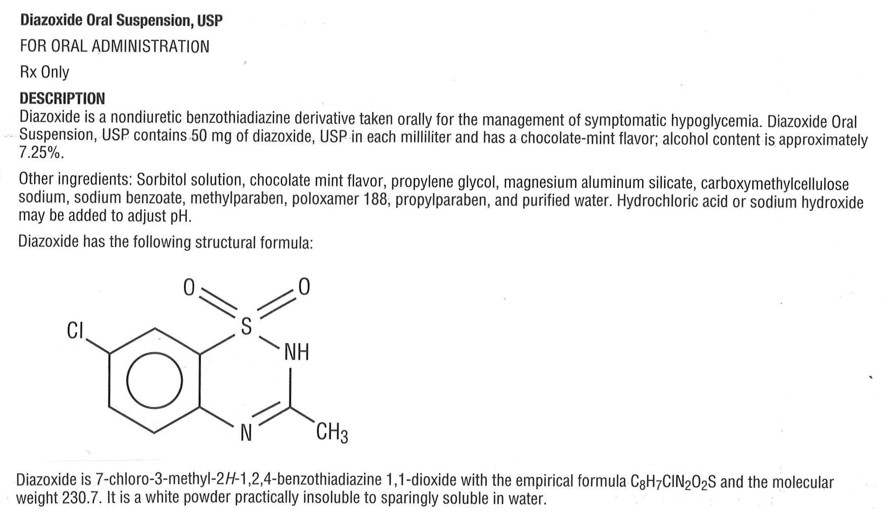
Diazoxide Oral Suspension, USP
Diazoxide Oral Solution by
Drug Labeling and Warnings
Diazoxide Oral Solution by is a Prescription medication manufactured, distributed, or labeled by TriRx Huntsville Pharmaceutical Services LLC, TriRx Huntsville Pharmaceutical Services, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
DIAZOXIDE ORAL SOLUTION- diazoxide oral suspension suspension
TriRx Huntsville Pharmaceutical Services LLC
----------
Diazoxide Oral Suspension, USP
| DIAZOXIDE ORAL SOLUTION
diazoxide oral suspension suspension |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - TriRx Huntsville Pharmaceutical Services LLC (117090286) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| TriRx Huntsville Pharmaceutical Services, LLC | 117090286 | manufacture(80432-057) | |
Revised: 1/2023
Document Id: f1863e5d-f2b6-2e47-e053-2a95a90af475
Set id: f1863e5d-f2b5-2e47-e053-2a95a90af475
Version: 1
Effective Time: 20230105
TriR
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.