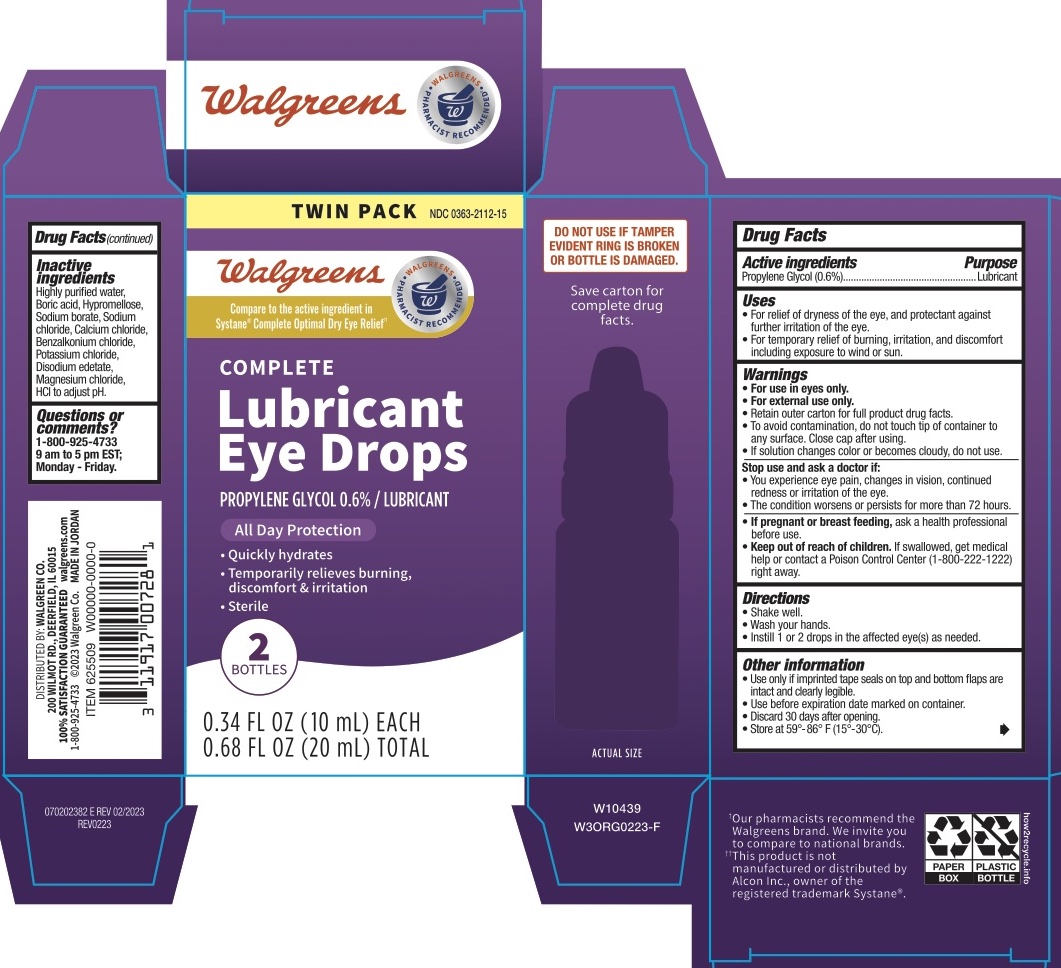
Walgreens Lubricant Eye Drops (Systane COMPLETE) Drug Facts
Walgreens Lubricant Eye Drops by
Drug Labeling and Warnings
Walgreens Lubricant Eye Drops by is a Otc medication manufactured, distributed, or labeled by WALGREEN COMPANY. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
WALGREENS LUBRICANT EYE DROPS- propylene glycol solution/ drops
WALGREEN COMPANY
----------
Walgreens Lubricant Eye Drops (Systane COMPLETE)
Drug Facts
Uses
- For relief of dryness of the eye, and protectant against further irritation of the eye.
- For temporary relief of buming, irritation, and discomfort including exposure to wind or sun.
Warnings
For use in eyes only.
For external use only.
Retain outer carton for full product drug facts.
To avoid contamination, do not touch tip of container to any surface. Close cap after using.
If solution changes color or becomes cloudy, do not use.
When using this product
Retain outer carton for full product drug facts.
To avoid contamination, do not touch tip of container to any surface. Close cap after using.
If solution changes color or becomes cloudy, do not use.
Stop use and ask a doctor if you experience any of the following:
You experience eye pain, changes in vision, continued redness or irritation of the eye.
The condition worsens or persists for more than 72 hours.
Other Information
Use only if imprinted tape seals on top and bottom flaps are intact and clearly legible.
Use before expiration date marked on container. Discard 30 days after opening.
Store at 59°-86° F (15°-30°C).
| WALGREENS LUBRICANT EYE DROPS
propylene glycol solution/ drops |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - WALGREEN COMPANY (008965063) |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.