Injection safely and effectively. See full prescribing information for Midazolam Injection.
MIDAZOLAM IN SODIUM CHLORIDE injection, for intravenous use, CIV
Initial U.S. Approval: 1985WARNING: PERSONNEL AND EQUIPMENT FOR MONITORING AND RESUSCITATION, AND RISKS FROM CONCOMITANT USE WITH OPIOID ANALGESICS AND OTHER SEDATIVE-HYPNOTICS
See full prescribing information for complete boxed warning.
- Only personnel trained in the administration of procedural sedation, and not involved in the conduct of the diagnostic or therapeutic procedure, should administer Midazolam Injection. (2.1, 5.1)
- Administering personnel must be trained in the detection and management of airway obstruction, hypoventilation, and apnea, including the maintenance of a patent airway, supportive ventilation, and cardiovascular resuscitation. (2.1, 5.1)
- Resuscitative drugs, and age- and size-appropriate equipment for bag/valve/mask assisted ventilation must be immediately available during administration of Midazolam Injection. (2.1, 5.1)
- Continuously monitor vital signs during sedation and through the recovery period. (2.1, 5.1)
- Concomitant use of benzodiazepines with opioid analgesics may result in profound sedation, respiratory depression, coma, and death. Continuously monitor patients for respiratory depression and depth of sedation. (5.2, 7.1)
INDICATIONS AND USAGE
Midazolam in 0.9% Sodium Chloride Injection is a benzodiazepine indicated for:
- continuous intravenous infusion for sedation of intubated and mechanically ventilated adult, pediatric, and neonatal patients as a component of anesthesia or during treatment in a critical care setting. (1)
DOSAGE AND ADMINISTRATION
- For intravenous injection only. Avoid intra-arterial injection or extravasation. (2.1)
- Individualize dosing and titrate to desired clinical response, taking into account patient age, clinical status, and concomitant use of other CNS depressants. (2.1)
- See Full Prescribing Information for complete dosage and administration information. (2)
DOSAGE FORMS AND STRENGTHS
Injection: 50 mg per 50 mL (1mg/mL) and 100 mg per 100 mL (1 mg/mL) in single-dose bags. (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
Cardiorespiratory Adverse Reactions: Serious cardiorespiratory adverse reactions have occurred, sometimes resulting in death or permanent neurologic injury. (5.3)
Paradoxical Behavior: Agitation, involuntary movements (including tonic/clonic movements and muscle tremor), hyperactivity and combativeness have been reported in both adult and pediatric patients. (5.4)
Dependence and Withdrawal with Long-Term Use: Use for several days to weeks may lead to physical dependence to midazolam. Do not abruptly discontinue midazolam. Gradually taper the dosage using a tapering schedule that is individualized to the patient. (5.5)
Debilitation and Comorbid Considerations: Higher risk adult and pediatric surgical patients, elderly patients and debilitated adult and pediatric patients require lower dosages, whether or not concomitant sedating medications have been administered. (5.6)
Risk of Intra-Arterial Injection: There have been limited reports of intra-arterial injection of midazolam. Adverse events have included local reactions, as well as isolated reports of seizure activity in which no clear causal relationship was established. (5.7)
Impaired Cognitive Function: Because of partial or complete impairment of recall, patients should not operate hazardous machinery or a motor vehicle until drug effects have subsided. (5.8)
Hypotension and Seizure in Preterm Infants and Neonates: Avoid rapid injection in the neonatal population. (5.9)
Neonatal Sedation and Withdrawal Syndrome: Receiving Midazolam in 0.9% Sodium Chloride Injection during pregnancy can result in neonatal sedation and/or neonatal withdrawal. (5.10, 8.1)
Pediatric Neurotoxicity: In developing animals, exposures greater than 3 hours cause neurotoxicity. Weigh benefits against potential risks when considering elective procedures in children under 3 years old. (5.11)
ADVERSE REACTIONS
The most common adverse reactions (≥15%) were decreased tidal volume, decreased respiratory rate, and apnea. (6)
To report SUSPECTED ADVERSE REACTIONS, contact WG Critical Care, LLC at 1-866-562-4708 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
Lactation: A lactating woman may pump and discard breast milk for 4 to 8 hours after treatment with midazolam (8.2).
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 5/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Instructions
2.2 General Dosing Information
2.3 Dosing Recommendations
2.4 Safe Discontinuation of Midazolam in 0.9% Sodium Chloride Injection After Long-Term Use
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Personnel and Equipment for Monitoring and Resuscitation
5.2 Risks from Concomitant Use with Opioids, Other Sedative Hypnotics, or Other Central Nervous
System Depressants5.3 Risk of Cardiorespiratory Adverse Reactions
5.4 Risk of Paradoxical Behavior
5.5 Risk of Dependence and Withdrawal with Long-Term Use of Midazolam in 0.9% Sodium Chloride Injection
5.6 Debilitation and Comorbid Considerations
5.7 Risk of Intra-Arterial Injection
5.8 Impaired Cognitive Function
5.9 Risk of Hypotension and Seizure in Preterm Infants and Neonates
5.10 Neonatal Sedation and Withdrawal Syndrome
5.11 Pediatric Neurotoxicity
5.12 Risk of Increased Intraocular Pressure in Patients with Glaucoma
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
7.1 Opioid Analgesics and Other Sedative Hypnotics
7.2 Cytochrome P450-3A4 Inhibitors
7.3 Saquinavir
7.4 Thiopental
7.5 Halothane
7.6 Pancuronium
7.7 Other Drugs Used in the Surgical Setting
7.8 Drug/Laboratory Test Interactions
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
9.2 Abuse
9.3. Dependence
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: PERSONNEL AND EQUIPMENT FOR MONITORING AND RESUSCITATION, AND RISKS FROM CONCOMITANT USE WITH OPIOID ANALGESICS AND OTHER SEDATIVE-HYPNOTICS Personnel and Equipment for Monitoring and ResuscitationOnly personnel trained in the administration of procedural sedation, and not involved in the conduct of the diagnostic or therapeutic procedure, should administer Midazolam in 0.9% Sodium Chloride Injection [see Dosage and Administration (2.1), Warnings and Precautions (5.1)].Administering personnel must be trained in the detection and management of airway obstruction, hypoventilation, and apnea, including the maintenance of a patent airway, supportive ventilation, and cardiovascular resuscitation [see Dosage and Administration (2.1), Warnings and Precautions (5.1)].Resuscitative drugs, and age- and size-appropriate equipment for bag/valve/mask assisted ventilation must be immediately available during administration of Midazolam in 0.9% Sodium Chloride Injection [see Dosage and Administration (2.1), Warnings and Precautions (5.1)].Continuously monitor vital signs during sedation and during the recovery period [see Dosage and Administration (2.1), Warnings and Precautions (5.1)].Risks from Concomitant Use with Opioid Analgesics and Other Sedative HypnoticsConcomitant use of benzodiazepines, including Midazolam in 0.9% Sodium Chloride Injection, and opioids may result in profound sedation, respiratory depression, coma, and death. Continuously monitor patients for respiratory depression and depth of sedation [see Warnings and Precautions (5.2) and Drug Interaction (7.1)].
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Instructions
- Midazolam in 0.9% Sodium Chloride Injection should only be administered intravenously. Avoid intra-arterial injection or extravasation [see Warnings and Precautions (5.7)].
- Only personnel trained in the administration of procedural sedation, and not involved in the conduct of the diagnostic or therapeutic procedure, should administer Midazolam in 0.9% Sodium Chloride Injection.
- Administering personnel must be trained in the detection and management of airway obstruction, hypoventilation, and apnea, including the maintenance of a patent airway, supportive ventilation, and cardiovascular resuscitation.
- Supplemental oxygen, resuscitative drugs, and age‑ and size-appropriate equipment for bag/valve/mask assisted ventilation must be immediately available during administration of Midazolam in 0.9% Sodium Chloride Injection. A benzodiazepine reversal agent should be immediately available.
- Continuously monitor vital signs during sedation and through the recovery period [see Warnings and Precautions (5.1)].
Midazolam must never be used without individualization of dosage particularly when used with other medications capable of producing central nervous system depression [Warnings and Precautions (5.2)].
Midazolam in 0.9% Sodium Chloride Injection can cause respiratory depression. It is a potent sedative agent that requires slow administration and individualization of dosage. Excessive single doses or rapid intravenous administration may result in respiratory depression, airway obstruction and/or arrest. Continuously monitor patients for early signs of hypoventilation, airway obstruction, and apnea using capnography, pulse oximetry, and clinical assessment [see Warnings and Precautions (5.3)].
Reactions such as agitation, involuntary movements, hyperactivity and combativeness have been reported in adult and pediatric patients. Should such reactions occur, the response to each dose of midazolam and all other drugs, including local anesthetics, should be evaluated before proceeding. Reversal of such responses with flumazenil has been reported in pediatric patients [see Warnings and Precautions (5.4)].
Visually inspect parenteral drug products for particulate matter and discoloration prior to administration, whenever solution and container permit. If solution is discolored or particulate matter is present, do not use.
2.2 General Dosing Information
Individualize dosing and titrate to desired clinical response, taking into account patient age, clinical status, and concomitant use of other CNS depressants. Titrate to effect with multiple small doses while continuously monitoring respiratory and cardiac function (i.e., pulse oximetry). To minimize the potential for oversedation, allow adequate time between doses to achieve peak central nervous system effect (3 to 5 minutes).
Adults and Pediatrics:
Sedation guidelines recommend a careful presedation history to determine how a patient's underlying medical conditions or concomitant medications might affect their response to sedation/analgesia as well as a physical examination including a focused examination of the airway for abnormalities. Further recommendations include appropriate presedation fasting.
Pediatrics:
Pediatric patients generally require higher dosages of midazolam (mg/kg) than adults. For deeply sedated pediatric patients a dedicated individual, other than the practitioner performing the procedure, should monitor the patient throughout the procedure.
Elderly and Debilitated Patients
Intravenous doses of midazolam should be decreased for elderly and for debilitated patients [see Warnings and Precautions (5.6)]. These patients will also probably take longer to recover completely after midazolam administration for the induction of anesthesia [see Warnings and Precautions (5.8)].
Monitoring
Patient response to sedative agents, and resultant respiratory status, is variable. Regardless of the intended level of sedation or route of administration, sedation is a continuum; a patient may move easily from light to deep sedation, with potential loss of protective reflexes. This is especially true in pediatric patients. Continuously monitor vital signs during sedation and through the recovery period [see Warnings and Precautions (5.1)].
2.3 Dosing Recommendations
Table 1 provides dosing recommendations for adult, pediatric, and neonatal patients.
Table 1. Dosing Recommendations for Continuous Intravenous Infusion in Adult, Pediatric, and Neonatal Patients
ADULT PATIENTS
If a loading dose is necessary to rapidly initiate sedation, 0.01 mg/kg to 0.05 mg/kg (approximately 0.5 mg to 4 mg for a typical adult) may be given slowly or infused over several minutes. This dose may be repeated at 10 to 15 minute intervals until adequate sedation is achieved. For maintenance of sedation, the usual initial infusion rate is 0.02 mg/kg/hr to 0.10 mg/kg/hr (1 mg/hr to 7 mg/hr). Higher loading or maintenance infusion rates may occasionally be required in some patients. Use the lowest recommended doses in patients with residual effects from anesthetic drugs, or in those concurrently receiving other sedatives or opioids.
Individual response to midazolam is variable. Titrate the infusion rate to the desired level of sedation, taking into account the patient's age, clinical status and current medications. In general, midazolam should be infused at the lowest rate that produces the desired level of sedation. Assess sedation at regular intervals and adjust the midazolam infusion rate. Finding the minimum effective infusion rate decreases the potential accumulation of midazolam and provides for the most rapid recovery once the infusion is terminated.
PEDIATRIC PATIENTS
UNLIKE ADULT PATIENTS, PEDIATRIC PATIENTS GENERALLY RECEIVE INCREMENTS OF MIDAZOLAM ON A MG/KG BASIS. As a group, pediatric patients generally require higher dosages of midazolam (mg/kg) than adults. Younger (less than six years) pediatric patients may require higher dosages (mg/kg) than older pediatric patients. In obese pediatric patients, calculate the dose based on ideal body weight.
Titrate the dose to the desired level of sedation. Assess for desired level of sedation and vital signs at regular intervals.
PRETERM AND TERM NEONATAL PATIENTS
Based on pharmacokinetic parameters and reported clinical experience in preterm and term neonates WHOSE TRACHEA WAS INTUBATED, initiate continuous intravenous infusions of Midazolam in Sodium Chloride Injection at a rate of 0.03 mg/kg/hr (0.5 mcg/kg/min) in neonates <32 weeks and 0.06 mg/kg/hr (1 mcg/kg/min) in neonates >32 weeks. Intravenous loading doses should not be used in neonates, rather the infusion may be run more rapidly for the first several hours to establish therapeutic plasma levels. Frequently assess the rate of infusion, particularly after the first 24 hours so as to administer the lowest possible effective dose and reduce the potential for drug accumulation. Hypotension may be observed in patients who are critically ill and in preterm and term infants, particularly those receiving fentanyl and/or when midazolam is administered rapidly.
When sedating preterm and former preterm neonates WHOSE TRACHEA WAS NOT INTUBATED, monitor respiratory parameters due to an increased risk of apnea.
2.4 Safe Discontinuation of Midazolam in 0.9% Sodium Chloride Injection After Long-Term Use
If Midazolam in 0.9% Sodium Chloride Injection is administered long-term (for several days to weeks), do not abruptly discontinue. Gradually taper the dosage in physically-dependent patients using a tapering schedule that is individualized to the patient. (see Warnings and Precautions (5.5).
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Personnel and Equipment for Monitoring and Resuscitation
- Prior to the intravenous administration of midazolam in any dose, ensure the immediate availability of oxygen, resuscitative drugs, age- and size-appropriate equipment for bag/valve/mask ventilation and intubation, and skilled personnel for the maintenance of a patent airway and support of ventilation.
- Only personnel trained in the administration of procedural sedation, and not involved in the conduct of the diagnostic or therapeutic procedure, should administer Midazolam in 0.9% Sodium Chloride Injection.
- Administering personnel must be trained in the detection and management of airway obstruction, hypoventilation, and apnea, including the maintenance of a patent airway, supportive ventilation, and cardiovascular resuscitation.
- Continuously monitor patients for early signs of hypoventilation, airway obstruction, or apnea, with means readily available (e.g., pulse oximetry). Hypoventilation, airway obstruction, and apnea can lead to hypoxia and/or cardiac arrest unless effective countermeasures are taken immediately.
- A benzodiazepine reversal agent (i.e., flumazenil) should be immediately available during administration of Midazolam in 0.9% Sodium Chloride Injection.
- Continuously monitor vital signs during the recovery period. Because intravenous midazolam can depress respiration [see Clinical Pharmacology (12)], especially when used concomitantly with opioid agonists and other sedatives [see Dosage and Administration (2)], it should be used for sedation/anxiolysis/amnesia only in the presence of personnel skilled in early detection of hypoventilation, maintaining a patent airway, and supporting ventilation.
5.2 Risks from Concomitant Use with Opioids, Other Sedative Hypnotics, or Other Central Nervous
System DepressantsConcomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death. Monitor patients for respiratory depression and sedation [see Warnings and Precautions (5.2) and Drug Interactions (7.1)].
Titrate the dose of Midazolam in 0.9% Sodium Chloride Injection when administered with opioid analgesics and sedative-hypnotics to the desired clinical response.
Continuously monitor sedated patients for hypotension, airway obstruction, hypoventilation, apnea, and oxygen desaturation. These cardiorespiratory effects may be more likely to occur in patients with obstructive sleep apnea, the elderly, and ASA‑PS III or IV patients.
Concomitant use of barbiturates, alcohol, or other central nervous system depressants may increase the risk of hypoventilation, airway obstruction, desaturation, or apnea and may contribute to profound and/or prolonged drug effect. Narcotic premedication also depresses the ventilatory response to carbon dioxide stimulation.
5.3 Risk of Cardiorespiratory Adverse Reactions
Serious cardiorespiratory adverse reactions have occurred after administration of midazolam. These have included respiratory depression, airway obstruction, oxygen desaturation, apnea, respiratory arrest and/or cardiac arrest, sometimes resulting in death or permanent neurologic injury. There have also been rare reports of hypotensive episodes requiring treatment during or after diagnostic or surgical manipulations particularly in adult or pediatric patients with hemodynamic instability. Hypotension occurred more frequently in the sedation studies in patients premedicated with an opioid. Excessive single doses or rapid intravenous administration may result in respiratory depression, airway obstruction and/or arrest. When used for sedation/anxiolysis/amnesia, midazolam should always be titrated slowly in adult or pediatric patients. Adverse hemodynamic events have been reported in pediatric patients with cardiovascular instability; rapid intravenous administration should also be avoided in this population. Continuously monitor patients for early signs of hypoventilation, airway obstruction, and apnea using capnography, pulse oximetry, and clinical assessment [see Dosage and Administration (2.2)].
5.4 Risk of Paradoxical Behavior
Reactions such as agitation, involuntary movements (including tonic/clonic movements and muscle tremor), hyperactivity and combativeness have been reported in both adult and pediatric patients. These reactions may be due to inadequate or excessive dosing or improper administration of midazolam; however, consideration should be given to the possibility of cerebral hypoxia or true paradoxical reactions. Should such reactions occur, the response to each dose of midazolam and all other drugs, including local anesthetics, should be evaluated before proceeding. Reversal of such responses with flumazenil has been reported in pediatric patients.
5.5 Risk of Dependence and Withdrawal with Long-Term Use of Midazolam in 0.9% Sodium Chloride Injection
The continued use of benzodiazepines for several days to weeks may lead to clinically significant physical dependence. If used for long-term use (i.e., for several days to weeks), abrupt discontinuation or rapid dosage reduction of midazolam, or administration of flumazenil, a benzodiazepine antagonist, may precipitate acute withdrawal reactions, including seizures, which can be life-threatening.
Patients at an increased risk of withdrawal adverse reactions after benzodiazepine discontinuation or rapid dosage reduction include those who take higher dosages (i.e., higher and/or more frequent doses) and those who have had longer durations of use.
After extended therapy, do not abruptly discontinue Midazolam in 0.9% Sodium Chloride Injection. When discontinuing midazolam in a physically-dependent patient, gradually taper the dosage using a tapering schedule that is individualized to the patient [see Dosage and Administration (2.3), Dependence (9.3)].
5.6 Debilitation and Comorbid Considerations
Higher risk adult and pediatric surgical patients, elderly patients and debilitated adult and pediatric patients require lower dosages, whether or not concomitant sedating medications have been administered. Adult or pediatric patients with COPD are unusually sensitive to the respiratory depressant effect of midazolam. Pediatric and adult patients undergoing procedures involving the upper airway such as upper endoscopy or dental care, are particularly vulnerable to episodes of desaturation and hypoventilation due to partial airway obstruction. Adult and pediatric patients with chronic renal failure and patients with congestive heart failure eliminate midazolam more slowly [see Clinical Pharmacology (12.3)]. Because elderly patients frequently have inefficient function of one or more organ systems and because dosage requirements have been shown to decrease with age, reduced initial dosage of midazolam is recommended, and the possibility of profound and/or prolonged effect should be considered.
Do not administer Midazolam in 0.9% Sodium Chloride Injection to adult or pediatric patients in shock or coma, or in acute alcohol intoxication with depression of vital signs. Particular care should be exercised in the use of intravenous midazolam in adult or pediatric patients with uncompensated acute illnesses, such as severe fluid or electrolyte disturbances.
5.7 Risk of Intra-Arterial Injection
There have been limited reports of intra-arterial injection of midazolam. Adverse events have included local reactions, as well as isolated reports of seizure activity in which no clear causal relationship was established. Precautions against unintended intra-arterial injection should be taken. Extravasation should also be avoided.
The safety and efficacy of Midazolam in 0.9% Sodium Chloride Injection following nonintravenous routes of administration have not been established. Midazolam in 0.9% Sodium Chloride Injection should only be administered intravenously.
5.8 Impaired Cognitive Function
Midazolam is associated with a high incidence of partial or complete impairment of recall for the next several hours. The decision as to when patients who have received injectable midazolam, particularly on an outpatient basis, may again engage in activities requiring complete mental alertness, operate hazardous machinery or drive a motor vehicle must be individualized. Gross tests of recovery from the effects of midazolam [see Clinical Pharmacology (12.3)] cannot be relied upon to predict reaction time under stress. It is recommended that no patient operate hazardous machinery or a motor vehicle until the effects of the drug, such as drowsiness, have subsided or until one full day after anesthesia and surgery, whichever is longer. For pediatric patients, particular care should be taken to assure safe ambulation.
5.9 Risk of Hypotension and Seizure in Preterm Infants and Neonates
Rapid injection should be avoided in the neonatal population. Midazolam administered rapidly as an intravenous injection (i.e., less than 2 minutes) has been associated with severe hypotension in neonates, particularly when the patient has also received fentanyl. Likewise, severe hypotension has been observed in neonates receiving a continuous infusion of midazolam who then receive a rapid intravenous injection of fentanyl. Seizures have been reported in several neonates following rapid intravenous administration.
The neonate also has reduced and/or immature organ function and is also vulnerable to profound and/or prolonged respiratory effects of midazolam.
5.10 Neonatal Sedation and Withdrawal Syndrome
Receiving Midazolam in 0.9% Sodium Chloride Injection late in pregnancy can result in sedation (respiratory depression, lethargy, hypotonia) and/or withdrawal symptoms (hyperreflexia, irritability, restlessness, tremors, inconsolable crying, and feeding difficulties) in the neonate. Monitor neonates exposed to Midazolam in 0.9% Sodium Chloride Injection during pregnancy or labor for signs of sedation and manage these neonates accordingly [see Use in Specific Populations (8.1)].
5.11 Pediatric Neurotoxicity
Published animal studies demonstrate that the administration of anesthetic and sedation drugs that block NMDA receptors and/or potentiate GABA activity increase neuronal apoptosis in the developing brain and result in long-term cognitive deficits when used for longer than 3 hours. The clinical significance of these findings is not clear. However, based on the available data, the window of vulnerability to these changes is believed to correlate with exposures in the third trimester of gestation through the first several months of life, but may extend out to approximately three years of age in humans [see Nonclinical Pharmacology (13.2)].
Some published studies in children suggest that similar deficits may occur after repeated or prolonged exposures to anesthetic agents early in life and may result in adverse cognitive or behavioral effects. These studies have substantial limitations, and it is not clear if the observed effects are due to the anesthetic/sedation drug administration or other factors such as the surgery or underlying illness.
Anesthetic and sedation drugs are a necessary part of the care of children needing surgery, other procedures, or tests that cannot be delayed, and no specific medications have been shown to be safer than any other. Decisions regarding the timing of any elective procedures requiring anesthesia should take into consideration the benefits of the procedure weighed against the potential risks.
5.12 Risk of Increased Intraocular Pressure in Patients with Glaucoma
Benzodiazepines, including Midazolam in 0.9% Sodium Chloride Injection, can increase intraocular pressure in patients with glaucoma. Measurements of intraocular pressure in patients without eye disease show a moderate lowering following induction with midazolam. Midazolam in 0.9% Sodium Chloride Injection may be used in patients with open-angle glaucoma only if they are receiving appropriate therapy. Patients with open-angle glaucoma may need to have their ophthalmologic status evaluated following treatment with Midazolam in 0.9% Sodium Chloride Injection. Midazolam in 0.9% Sodium Chloride Injection is contraindicated in patients with narrow-angle glaucoma.
-
6 ADVERSE REACTIONS
The following serious adverse reactions are discussed in greater detail in other sections:
- Cardiorespiratory Adverse Reactions [see Warnings and Precautions (5.3)]
- Paradoxical Behavior [see Warnings and Precautions (5.4)]
- Dependence and Withdrawal [see Warnings and Precautions (5.5)]
- Impaired Cognitive Function [see Warnings and Precautions (5.8)]
- Hypotension and Seizure in Preterm Infants and Neonates [see Warnings and Precautions (5.9)]
- Neonatal Sedation and Withdrawal Syndrome [see Warnings and Precautions (5.10), Use in Specific Populations (8.1)]
- Pediatric Neurotoxicity [see Warnings and Precautions (5.11)]
The following adverse reactions have been identified from literature or postmarketing reports of midazolam. Because some of these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Fluctuations in vital signs were the most frequently seen findings following parenteral administration of midazolam in adults and included decreased tidal volume and/or respiratory rate decrease (23.3% of patients following intravenous administration) and apnea (15.4% of patients following intravenous administration), as well as variations in blood pressure and pulse rate. The majority of serious adverse reactions, particularly those associated with oxygenation and ventilation, have been reported when midazolam is administered with other medications capable of depressing the central nervous system. The incidence of such events is higher in patients undergoing procedures involving the airway without the protective effect of an endotracheal tube, (e.g., upper endoscopy and dental procedures).
Adults
Table 2: Additional Adverse Reactions Reported Subsequent to Intravenous Administration as a Single Sedative/anxiolytic/amnestic Agent in Adult Patients:
hiccoughs (3.9%)
Local effects at the intravenous site
nausea (2.8%)
tenderness (5.6%)
vomiting (2.6%)
pain during injection (5.0%)
coughing (1.3%)
redness (2.6%)
"oversedation" (1.6%)
induration (1.7%)
headache (1.5%)
phlebitis (0.4%)
drowsiness (1.2%)
Pediatric Patients
The following adverse events related to the use of intravenous midazolam in pediatric patients were reported in the medical literature: desaturation 4.6%, apnea 2.8%, hypotension 2.7%, paradoxical reactions 2.0%, hiccough 1.2%, seizure-like activity 1.1% and nystagmus 1.1%. The majority of airway-related events occurred in patients receiving other CNS depressing medications and in patients where midazolam was not used as a single sedating agent.
Neonates
There have been reports of hypotensive episodes and seizures following the administration of midazolam to neonates, [see Warnings and Precautions (5.9)].
Other Adverse Reactions Occurring at an Incidence of <1.0% Following Intravenous Injection as a Single Sedative/Anxiolytic/Amnesia Agent
Respiratory: Laryngospasm, bronchospasm, dyspnea, hyperventilation, wheezing, shallow respirations, airway obstruction, tachypnea
Cardiovascular: Bigeminy, premature ventricular contractions, vasovagal episode, bradycardia, tachycardia, nodal rhythm
Gastrointestinal: Acid taste, excessive salivation, retching
CNS/Neuromuscular: Retrograde amnesia, euphoria, hallucination, confusion, argumentativeness, nervousness, anxiety, grogginess, restlessness, emergence delirium or agitation, prolonged emergence from anesthesia, dreaming during emergence, sleep disturbance, insomnia, nightmares, athetoid movements, seizure-like activity, ataxia, dizziness, dysphoria, slurred speech, dysphonia, paresthesia
Special Senses: Blurred vision, diplopia, nystagmus, pinpoint pupils, cyclic movements of eyelids, visual disturbance, difficulty focusing eyes, ears blocked, loss of balance, light-headedness
Integumentary: Hive-like elevation at injection site, swelling or feeling of burning, warmth or coldness at injection site
Hypersensitivity: Allergic reactions including anaphylactic reactions, hives, rash, pruritus
Miscellaneous: Yawning, lethargy, chills, weakness, toothache, faint feeling, hematoma
-
7 DRUG INTERACTIONS
7.1 Opioid Analgesics and Other Sedative Hypnotics
The concomitant use of benzodiazepines and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration. Benzodiazepines interact at GABAA sites and opioids interact primarily at mu receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid-related respiratory depression exists. Monitor patients closely for respiratory depression and sedation.
The sedative effect of intravenous midazolam is accentuated by any concomitantly administered medication which depresses the central nervous system, particularly opioids (e.g., morphine, meperidine and fentanyl) and also secobarbital and droperidol. Consequently, the dosage of midazolam should be adjusted according to the type and amount of concomitant medications administered and the desired clinical response [see Dosage and Administration (2)].
7.2 Cytochrome P450-3A4 Inhibitors
Concomitant administration with drugs that are known to inhibit the P450-3A4 enzyme system, such as cimetidine (not ranitidine), erythromycin, diltiazem, verapamil, ketoconazole and itraconazole, may result in prolonged sedation due to a decrease in plasma clearance of midazolam.
The effect of single oral doses of 800 mg cimetidine and 300 mg ranitidine on steady-state concentrations of midazolam was examined in a randomized crossover study (n=8). Cimetidine increased the mean midazolam steady-state concentration from 57 to 71 ng/mL. Ranitidine increased the mean steady-state concentration to 62 ng/mL. No change in choice reaction time or sedation index was detected after dosing with the H2 receptor antagonists.
In a placebo-controlled study, erythromycin administered as a 500 mg dose, three times a day, for 1 week (n=6), reduced the clearance of midazolam following a single 0.5 mg/kg intravenous dose. The half-life was approximately doubled.
The effects of diltiazem (60 mg three times a day) and verapamil (80 mg three times a day) on the pharmacokinetics and pharmacodynamics of oral midazolam were investigated in a three-way crossover study (n=9). The half-life of midazolam increased from 5 to 7 hours when midazolam was taken in conjunction with verapamil or diltiazem. No interaction was observed in healthy subjects between midazolam and nifedipine.
7.3 Saquinavir
In a placebo-controlled study, saquinavir administered as a 1200 mg dose, tid, for 5 days (n=12), a 56% reduction in the clearance of midazolam following a single 0.05 mg/kg intravenous dose was observed. The half-life was approximately doubled.
7.4 Thiopental
A moderate reduction in induction dosage requirements of thiopental (about 15%) has been noted following use of intramuscular midazolam for premedication in adults.
7.5 Halothane
The intravenous administration of midazolam decreases the minimum alveolar concentration (MAC) of halothane required for general anesthesia. This decrease correlates with the dose of midazolam administered; no similar studies have been carried out in pediatric patients but there is no scientific reason to expect that pediatric patients would respond differently than adults.
7.6 Pancuronium
Although the possibility of minor interactive effects has not been fully studied, midazolam and pancuronium have been used together in patients without noting clinically significant changes in dosage, onset or duration in adults. Midazolam does not protect against the characteristic circulatory changes noted after administration of succinylcholine or pancuronium and does not protect against the increased intracranial pressure noted following administration of succinylcholine. Midazolam does not cause a clinically significant change in dosage, onset or duration of a single intubating dose of succinylcholine; no similar studies have been carried out in pediatric patients but there is no scientific reason to expect that pediatric patients would respond differently than adults.
7.7 Other Drugs Used in the Surgical Setting
No significant adverse interactions with commonly used premedications or drugs used during anesthesia and surgery (including atropine, scopolamine, glycopyrrolate, diazepam, hydroxyzine, d-tubocurarine, succinylcholine and other nondepolarizing muscle relaxants) or topical local anesthetics (including lidocaine, dyclonine HCl and Cetacaine) have been observed in adults or pediatric patients. In neonates, however, severe hypotension has been reported with concomitant administration of fentanyl. This effect has been observed in neonates on an infusion of midazolam who received a rapid injection of fentanyl and in patients on an infusion of fentanyl who have received a rapid injection of midazolam.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Neonates born to mothers using benzodiazepines, including midazolam, late in pregnancy have been reported to experience symptoms of sedation and/or neonatal withdrawal [see Warnings and Precautions (5.10), and Clinical Considerations]. Available data from published observational studies of pregnant women exposed to benzodiazepines do not report a clear association with benzodiazepines and major birth defects (see Data).
Available data from randomized controlled trials, cohort studies and case reports over several decades with midazolam use in pregnant women for anesthesia have not identified a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Most of the reported exposures to midazolam occurred at the time of cesarean delivery. Rare case reports of the prolonged use of midazolam in pregnant women for sedation in a critical care setting are insufficient to evaluate for a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes (see Data).
In pregnant rats and rabbits, midazolam did not cause adverse effects to the fetus at doses of up to 1.85 times the human induction dose of 0.35 mg/kg based on body surface area comparisons.
Published studies in pregnant primates demonstrate that the administration of anesthetic and sedation drugs that block NMDA receptors and/or potentiate GABA activity during the period of peak brain development increases neuronal apoptosis in the developing brain of the offspring when used for longer than 3 hours. There are no data on pregnancy exposures in primates corresponding to periods prior to the third trimester in humans (see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Benzodiazepines cross the placenta and may produce respiratory depression, hypotonia, and sedation in neonates. Monitor neonates exposed to midazolam during pregnancy or labor for signs of sedation, respiratory depression, hypotonia, and feeding problems. Monitor neonates exposed to midazolam during pregnancy for signs of withdrawal. Manage these neonates accordingly [see Warnings and Precautions (5.2)].
Data
Human Data
Published data from observational studies on the use of benzodiazepines during pregnancy do not report a clear association with benzodiazepines and major birth defects. Although early studies reported an increased risk of congenital malformations with diazepam and chlordiazepoxide, there was no consistent pattern noted. In addition, the majority of more recent case-control and cohort studies of benzodiazepine use during pregnancy, which were adjusted for confounding exposures to alcohol, tobacco and other medications, have not confirmed these findings.
Animal Data
Pregnant rats were treated with midazolam using intravenous doses of 0.2, 1, and 4 mg/kg/day (0.09, 0.46, and 1.85 times the human induction dose of 0.35 mg/kg based on body surface area comparisons) during the period of organogenesis (Gestation Day 7 through 15). Midazolam did not cause adverse effects to the fetus at doses of up to 1.85 times the human induction dose. All doses produced slight to moderate ataxia. The high dose produced a 5% decrease in maternal body weight gain compared to control.
Pregnant rabbits were treated with midazolam using intravenous doses of 0.2, 0.6, and 2 mg/kg/day (0.09, 0.46, and 1.85 times the human induction dose of 0.35 mg/kg based on body surface area comparisons) during the period of organogenesis (Gestation Day 7 to 18). Midazolam did not cause adverse effects to the fetus at doses of up to 1.85 times the human induction dose. The high dose was associated with findings of ataxia and sedation but no evidence of maternal toxicity.
Pregnant rats were administered midazolam using intravenous doses of 0.2, 1, and 4 mg/kg/day (0.09, 0.46, and 1.85 times the human induction dose of 0.35 mg/kg based on body surface area comparisons) during late gestation and through lactation (Gestation Day 15 through Lactation Day 21). All doses produced ataxia. The high dose produced a slight decrease in maternal body weight gain compared to control. There were no clear adverse effects noted in the offspring. The study included no functional assessments of the pups, such as learning and memory testing or reproductive capacity.
In a published study in primates, administration of an anesthetic dose of ketamine for 24 hours on Gestation Day 122 increased neuronal apoptosis in the developing brain of the fetus. In other published studies, administration of either isoflurane or propofol for 5 hours on Gestation Day 120 resulted in increased neuronal and oligodendrocyte apoptosis in the developing brain of the offspring. With respect to brain development, this time period corresponds to the third trimester of gestation in the human. The clinical significance of these findings is not clear; however, studies in juvenile animals suggest neuroapoptosis correlates with long-term cognitive deficits [see Warnings and Precautions (5.8), Use in Specific Populations (8.4), Nonclinical Pharmacology (13.2)].
8.2 Lactation
Risk Summary
There are reports of sedation, poor feeding, and poor weight gain in infants exposed to benzodiazepines through breast milk. Based on data from published studies, midazolam is present in human milk in low levels (see Data). There are no data on the effects of midazolam on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Midazolam in 0.9% Sodium Chloride Injection and any potential adverse effects on the breastfed infant from Midazolam in 0.9% Sodium Chloride Injection or from the underlying maternal condition.
Clinical Considerations
Infants exposed to midazolam through breast milk should be monitored for sedation, poor feeding, and poor weight gain. A lactating woman may consider interrupting breastfeeding and pumping and discarding breast milk during treatment for a range of at least 4 to 8 hours after midazolam administration in order to minimize drug exposure to a breastfed infant.
Data
Published clinical lactation studies describe the presence of midazolam in human milk at low levels 4 to 8 hours after midazolam administration. These lactation studies have limitations including poor methodology and lack of validated analytical methods. Published study guidelines recommend pumping and discarding breast milk for a range of at least 4 to 8 hours after treatment with midazolam. No safety signals have been identified in breastfed infants exposed to midazolam.
8.4 Pediatric Use
The safety and efficacy of midazolam for sedation/anxiolysis/amnesia following continuous infusion have been established in pediatric and neonatal patients. UNLIKE ADULT PATIENTS, PEDIATRIC PATIENTS GENERALLY RECEIVE INCREMENTS OF MIDAZOLAM ON A MG/KG BASIS. As a group, pediatric patients generally require higher dosages of midazolam (mg/kg) than do adults. Younger (less than six years) pediatric patients may require higher dosages (mg/kg) than older pediatric patients, and may require closer monitoring. In obese PEDIATRIC PATIENTS, the dose should be calculated based on ideal body weight. When midazolam is given in conjunction with opioids or other sedatives, the potential for respiratory depression, airway obstruction, or hypoventilation is increased. The health care practitioner who uses this medication in pediatric patients should be aware of and follow accepted professional guidelines for pediatric sedation appropriate to their situation.
Midazolam should not be administered by rapid injection in the neonatal population. Severe hypotension and seizures have been reported following rapid intravenous administration, particularly, with concomitant use of fentanyl.
Animal Data
Published juvenile animal studies demonstrate that the administration of anesthetic and sedation drugs, such as Midazolam in 0.9% Sodium Chloride Injection, that either block NMDA receptors or potentiate the activity of GABA during the period of rapid brain growth or synaptogenesis, results in widespread neuronal and oligodendrocyte cell loss in the developing brain and alterations in synaptic morphology and neurogenesis. Based on comparisons across species, the window of vulnerability to these changes is believed to correlate with exposures in the third trimester of gestation through the first several months of life, but may extend out to approximately 3 years of age in humans.
In primates, exposure to 3 hours of ketamine that produced a light surgical plane of anesthesia did not increase neuronal cell loss, however, treatment regimens of 5 hours or longer of isoflurane increased neuronal cell loss. Data from isoflurane-treated rodents and ketamine-treated primates suggest that the neuronal and oligodendrocyte cell losses are associated with prolonged cognitive deficits in learning and memory. The clinical significance of these nonclinical findings is not known, and healthcare providers should balance the benefits of appropriate anesthesia in pregnant women, neonates, and young children who require procedures with the potential risks suggested by the nonclinical data [see Warnings and Precautions (5.8) and Nonclinical Pharmacology (13.2)].
8.5 Geriatric Use
Because geriatric patients may have altered drug distribution and diminished hepatic and/or renal function, reduced doses of midazolam are recommended. Doses of Midazolam in 0.9% Sodium Chloride Injection should be decreased for elderly and for debilitated patients [see Warnings and Precautions (5.6) and Dosage and Administration (2)] and subjects over 70 years of age may be particularly sensitive. These patients will also probably take longer to recover completely after midazolam administration for the induction of anesthesia. Administration of intravenous midazolam to elderly and/or high-risk surgical patients has been associated with rare reports of death under circumstances compatible with cardiorespiratory depression. In most of these cases, the patients also received other central nervous system depressants capable of depressing respiration, especially opioids [see Dosage and Administration (2)].
Midazolam is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
-
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
Midazolam in 0.9% Sodium Chloride Injection contains midazolam, a Schedule IV controlled substance.
9.2 Abuse
Midazolam in 0.9% Sodium Chloride Injection contains the benzodiazepine, midazolam. Benzodiazepines are a class of sedative drugs with a known potential for abuse. Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects.
Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a health care provider or for whom it was not prescribed. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence. Both abuse and misuse may lead to addiction. Midazolam was actively self-administered in primate models used to assess the positive reinforcing effects of psychoactive drugs. Midazolam produced physical dependence of a mild to moderate intensity in cynomolgus monkeys after 5 to 10 weeks of administration. Available data concerning the drug abuse and dependence potential of midazolam suggest that its abuse potential is at least equivalent to that of diazepam.
9.3. Dependence
Midazolam may produce physical dependence after long-term use. Physical dependence is a state that develops as a result of physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug. If Midazolam in 0.9% Sodium Chloride Injection is administered long-term (i.e., for several days to weeks), abrupt discontinuation or rapid dosage reduction, or administration of flumazenil, a benzodiazepine antagonist, may precipitate acute withdrawal reactions, including seizures, which can be life-threatening. Patients at an increased risk of withdrawal adverse reactions after benzodiazepine discontinuation or rapid dosage reduction include those who take higher dosages (i.e., higher and/or more frequent doses) and those who have had longer durations of use [see Warnings and Precautions (5.5)].
To reduce the risk of withdrawal reactions, after extended therapy, do not abruptly discontinue Midazolam in 0.9% Sodium Chloride Injection. Gradually taper the dosage using a tapering schedule that is individualized to the patient.
Acute Withdrawal Signs and Symptoms
Acute withdrawal signs and symptoms have included abnormal involuntary movements, anxiety, blurred vision, cognitive disorder, depersonalization, depression, derealization, dizziness, fatigue, gastrointestinal adverse reactions (e.g., nausea, vomiting, diarrhea, weight loss, decreased appetite), headache, hyperacusis, hypertension, irritability, insomnia, memory impairment, muscle pain and stiffness, panic attacks, photophobia, restlessness, tachycardia, and tremor. More severe acute withdrawal signs and symptoms, including life-threatening reactions, have included catatonia, convulsions, delirium tremens, depression, hallucinations, homicidal thoughts, mania, psychosis, and suicidality
Protracted Withdrawal Syndrome
Protracted withdrawal syndrome is characterized by anxiety, cognitive impairment, depression, insomnia, formication, motor symptoms (e.g., weakness, tremor, muscle twitches), paresthesia, and tinnitus that persists beyond 4 to 6 weeks after initial benzodiazepine withdrawal. Protracted withdrawal symptoms may last weeks to more than 12 months.
Tolerance
Midazolam may produce tolerance after long-term use. Tolerance is a physiological state characterized by a reduced response to a drug after repeated administration (i.e., a higher dose of a drug is required to produce the same effect that was once obtained at a lower dose). Tolerance may develop within days or weeks of the therapeutic effects of Midazolam; however, little tolerance develops to the amnestic reactions and other cognitive impairments caused by benzodiazepines.
-
10 OVERDOSAGE
Clinical Presentation
Overdosage of benzodiazepines is characterized by central nervous system depression ranging from drowsiness to coma. In mild to moderate cases, symptoms can include drowsiness, confusion, dysarthria, lethargy, hypnotic state, diminished reflexes, ataxia, and hypotonia. Rarely, paradoxical or disinhibitory reactions (including agitation, irritability, impulsivity, violent behavior, confusion, restlessness, excitement, and talkativeness) may occur. In severe overdosage cases, patients may develop respiratory depression and coma. Overdosage of benzodiazepines in combination with other CNS depressants (including alcohol and opioids) may be fatal [see Warnings and Precautions (5.2)]. Markedly abnormal (lowered or elevated) blood pressure, heart rate, or respiratory rate raise the concern that additional drugs and/or alcohol are involved in the overdosage. No evidence of specific organ toxicity from midazolam overdosage has been reported.
Management of Overdosage
In managing benzodiazepine overdosage, employ general supportive measures, including intravenous fluids and airway management. Flumazenil, a specific benzodiazepine receptor antagonist indicated for the complete or partial reversal of the sedative effects of benzodiazepines in the management of benzodiazepine overdosage, can lead to withdrawal and adverse reactions, including seizures, particularly in the context of mixed overdosage with drugs that increase seizure risk (e.g., tricyclic and tetracyclic antidepressants) and in patients with long-term benzodiazepine use and physical dependency. The risk of withdrawal seizures with flumazenil use may be increased in patients with epilepsy. Flumazenil is contraindicated in patients who have received a benzodiazepine for control of a potentially life-threatening condition (e.g., status epilepticus). If the decision is made to use flumazenil, it should be used as an adjunct to, not as a substitute for, supportive management of benzodiazepine overdosage. See the flumazenil injection Prescribing Information.
Consider contacting the Poison Help Line (1-800-222-1222) or medical toxicologist for additional overdosage management for recommendations.
-
11 DESCRIPTION
Midazolam in 0.9% Sodium Chloride Injection is a benzodiazepine available as a sterile, preservative-free, nonpyrogenic solution of midazolam and sodium chloride in water for injection for intravenous use. Each single-dose bag of Midazolam in 0.9% Sodium Chloride Injection contains either 50 mg/50 mL (1 mg/mL) or 100 mg/100 mL (1 mg/mL) of midazolam and 9 mg/mL of sodium chloride in water for injection. Midazolam in Sodium Chloride Injection may contain hydrochloric acid and/or sodium hydroxide for pH adjustment. The pH is approximately 2.5 - 3.5.
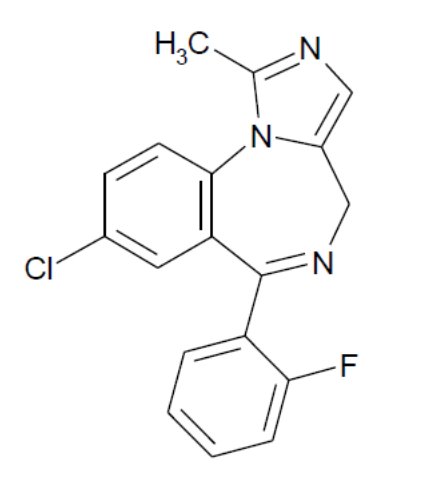
Midazolam is a white to light yellow crystalline compound, insoluble in water, freely soluble in ethanol, soluble in methanol. Chemically, midazolam is 8-chloro-6-(2-fluorophenyl)-1-methyl-4H-imidazo[1,5-a][1,4]benzodiazepine. Midazolam has the empirical formula C18H13ClFN3, a calculated molecular weight of 325.77 and the following structural formula:
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Midazolam is a short-acting benzodiazepine central nervous system (CNS) depressant.
12.2 Pharmacodynamics
The effects of midazolam on the CNS are dependent on the dose administered, the route of administration, and the presence or absence of other medications.
Time to Onset
Sedation in adult and pediatric patients is achieved within 3 to 5 minutes after intravenous injection; the time of onset is affected by total dose administered and the concurrent administration of opioid premedication. Seventy-one percent of the adult patients in endoscopy studies had no recall of introduction of the endoscope; 82% of the patients had no recall of withdrawal of the endoscope. In one study of pediatric patients undergoing lumbar puncture or bone marrow aspiration, 88% of patients had impaired recall vs 9% of the placebo controls. In another pediatric oncology study, 91% of midazolam treated patients were amnestic compared with 35% of patients who had received fentanyl alone.
When midazolam is given intravenous as an anesthetic induction agent, induction of anesthesia occurs in approximately 1.5 minutes when opioid premedication has been administered and in 2 to 2.5 minutes without opioid premedication or other sedative premedication. Some impairment in a test of memory was noted in 90% of the patients studied.
Midazolam, used as directed, does not delay awakening from general anesthesia in adults. Gross tests of recovery after awakening (orientation, ability to stand and walk, suitability for discharge from the recovery room, return to baseline Trieger competency) usually indicate recovery within 2 hours but recovery may take up to 6 hours in some cases. When compared with patients who received thiopental, patients who received midazolam generally recovered at a slightly slower rate. Recovery from anesthesia or sedation for procedures in pediatric patients depends on the dose of midazolam administered, coadministration of other medications causing CNS depression and duration of the procedure.
In patients without intracranial lesions, induction of general anesthesia with intravenous midazolam is associated with a moderate decrease in cerebrospinal fluid pressure (lumbar puncture measurements), similar to that observed following intravenous thiopental. Preliminary data in neurosurgical patients with normal intracranial pressure but decreased compliance (subarachnoid screw measurements) show comparable elevations of intracranial pressure with midazolam and with thiopental during intubation. No similar studies have been reported in pediatric patients.
Intravenous induction doses of midazolam depress the ventilatory response to carbon dioxide stimulation for 15 minutes or more beyond the duration of ventilatory depression following administration of thiopental in adults. Impairment of ventilatory response to carbon dioxide is more marked in adult patients with chronic obstructive pulmonary disease (COPD). Sedation with intravenous midazolam does not adversely affect the mechanics of respiration (resistance, static recoil, most lung volume measurements); total lung capacity and peak expiratory flow decrease significantly but static compliance and maximum expiratory flow at 50% of awake total lung capacity (Vmax) increase.
In cardiac hemodynamic studies in adults, intravenous induction of general anesthesia with midazolam was associated with a slight to moderate decrease in mean arterial pressure, cardiac output, stroke volume and systemic vascular resistance. Slow heart rates (less than 65/minute), particularly in patients taking propranolol for angina, tended to rise slightly; faster heart rates (e.g., 85/minute) tended to slow slightly. In pediatric patients, a comparison of intravenous midazolam (500 mcg/kg) with propofol (2.5 mg/kg) revealed a mean 15% decrease in systolic blood pressure in patients who had received intravenous midazolam vs a mean 25% decrease in systolic blood pressure following propofol.
Plasma Concentration-Efficacy Relationships
Concentration-efficacy relationships (after an intravenous dose) have been demonstrated for a variety of pharmacodynamic measures (eg, reaction time, eye movement, sedation) and are associated with extensive intersubject variability. Logistic regression analysis of sedation scores and steady-state plasma concentration indicated that at plasma concentrations greater than 100 ng/mL there was at least a 50% probability that patients would be sedated, but respond to verbal commands (sedation score=3). At 200 ng/mL there was at least a 50% probability that patients would be asleep, but respond to glabellar tap (sedation score=4).
12.3 Pharmacokinetics
Midazolam's activity is primarily due to the parent drug. Elimination of the parent drug takes place via hepatic metabolism of midazolam to hydroxylated metabolites that are conjugated and excreted in the urine. Six single-dose pharmacokinetic studies involving healthy adults yield pharmacokinetic parameters for midazolam in the following ranges: volume of distribution (Vd), 1.0 to 3.1 L/kg; elimination half-life, 1.8 to 6.4 hours (mean approximately 3 hours); total clearance (Cl), 0.25 to 0.54 L/hr/kg. In a parallel group study, there was no difference in the clearance, in subjects administered 0.15 mg/kg (n=4) and 0.30 mg/kg (n=4) intravenous doses indicating linear kinetics. The clearance was successively reduced by approximately 30% at doses of 0.45 mg/kg (n=4) and 0.6 mg/kg (n=5) indicating non-linear kinetics in this dose range.
Absorption
Following intramuscular administration, Cmax for midazolam and its 1-hydroxy metabolite were approximately one-half of those achieved after intravenous injection.
Distribution
The volume of distribution (Vd) determined from six single-dose pharmacokinetic studies involving healthy adults ranged from 1.0 to 3.1 L/kg. Female gender, old age, and obesity are associated with increased values of midazolam Vd. In humans, midazolam has been shown to cross the placenta and enter into fetal circulation and has been detected in human milk and CSF (see Clinical Pharmacology, Special Populations).
In adults and pediatric patients older than 1 year, midazolam is approximately 97% bound to plasma protein, principally albumin and that for 1-hydroxy metabolite is about 89%.
Elimination
Metabolism
In vitro studies with human liver microsomes indicate that the biotransformation of midazolam is mediated by cytochrome P450-3A4. This cytochrome also appears to be present in gastrointestinal tract mucosa as well as liver. Sixty to seventy percent of the biotransformation products is 1-hydroxy-midazolam (also termed alpha-hydroxy-midazolam) while 4-hydroxy-midazolam constitutes 5% or less. Small amounts of a dihydroxy derivative have also been detected but not quantified. The principal urinary excretion products are glucuronide conjugates of the hydroxylated derivatives.
Drugs that inhibit the activity of cytochrome P450-3A4 may inhibit midazolam clearance and elevate steady-state midazolam concentrations.
Studies of the intravenous administration of 1-hydroxy-midazolam in humans suggest that 1-hydroxy-midazolam is at least as potent as the parent compound and may contribute to the net pharmacologic activity of midazolam. In vitro studies have demonstrated that the affinities of 1- and 4-hydroxy-midazolam for the benzodiazepine receptor are approximately 20% and 7%, respectively, relative to midazolam.
Excretion
Clearance of midazolam is reduced in association with old age, congestive heart failure, liver disease (cirrhosis) or conditions which diminish cardiac output and hepatic blood flow.
The principal urinary excretion product is 1-hydroxy-midazolam in the form of a glucuronide conjugate; smaller amounts of the glucuronide conjugates of 4-hydroxy- and dihydroxy-midazolam are detected as well. The amount of midazolam excreted unchanged in the urine after a single intravenous dose is less than 0.5% (n=5). Following a single intravenous infusion in 5 healthy volunteers, 45% to 57% of the dose was excreted in the urine as 1-hydroxymethyl midazolam conjugate.
Pharmacokinetics-Continuous Infusion
The pharmacokinetic profile of midazolam following continuous infusion, based on 282 adult subjects, has been shown to be similar to that following single-dose administration for subjects of comparable age, gender, body habitus and health status. However, midazolam can accumulate in peripheral tissues with continuous infusion. The effects of accumulation are greater after long-term infusions than after short-term infusions. The effects of accumulation can be reduced by maintaining the lowest midazolam infusion rate that produces satisfactory sedation.
Infrequent hypotensive episodes have occurred during continuous infusion; however, neither the time to onset nor the duration of the episode appeared to be related to plasma concentrations of midazolam or alpha-hydroxy-midazolam. Further, there does not appear to be an increased chance of occurrence of a hypotensive episode with increased loading doses.
Patients with renal impairment may have longer elimination half-lives for midazolam [see Clinical Pharmacology (12.3)].
Specific Populations
Changes in the pharmacokinetic profile of midazolam due to drug interactions, physiological variables, etc., may result in changes in the plasma concentration-time profile and pharmacological response to midazolam in these patients. For example, patients with acute renal failure appear to have a longer elimination half-life for midazolam and may experience delayed recovery [see Clinical Pharmacology (12.3)]. In other groups, the relationship between prolonged half-life and duration of effect has not been established.
Age: Pediatrics and Neonates
In pediatric patients aged 1 year and older, the pharmacokinetic properties following a single dose of midazolam reported in 10 separate studies of midazolam are similar to those in adults. Weight-normalized clearance is similar or higher (0.19 to 0.80 L/hr/kg) than in adults and the terminal elimination half-life (0.78 to 3.3 hours) is similar to or shorter than in adults. The pharmacokinetic properties during and following continuous intravenous infusion in pediatric patients in the operating room as an adjunct to general anesthesia and in the intensive care environment are similar to those in adults.
In seriously ill neonates, however, the terminal elimination half-life of midazolam is substantially prolonged (6.5 to 12.0 hours) and the clearance reduced (0.07 to 0.12 L/hr/kg) compared to healthy adults or other groups of pediatric patients. It cannot be determined if these differences are due to age, immature organ function or metabolic pathways, underlying illness or debility.
Age: Geriatric
In three parallel group studies, the pharmacokinetics of midazolam administered intravenous or intramuscular were compared in young (mean age 29, n=52) and healthy elderly subjects (mean age 73, n=53). Plasma half-life was approximately two-fold higher in the elderly. The mean Vd based on total body weight increased consistently between 15% to 100% in the elderly. The mean Cl decreased approximately 25% in the elderly in two studies and was similar to that of the younger patients in the other.
Obese
In a study comparing normals (n=20) and obese patients (n=20) the mean half-life was greater in the obese group (5.9 vs 2.3 hrs). This was due to an increase of approximately 50% in the Vd corrected for total body weight. The clearance was not significantly different between groups.
Congestive Heart Failure
In patients suffering from congestive heart failure, there appeared to be a two-fold increase in the elimination half-life, a 25% decrease in the plasma clearance and a 40% increase in the volume of distribution of midazolam.
Hepatic Impairment
Midazolam pharmacokinetics were studied after an intravenous single dose (0.075 mg/kg) was administered to 7 patients with biopsy proven alcoholic cirrhosis and 8 control patients. The mean half-life of midazolam increased 2.5-fold in the alcoholic patients. Clearance was reduced by 50% and the Vd increased by 20%. In another study in 21 male patients with cirrhosis, without ascites and with normal kidney function as determined by creatinine clearance, no changes in the pharmacokinetics of midazolam or 1-hydroxy-midazolam were observed when compared to healthy individuals.
Renal Impairment
Patients with renal impairment may have longer elimination half-lives for midazolam and its metabolites which may result in slower recovery.
Midazolam and 1-hydroxy-midazolam pharmacokinetics in 6 ICU patients who developed acute renal failure (ARF) were compared with a normal renal function control group. Midazolam was administered as an infusion (5 to 15 mg/hours). Midazolam clearance was reduced (1.9 vs 2.8 mL/min/kg) and the half-life was prolonged (7.6 vs 13 hours) in the ARF patients. The renal clearance of the 1-hydroxy-midazolam glucuronide was prolonged in the ARF group (4 vs 136 mL/min) and the half-life was prolonged (12 vs >25 hours). Plasma levels accumulated in all ARF patients to about ten times that of the parent drug. The relationship between accumulating metabolite levels and prolonged sedation is unclear.
In a study of chronic renal failure patients (n=15) receiving a single intravenous dose, there was a two-fold increase in the clearance and volume of distribution but the half-life remained unchanged. Metabolite levels were not studied.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Midazolam maleate was administered with diet in mice and rats for 2 years at dosages of 1, 9, or 80 mg/kg/day. In female mice in the highest dose group there was a marked increase in the incidence of hepatic tumors. In high-dose male rats there was a small but statistically significant increase in benign thyroid follicular cell tumors. Dosages of 9 mg/kg/day of midazolam maleate (4 times a human induction dose of 0.35 mg/kg based on body surface area comparison) do not increase the incidence of tumors. The pathogenesis of induction of these tumors is not known. These tumors were found after chronic administration, whereas human use will ordinarily be of single or several doses.
Mutagenesis
Midazolam did not have mutagenic activity in Salmonella typhimurium (5 bacterial strains), Chinese hamster lung cells (V79), human lymphocytes or in the micronucleus test in mice.
Impairment of Fertility
Male rats were treated orally with 1, 4, or 16 mg/kg midazolam beginning 62 days prior to mating with female rats treated with the same doses for 14 days prior to mating to Gestation Day 13 or Lactation Day 21. The high dose produced an equivalent exposure (AUC) as 4 mg/kg intravenous midazolam (1.85 times the human induction dose of 0.35 mg/kg based on body surface area comparison). There were no adverse effects on either male or female fertility noted.
13.2 Animal Toxicology and/or Pharmacology
Published studies in animals demonstrate that the use of anesthetic agents during the period of rapid brain growth or synaptogenesis results in widespread neuronal and oligodendrocyte cell loss in the developing brain and alterations in synaptic morphology and neurogenesis. Based on comparisons across species, the window of vulnerability to these changes is believed to correlate with exposures in the third trimester through the first several months of life, but may extend out to approximately 3 years of age in humans.
In primates, exposure to 3 hours of an anesthetic regimen that produced a light surgical plane of anesthesia did not increase neuronal cell loss, however, treatment regimens of 5 hours or longer increased neuronal cell loss. Data in rodents and in primates suggest that the neuronal and oligodendrocyte cell losses are associated with subtle but prolonged cognitive deficits in learning and memory. The clinical significance of these nonclinical findings is not known, and health care providers should balance the benefits of appropriate anesthesia in neonates and young children who require procedures against the potential risks suggested by the nonclinical data [Warnings and Precautions (5.8)].
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Midazolam in 0.9% Sodium Chloride Injection is a clear, colorless solution supplied in single-dose bags with an aluminum overwrap available as:
Total Strength per Total Volume
Strength per mL
10 single-dose bags NDC
Bag and Overwrap NDC
*50 mg per 50 mL
1 mg/mL
44567-610-10
44567-610-01
100 mg per 100 mL
1 mg/mL
44567-611-10
44567-611-01
*Partial fill container 50 mL volume in 100 mL container
Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Protect from Freezing. Individual containers may be used up to 48 hours after initial penetration. Discard unused portion.
-
17 PATIENT COUNSELING INFORMATION
Alcohol and Current Medication
Advise patients to notify their healthcare provider about alcohol or medication use, especially blood pressure medication and antibiotics. Alcohol and other CNS depressants, such as opioid analgesic and benzodiazepines, can have an additive effect when administered with Midazolam in 0.9% Sodium Chloride Injection [see Warnings and Precautions (5.2), Drug Interactions (7.1)].Effect of Anesthetic and Sedation Drugs on Early Brain Development\
Studies conducted in young animals and children suggest repeated or prolonged use of general anesthetic or sedation drugs in children younger than 3 years may have negative effects on their developing brains. Discuss with parents and caregivers the benefits, risks, and timing and duration of surgery or procedures requiring anesthetic and sedation drugs [see Warnings and Precautions (5.11), Use in Specific Populations (8.1)].Pregnancy
Advise pregnant females exposed to midazolam late in pregnancy can result in sedation (respiratory depression, lethargy, hypotonia) and/or withdrawal symptoms (hyperreflexia, irritability, restlessness, tremors, inconsolable crying, and feeding difficulties) in newborns. Instruct patients to inform their healthcare provider if they are pregnant during treatment with Midazolam in 0.9% Sodium Chloride Injection [see Warnings and Precautions (5.10), Use in Specific Populations (8.1)].
Lactation
Instruct patients to notify their healthcare provider if they are breastfeeding or intend to breastfeed. Instruct breastfeeding patients receiving midazolam to monitor infants for excessive sedation, poor feeding, and poor weight gain, and to seek medical attention if they notice these signs. A lactating woman may consider pumping and discarding breastmilk for at least 4 to 8 hours after receiving midazolam for sedation or anesthesia to minimize drug exposure to a breastfed infant [see Use in Specific Populations (8.2)].
Residual Sedation and Amnesia
Advise patients that they may experience residual sedation and amnesia. The decision as to when patients who have received injectable midazolam, particularly on an outpatient basis, may again engage in activities requiring complete mental alertness, operate hazardous machinery, or drive a motor vehicle must be individualized [see Warnings and Precautions (5.8)].Withdrawal
Advise patients that receive midazolam in a critical care setting over an extended period of time that they may experience symptoms of withdrawal following abrupt discontinuation.
Discard unused portion.
Manufactured for:
WG Critical Care, LLC
Paramus, NJ 07652
Made in Switzerland
Patented. See www.wgcriticalcare.com/patents
-
Package/Label Display Panel
High Alert Medication
NDC: 44567-610-01 Do Not Dilute 50 mL
Midazolam
in 0.9% Sodium Chloride
Injection50 mg per 50 mL
(1 mg per mL)
-
Package/Label Display Panel
High Alert Medication
NDC: 44567-611-01 Do Not Dilute 100 mL
Midazolam
in 0.9% Sodium Chloride
Injection100 mg per 100 mL (1 mg per mL)
-
INGREDIENTS AND APPEARANCE
MIDAZOLAM IN SODIUM CHLORIDE
midazolam injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 44567-610 Route of Administration INTRAVENOUS DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MIDAZOLAM (UNII: R60L0SM5BC) (MIDAZOLAM - UNII:R60L0SM5BC) MIDAZOLAM 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) 9 mg in 1 mL HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Other Ingredients Ingredient Kind Ingredient Name Quantity Does not contain NATURAL LATEX RUBBER (UNII: 2LQ0UUW8IN) 0 in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 44567-610-10 10 in 1 CARTON 04/19/2021 1 NDC: 44567-610-01 50 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA211844 12/31/2020 MIDAZOLAM IN SODIUM CHLORIDE
midazolam injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 44567-611 Route of Administration INTRAVENOUS DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MIDAZOLAM (UNII: R60L0SM5BC) (MIDAZOLAM - UNII:R60L0SM5BC) MIDAZOLAM 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) 9 mg in 1 mL HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Other Ingredients Ingredient Kind Ingredient Name Quantity Does not contain NATURAL LATEX RUBBER (UNII: 2LQ0UUW8IN) 0 in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 44567-611-10 10 in 1 CARTON 04/19/2021 1 NDC: 44567-611-01 100 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA211844 12/31/2020 Labeler - WG Critical Care, LLC. (829274633) Establishment Name Address ID/FEI Business Operations InfoRLife SA 481732753 MANUFACTURE(44567-610, 44567-611)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.