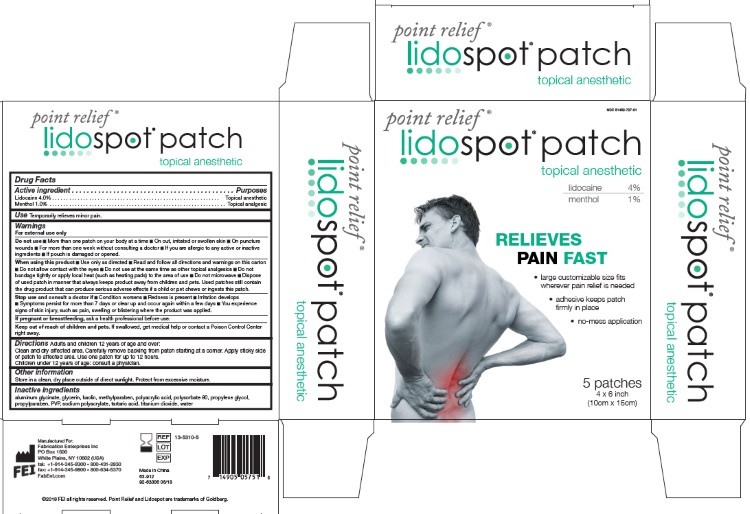
POINT RELIEF LIDOSPOT WITH MENTHOL- lidocaine, menthol patch
Point Relief LidoSpot with Menthol by
Drug Labeling and Warnings
Point Relief LidoSpot with Menthol by is a Otc medication manufactured, distributed, or labeled by Fabrication Enterprises, Foshan Aqua Gel Biotech Co.,Ltd.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Drug Facts
- ACTIVE INGREDIENT
- PURPOSE
- USES
-
WARNINGS
- For External use only. Use only as directed.
- Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
■ More than one patch on your body at a time
■ On cut, irritated or swollen skin
■ On puncture wounds
■ For more than one week without consulting a doctor
■ If you are allergic to any active or inactive ingredients
■ If pouch is damaged or opened.
WHEN USING:
■ Use only as directed
■ Read and follow all directions and warnings on this carton
■ Do not allow contact with the eyes
■ Do not use at the same time as other topical analgesics
■ Do not bandage tightly or apply local heat (such as heating pads) to the area of use
■ Do not microwave
■ Dispose of used patch in manner that always keeps product away from children and pets. Used patches still contain
the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch. - DIRECTIONS Adults and children 12 years of age and over:
- INACTIVE INGREDIENTS
- STORAGE AND HANDLING
- Manufactured For:Fabrication Enterprises IncWhite Plains, NY 10602www.FabEnt.com
-
INGREDIENTS AND APPEARANCE
POINT RELIEF LIDOSPOT WITH MENTHOL
lidocaine, menthol patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 51452-912 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 4 g in 100 g MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL, UNSPECIFIED FORM 1 g in 100 g Inactive Ingredients Ingredient Name Strength DIHYDROXYALUMINUM AMINOACETATE ANHYDROUS (UNII: 1K713C615K) GLYCERIN (UNII: PDC6A3C0OX) KAOLIN (UNII: 24H4NWX5CO) METHYLPARABEN (UNII: A2I8C7HI9T) POLYACRYLIC ACID (250000 MW) (UNII: 9G2MAD7J6W) POLYSORBATE 80 (UNII: 6OZP39ZG8H) PROPYLENE GLYCOL 1-(2-METHYLBUTYRATE) (UNII: 9Q5W5G6461) PROPYLPARABEN (UNII: Z8IX2SC1OH) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM POLYACRYLATE (2500000 MW) (UNII: 05I15JNI2J) TARTARIC ACID (UNII: W4888I119H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 51452-912-05 5 in 1 POUCH 07/01/2018 1 8 g in 1 PATCH; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 07/01/2018 Labeler - Fabrication Enterprises (070577218) Establishment Name Address ID/FEI Business Operations Foshan Aqua Gel Biotech Co.,Ltd. 529128763 manufacture(51452-912)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.