ZOLYMBUS- bimatoprost gel
ZOLYMBUS by
Drug Labeling and Warnings
ZOLYMBUS by is a Prescription medication manufactured, distributed, or labeled by Thea Pharma Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ZOLYMBUS™ safely and effectively. See full prescribing information for ZOLYMBUS™.
ZOLYMBUS™ (bimatoprost ophthalmic gel) 0.01%, for topical ophthalmic use
Initial U.S. Approval: 2001INDICATIONS AND USAGE
ZOLYMBUS™ is a prostaglandin analog indicated for the reduction of elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension. (1)
DOSAGE AND ADMINISTRATION
One drop in the affected eye(s) once daily in the evening. (2)
DOSAGE FORMS AND STRENGTHS
Ophthalmic gel: 0.01% bimatoprost in a single-dose container. (3)
CONTRAINDICATIONS
Hypersensitivity. (4)
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
Most common adverse reactions (≥10%) are conjunctival hyperemia (14%) and eye irritation (11%). ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Thea Pharma Inc. at 1-833-838-4028 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
Use in pediatric patients below the age of 16 years is not recommended because of potential safety concerns related to increased pigmentation following long-term chronic use. ( 8.4)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 9/2025
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Pigmentation
5.2 Eyelash Changes
5.3 Intraocular Inflammation
5.4 Macular Edema
5.5 Contact Lens Use
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
The recommended dosage is one drop in the affected eye(s) once daily in the evening. ZOLYMBUS should not be administered more than once daily since it has been shown that more frequent administration of prostaglandin analogs may lessen the IOP lowering effect.
Reduction of the IOP starts approximately 4 hours after the first administration with maximum effect reached within approximately 8 to 12 hours.
ZOLYMBUS may be used concomitantly with other topical ophthalmic drug products to lower IOP. If more than one topical ophthalmic drug is being used, the drugs should be administered at least five (5) minutes apart.If one dose is missed, treatment should continue with the next dose as normal.
The gel from one single-dose container is to be used immediately after opening for administration to one or both eyes. Since sterility cannot be maintained after the single-dose container is opened, discard the open container and the remaining contents immediately after administration. - 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
ZOLYMBUS is contraindicated in patients with hypersensitivity to bimatoprost or to any of the ingredients [see Adverse Reactions (6.2)] .
-
5 WARNINGS AND PRECAUTIONS
5.1 Pigmentation
Bimatoprost has been reported to cause changes to pigmented tissues. The most frequently reported changes have been increased pigmentation of the iris, periorbital tissue (eyelid) and eyelashes. Pigmentation is expected to increase as long as bimatoprost is administered. The pigmentation change is due to increased melanin content in the melanocytes rather than to an increase in the number of melanocytes. After discontinuation of bimatoprost, pigmentation of the iris is likely to be permanent, while pigmentation of the periorbital tissue and eyelash changes have been reported to be reversible in some patients. Patients who receive treatment should be informed of the possibility of increased pigmentation. The long term effects of increased pigmentation are not known.
Iris color change may not be noticeable for several months to years. Typically, the brown pigmentation around the pupil spreads concentrically towards the periphery of the iris and the entire iris or parts of the iris become more brownish. Neither nevi nor freckles of the iris appear to be affected by treatment. While treatment with ZOLYMBUS can be continued in patients who develop noticeably increased iris pigmentation, these patients should be examined regularly.
5.2 Eyelash Changes
ZOLYMBUS may gradually change eyelashes and vellus hair in the treated eye. These changes include increased length, thickness, and number of lashes. Eyelash changes are usually reversible upon discontinuation of treatment.
5.3 Intraocular Inflammation
Prostaglandin analogs, including bimatoprost, have been reported to cause intraocular inflammation. ZOLYMBUS should be used with caution in patients with active intraocular inflammation (e.g., iritis/uveitis) because inflammation may be exacerbated.
5.4 Macular Edema
Macular edema, including cystoid macular edema, has been reported during treatment with bimatoprost ophthalmic products. ZOLYMBUS should be used with caution in aphakic patients, in pseudophakic patients with a torn posterior lens capsule, or in patients with known risk factors for macular edema.
-
6 ADVERSE REACTIONS
The following adverse reactions are described elsewhere in the labeling:
- Pigmentation including blepharal pigmentation and iris hyperpigmentation [see Warnings and Precautions (5.1)]
- Eyelash Changes [see Warnings and Precautions (5.2)]
- Intraocular Inflammation [see Warnings and Precautions (5.3)]
- Macular Edema [see Warnings and Precautions (5.4)]
- Hypersensitivity [see Contraindications (4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In the two clinical trials conducted with ZOLYMBUS (bimatoprost ophthalmic gel) 0.01% comparing it to preserved bimatoprost ophthalmic solution 0.01%, the most frequently reported ocular adverse reactions were conjunctival hyperemia (14%), eye irritation (11%), eye pruritus (9%) and foreign body sensation in eyes (8%). Other adverse drug reactions (reported in 1% to 6% of patients) with ZOLYMBUS in the studies included dry eye, abnormal sensation in eye, lacrimation increased, eye pain, and vision blurred.6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of topical bimatoprost products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These reactions, which have been chosen for inclusion due to either their seriousness, frequency of reporting, possible causal connection to bimatoprost, or a combination of these factors include: asthma-like symptoms, dizziness, dry eye, dyspnea, eye discharge, eye edema, foreign body sensation, headache, hypersensitivity including signs and symptoms of eye allergy and allergic dermatitis, hypertension, lacrimation increased, periorbital and lid changes associated with periorbital fat atrophy leading to skin tightness, deepening of the eyelid sulcus, eyelid ptosis, enophthalmos, and eyelid retraction; and photophobia.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate and well-controlled studies of ZOLYMBUS (bimatoprost ophthalmic gel) 0.01% administration in pregnant women. There is no increase in the risk of major birth defects or miscarriages based on bimatoprost post-marketing experience.In embryofetal developmental studies, administration of bimatoprost to pregnant mice and rats during organogenesis, resulted in abortion and early delivery at oral doses at least 33 times (mice) or 94 times (rats) the human exposure to bimatoprost solution 0.03% dosed bilaterally once daily (based on blood area under the curve [AUC] levels). These adverse effects were not observed at 2.6 times (mice) and 47 times (rats) the human exposure to bimatoprost 0.03% dosed bilaterally once daily (based on blood AUC levels).
In pre/postnatal development studies, administration of bimatoprost to pregnant rats from organogenesis to the end of lactation resulted in reduced gestation length and fetal body weight, and increased fetal and pup mortality at oral doses at least 41 times the human systemic exposure to bimatoprost solution 0.03% dosed bilaterally once daily (based on blood AUC levels). No adverse effects were observed in rat offspring at exposures estimated at 14 times the human exposure to bimatoprost solution 0.03% dosed bilaterally once daily (based on blood AUC levels).
Because animal reproductive studies are not always predictive of human response ZOLYMBUS should be administered during pregnancy only if the potential benefit justifies the potential risk to the fetus.
The background risk of major birth defects and miscarriage for the indicated population is unknown. However, the background risk in the U.S. general population of major birth defects is 2 to 4%, and of miscarriage is 15 to 20%, of clinically recognized pregnancies.
Data
Animal Data
In an embryofetal development rat study, abortion was observed in pregnant rats administered bimatoprost orally during organogenesis at 0.6 mg/kg/day (94 times the human systemic exposure to bimatoprost solution 0.03% dosed bilaterally once daily, based on AUC). The No Observed Adverse Effect Level (NOAEL) for abortion was 0.3 mg/kg/day (estimated at 47 times the human systemic exposure to bimatoprost solution 0.03% dosed bilaterally once daily, based on AUC). No abnormalities were observed in rat fetuses at doses up to 0.6 mg/kg/day.In an embryofetal development mouse study, abortion and early delivery were observed in pregnant mice administered bimatoprost orally during organogenesis at doses greater than or equal to 0.3 mg/kg/day (33 times the human systemic exposure to bimatoprost solution 0.03% dosed bilaterally once daily, based on AUC). The NOAEL for abortion and early delivery was 0.1 mg/kg/day (2.6 times the human systemic exposure to bimatoprost solution 0.03% dosed bilaterally once daily, based on AUC). No abnormalities were observed in mouse fetuses at doses up to 0.6 mg/kg/day (72 times the human systemic exposure to bimatoprost solution 0.03% dosed bilaterally once daily based on AUC).
In a pre/postnatal development study, treatment of pregnant rats with bimatoprost orally from gestation day 7 to lactation day 20 resulted in reduced gestation length, increased late resorptions, fetal deaths, and postnatal pup mortality, and reduced pup body weight at doses greater than or equal to 0.3 mg/kg/day. These effects were observed at exposures at least 41 times the human systemic exposure to bimatoprost solution 0.03% dosed bilaterally once daily, based on AUC. The NOAEL for postnatal development and mating performance of the offspring was 0.1 mg/kg/day (estimated at 14 times the human systemic exposure to bimatoprost solution 0.03% dosed bilaterally once daily, based on AUC).
8.2 Lactation
Risk Summary
It is not known whether topical ocular treatment with ZOLYMBUS could result in sufficient systemic absorption to produce detectable quantities in human milk. In animal studies, bimatoprost has been shown to be present in breast milk of lactating rats at an intravenous dose (i.e., 1 mg/kg) 970 times the recommended human ophthalmic dose (on a mg/m 2basis), however, no animal data is available at clinically relevant doses.The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for ZOLYMBUS and any potential adverse effects on the breastfed child from ZOLYMBUS.
-
10 OVERDOSAGE
No information is available on overdosage in humans. If overdose with ZOLYMBUS occurs, treatment should be symptomatic.
In oral (by gavage) mouse and rat studies, doses up to 100 mg/kg/day did not produce any toxicity. This dose expressed as mg/m 2is at least 90 times higher than the accidental dose of the entire content of a pack of ZOLYMBUS (30 x 0.3 g single-dose containers; 9 g) for a 10 kg child.
-
11 DESCRIPTION
ZOLYMBUS contains bimatoprost, a synthetic prostamide analog with ocular hypotensive activity, for topical ophthalmic use. The chemical name is 5-Heptenamide, 7-[(1R,2R,3R,5S)-3,5-dihydroxy-2- [(1E,3S)-3-hydroxy-5-phenyl-1-penten-1-yl]cyclopentyl]-N-ethyl, (5Z)-, with the molecular weight 415.58 and molecular formula is C 25H 37NO 4. Its chemical structure is:
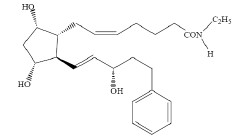
Bimatoprost is a crystalline powder, which is very soluble in ethyl alcohol and methyl alcohol and slightly soluble in water. ZOLYMBUS is a sterile, colorless, opalescent ophthalmic gel with an osmolality of approximately 300 mOsmol/kg.
Each container contains 0.03 mg of bimatoprost and the following inactive ingredients: carbomer, polyethylene glycol, sodium acetate trihydrate, sorbitol, sodium hydroxide and water for injection with a pH range from 6.9 to 7.9.
ZOLYMBUS does not contain a preservative. -
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Bimatoprost, a prostaglandin analog, is a synthetic structural analog of prostaglandin with ocular hypotensive activity. It selectively mimics the effects of naturally occurring substances, prostamides. Bimatoprost is believed to lower intraocular pressure (IOP) in humans by increasing outflow of aqueous humor through both the trabecular meshwork and uveoscleral routes. Elevated IOP presents a major risk factor for glaucomatous field loss. The higher the level of IOP, the greater the likelihood of optic nerve damage and visual field loss.
12.3 Pharmacokinetics
Absorption
After one drop of bimatoprost ophthalmic gel 0.01% was administered once daily to both eyes of 20 healthy participants for two weeks, blood concentrations peaked within 10 minutes after dosing and were below the lower limit of detection (5 pg/mL) in most subjects within 30 minutes after dosing. Mean C maxvalues were similar on days 1 and 14 at 0.021 ng/mL and 0.028 ng/mL, respectively, indicating that a steady state was reached. There was no significant systemic drug accumulation over time.Distribution
Bimatoprost is moderately distributed into body tissues with a steady-state volume of distribution of 0.67 L/kg. In human blood, bimatoprost resides mainly in the plasma. Approximately 12% of bimatoprost remains unbound in human plasma.Elimination
Metabolism
Bimatoprost is the major circulating species in the blood once it reaches the systemic circulation following ocular dosing. Bimatoprost then undergoes oxidation, N-deethylation and glucuronidation to form a diverse variety of metabolites.
Excretion
Following an intravenous dose of radiolabeled bimatoprost (3.12 mcg/kg) to six healthy subjects, the maximum blood concentration of unchanged drug was 12.2 ng/mL and decreased rapidly with an elimination half-life of approximately 45 minutes. The total blood clearance of bimatoprost was 1.5 L/hr/kg. Up to 67% of the administered dose was excreted in the urine while 25% of the dose was recovered in the feces. -
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Bimatoprost was not carcinogenic in either mice or rats when administered by oral gavage for 104 weeks at doses up to 2 mg/kg/day and 1 mg/kg/day, respectively (192 and 291 times the estimated human systemic exposure to bimatoprost solution 0.03% dosed bilaterally once daily, respectively, based on blood AUC levels).Mutagenesis
Bimatoprost was not mutagenic or clastogenic in the Ames test, in the mouse lymphoma test, or in the in vivo mouse micronucleus tests.Impairment of Fertility
Bimatoprost did not impair fertility in male or female rats up to doses of 0.6 mg/kg/day (at least 103 times the recommended human exposure to bimatoprost solution 0.03% dosed bilaterally once daily based on blood AUC levels). - 14 CLINICAL STUDIES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
ZOLYMBUS (bimatoprost ophthalmic gel) 0.01% is supplied as a colorless, opalescent gel in blow-fill-seal single-dose container packaged in foil pouches.
10 single-dose containers per pouch.
NDC: 82584-010-30; Carton of 30 single-dose containersStorage:
Store at 20°C to 30°C (68°F to 86°F).
Protect from light. Store in the original pouch. After the pouch is opened, the single-dose containers may be stored in the opened foil pouch for up to 14 days at 20°C to 30°C (68°F to 86°F).
Patient should be advised to write down the date the foil pouch is opened in the space provided on the pouch.
Discard any unused containers 14 days after first opening the pouch.Single-dose container. Discard unused portion.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Potential for Pigmentation
Advise patients about the potential for increased brown pigmentation of the iris, which may be permanent. Also inform patients about the possibility of eyelid skin darkening, which may be reversible after discontinuation of ZOLYMBUS (bimatoprost ophthalmic gel).
Potential for Eyelash Changes
Inform patients of the possibility of eyelash and vellus hair changes in the treated eye during treatment with ZOLYMBUS. These changes may result in a disparity between eyes in length, thickness, pigmentation, number of eyelashes or vellus hairs, and/or direction of eyelash growth. Eyelash changes are usually reversible upon discontinuation of treatment.
Handling the Container
Advise patients that ZOLYMBUS is a sterile gel that does not contain a preservative. The drops are supplied in a single-dose container. The gel from one single-dose container is to be used immediately after opening for administration to one or both eyes. Since sterility cannot be maintained after the single-dose container is opened, discard the open container and the remaining contents immediately after administration. Open a new single-dose container every time you use ZOLYMBUS.
When to Seek Physician Advice
Advise patients that if they develop an intercurrent ocular condition (e.g., trauma or infection), have ocular surgery, or develop any ocular reactions, particularly conjunctivitis and eyelid reactions, they should immediately seek their physician’s advice concerning the continued use of ZOLYMBUS.Contact Lens Use
Advise patients that contact lenses should be removed prior to administration of ZOLYMBUS. Lenses may be reinserted 15 minutes following administration of ZOLYMBUS.Use with Other Ophthalmic Drugs
Advise patients that if more than one topical ophthalmic drug is being used, the drugs should be administered at least five (5) minutes apart.If a Dose is Missed
Advise patients that if one dose is missed, treatment should continue with the next dose as normal.Manufactured for: Thea Pharma Inc.Waltham, MA 02451.
U.S. Patent N°. 10,314,780. ©2025, Laboratoires Théa. All rights reserved. ZOLYMBUS™ is a trademark of Laboratoires Théa. -
PATIENT PACKAGE INSERT
PATIENT INFORMATION
ZOLYMBUS TM(zoh LIM bus)
(bimatoprost ophthalmic gel 0.01%)
for topical ophthalmic useThis Patient Information has been approved by the U.S. Food and Drug Administration. Approved: 09/2025 What is ZOLYMBUS?
ZOLYMBUS is a prescription eye gel used to lower the pressure in the eye (intraocular pressure) in people with open-angle glaucoma or ocular hypertension when their eye pressure is too high. ZOLYMBUS belongs to a group of medicines called prostaglandin analogs.
ZOLYMBUS is not recommended in children younger than 16 years of age because of the potential for eye and eyelid color changes.Do not use ZOLYMBUS ifyou are allergic to bimatoprost or any of the ingredients in ZOLYMBUS. See the end of this Patient Information leaflet for a complete list of ingredients in ZOLYMBUS. Before using ZOLYMBUS, tell your healthcare provider about all your medical conditions, including if you: - have or have had eye problems including any surgery on your eye or eyes
- are using any other eye medicines
- are pregnant or plan to become pregnant. It is not known if ZOLYMBUS will harm your unborn baby. If you become pregnant while using ZOLYMBUS talk to your healthcare provider right away.
- are breastfeeding or plan to breastfeed. It is not known if ZOLYMBUS passes into your breast milk. Talk to your healthcare provider about the best way to feed your baby if you use ZOLYMBUS.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.How should I use ZOLYMBUS?
Read the Instructions for Use at the end of this Patient Information leaflet for additional instructions about the right way to use ZOLYMBUS.- Use 1 drop of ZOLYMBUS in your affected eye or eyes each evening.Talk to your healthcare provider or pharmacist if you are not sure how to use ZOLYMBUS.
- Your ZOLYMBUS may not work as well if you use it more than 1 time each evening.
- If you miss a dose of ZOLYMBUS, skip the missed dose and use the next dose at your regular time.
- If you use other medicines in your eye, wait at least 5 minutes between using ZOLYMBUS and your other eye medicines.
- Contact lenses should be taken out before using ZOLYMBUS and you should wait at least 15 minutes after giving the dose of ZOLYMBUS before putting the contact lenses back into your eyes.
- Use your ZOLYMBUS right away after opening for use in 1 or both eyes. Each ZOLYMBUS single-dose container is sterile and is to be used 1 time then thrown away. Do not save any ZOLYMBUS that may be left over after you use your medicine. Using ZOLYMBUS that is not sterile may cause other eye problems.
What are the possible side effects of ZOLYMBUS?
ZOLYMBUS may cause serious side effects including:- changes in the color of your eye (iris).Your iris may become more brown in color while using ZOLYMBUS. This color change may not go away when you stop using ZOLYMBUS. If ZOLYMBUS is used in 1 eye only, the color of that eye may always be a different color from the color of your other eye.
- darkening of the color of the skin around your eye (eyelid).These skin changes usually go away when you stop using ZOLYMBUS.
- increasing the length, thickness, color, and number of your eyelashes.These eyelash changes usually go away when you stop using ZOLYMBUS.
- hair growth on skin around the treated eye.This hair growth usually goes away when you stop using ZOLYMBUS.
- inner eye swelling(inflammation). Swelling or thickening may occur in the center of your eye (macular edema) and swelling in the middle layer of your eye (uvea, intraocular inflammation). Tell your healthcare provider if you experience eye inflammation or have a known risk of macular edema.
- redness of and around the eye (conjunctival hyperemia)
- eye irritation
- itchy eyes (eye pruritus)
- dry eye
- foreign body sensation in the eyes
- increase of tears in the eye (increased lacrimation)
- eye pain
- blurry vision
Tell your healthcare provider right away if you have any new eye problems while using ZOLYMBUS, including:
an eye injuryan eye infection
eye surgery
swelling and redness of and around your eye (conjunctivitis)
problems with your eyelids
Additionally, the following side effects have been reported in other bimatoprost medicines like ZOLYMBUS, when used in the eye (topical use):
asthma-like symptoms
dizziness
shortness of breath (dyspnea)
eye discharge (eye mucus)
inflammation of the eye (eye edema)
headache
signs and symptoms of eye allergy and allergic inflammation of the skin (allergic dermatitis)
hypertension
periorbital and lid changes associated with loss of fat leading to skin tightness
deepening of the eyelid crease (eyelid sulcus)
drooping eyelid (eyelid ptosis)
sunken eye (enophtalmos)
eyelid retraction
sensitivity to light (photophobia)
Tell your healthcare provider if you have any side effect that bothers you or does not go away.These are not all the possible side effects of ZOLYMBUS. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA- 1088.How should I store ZOLYMBUS?
Before opening the foil pouches:
Keep the ZOLYMBUS single-dose containers in their original foil pouch. Protect from light.Store the unopened foil pouches between 68°F to 86°F (20°C to 30°C).
Do not open the foil pouch containing ZOLYMBUS until you are ready to use the eye drops.
After opening the foil pouch:
Store the single-dose containers in the opened foil pouch between 68°F to 86°F (20°C to 30°C), for up to 14 days.
Write down the date of first opening the foil pouch in the space provided on the pouch.
Throw away all unused ZOLYMBUS single-dose containers in the opened foil pouch after 14 days.
Keep ZOLYMBUS and all medicines out of the reach of children.General information about the safe and effective use of ZOLYMBUS.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ZOLYMBUS for a condition for which it was not prescribed. Do not give ZOLYMBUS to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your pharmacist or healthcare provider for information about ZOLYMBUS that is written for health professionals.What are the ingredients in ZOLYMBUS?
Active ingredients:bimatoprost
Inactive ingredients:carbomer, polyethylene glycol, sodium acetate trihydrate, sorbitol, sodium hydroxide and water.ZOLYMBUS does not contain a preservative.
Manufactured for: Thea Pharma Inc.Waltham, MA 02451. U.S. Patent N°. 10,314,780. ©2025, Laboratoires Théa. All rights reserved. ZOLYMBUS™ is a trademark of Laboratoires Théa.
-
INSTRUCTIONS FOR USE
Instructions for Use
ZOLYMBUS™ (zoh LIM bus)
(bimatoprost ophthalmic gel) 0.01%
for topical ophthalmic useRead this Instructions for Use before using your ZOLYMBUS™ and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your healthcare provider about your medical condition or your treatment. Important Information You Need to Know Before Using ZOLYMBUS: - ZOLYMBUS is for use in the eye. Do not swallow ZOLYMBUS.
- ZOLYMBUS single-dose containers are packaged in a foil pouch.
- Do notuse the ZOLYMBUS single-dose containers if the foil pouch is already opened when you receive it.
- Write down the date you open the foil pouch in the space provided on the pouch.
- Use 1 drop of ZOLYMBUS in your affected eye or eyes each evening.
- If you use other eye medicines dropped into the eye (topically applied ophthalmic medicines), use the medicines at least 5 minutes before or 5 minutes after the use of ZOLYMBUS.
- Do notopen the ZOLYMBUS single-dose container until you are ready to use the eye drops.
Please follow these instructions to use ZOLYMBUS: Step 1. Wash your hands and sit or stand comfortably. Step 2. - Open the foil pouch containing a strip of 10 single-dose containers.
- Write down the date of first opening on the foil pouch.
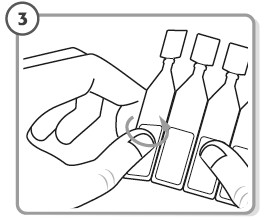
Step 3. - Take the strip of single-dose containers from the foil pouch.
- Break off 1 single-dose container from the strip.
- Place the remaining strip of unopened single-dose containers back in the foil pouch and fold the edge to close the pouch.
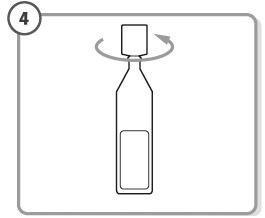
Step 4. - Hold the single-dose container upright.
- Make sure that your ZOLYMBUS medicine is in the bottom part of the single-dose container.
- Twist open the top of the single-dose container as shown. Do not touch the tip after opening the container.
Step 5. - Tilt your head backwards. If you are unable to tilt your head, lie down.
- Use your finger to gently pull down the lower eyelid of your affected eye.
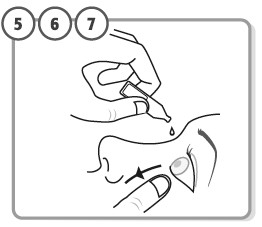
Step 6. Place the tip of the single-dose container close to, but not touching your eye. Step 7. Squeeze the single-dose container gently so that only 1 drop goes into your eye, then release the lower eyelid.
If the drop misses your eye completely, try again.- If your healthcare provider has told you to use ZOLYMBUS drops in both eyes, repeat Step 5 to Step 7 for your other eye.
- Each single-dose container contains enough drops for both eyes.
- Throw away the single-dose container after use. Do not keep it to use it again even if there is product left in the container.To lessen the chance of infection, a new single-dose container must be opened each time you are ready to use ZOLYMBUS.
- Place the folded foil pouch back in the carton. The unopened single-dose containers must be used within 14 days after opening the foil pouch. Protect from light.

Manufactured for: Thea Pharma Inc.Waltham, MA 02451.
U.S. Patent N°. 10,314,780. ©2025, Laboratoires Théa. All rights reserved. ZOLYMBUS™ is a trademark of Laboratoires Théa.This Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration. Approved: 09/2025 -
PRINCIPAL DISPLAY PANEL -ophthalmic gel 0.01%; Carton of 30 single-dose containers
NDC: 82584-010-30
ZOLYMBUS™
(bimatoprost ophthalmic gel)
0.01%
Contains:
Active:bimatoprost 0.1 mg/g.
Inactive ingredients:carbomer, sodium acetate
trihydrate, polyethylene glycol, sodium hydroxide (for
pH adjustment), sorbitol and water for injection.
Recommended Dosage:one drop in the affected eye(s)
once daily in the evening.
Single-dose container. Discard unused portion.
Storage:Store at 20°C to 30°C (68°F to 86°F).
See Prescribing Information.
30 single-dose containers: 3 pouches x 10 single-dose
containers (0.3 g each).
Store in the original pouch. After the pouch is opened, the
single-dose containers may be stored in the opened foil
pouch for up to 14 days.
Discard any unused containers 14 days after first
opening the pouch.
Manufactured for Thea Pharma Inc.
Waltham, MA 02451
U.S. Patent N°. 10,314,780. ©2024, Laboratoires Théa.
All rights reserved. ZOLYMBUS™ is a trademark of
Laboratoires Théa.
Théa Logo
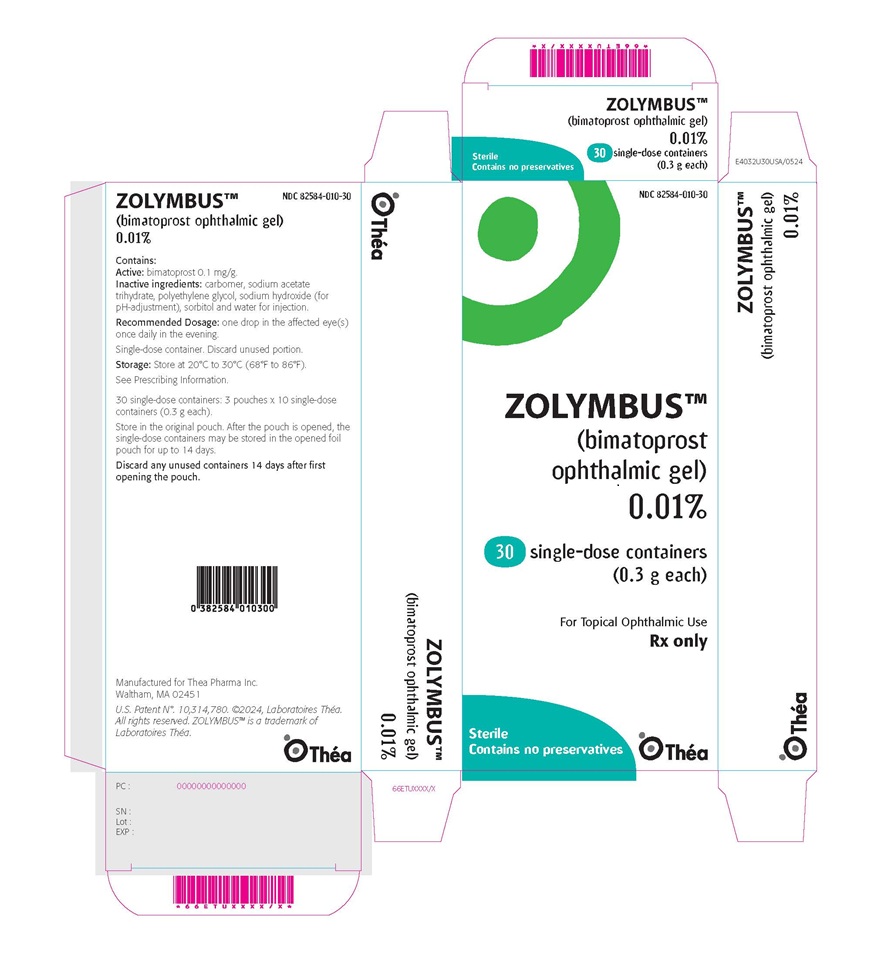
-
PRINCIPAL DISPLAY PANEL - ophthalmic gel 0.01%; Pouch of 10 single-dose containers
NDC: 82584-010-81
Recommended Dosage:one drop in the affected eye(s) once daily in the evening.
Single-dose container. Discard unused portion.
See carton for inactive ingredients.
Storage:Store at 20°C to 30°C (68°F to 86°F).
Protect from light. Store in the original pouch. After the pouch is opened, the single-use containers may be stored in the
opened foil pouch for up to 14 days.
Discard any unused containers 14 days after first opening the pouch.
Write down the date you open the pouch. __ / __ / __ZOLYMBUS™
(bimatoprost ophthalmic gel) 0.01%
For Topical Ophthalmic Use
10 single-dose containers (0.3 g each)
Rx only
Sterile - Contains no preservatives
Manufactured for Thea Pharma Inc.
Waltham, MA 02451Théa Logo
Lot:
EXP:

-
INGREDIENTS AND APPEARANCE
ZOLYMBUS
bimatoprost gelProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 82584-010 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BIMATOPROST (UNII: QXS94885MZ) (BIMATOPROST - UNII:QXS94885MZ) BIMATOPROST 0.1 mg in 1 g Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER TYPE B (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: HHT01ZNK31) POLYETHYLENE GLYCOL 4000 (UNII: 4R4HFI6D95) SODIUM ACETATE (UNII: 4550K0SC9B) SODIUM HYDROXIDE (UNII: 55X04QC32I) SORBITOL (UNII: 506T60A25R) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 82584-010-30 3 in 1 CARTON 02/26/2026 1 NDC: 82584-010-81 10 in 1 POUCH 1 0.3 g in 1 VIAL, SINGLE-DOSE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 2 NDC: 82584-010-10 1 in 1 CARTON 02/26/2026 2 10 in 1 POUCH 2 0.3 g in 1 VIAL, SINGLE-DOSE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 3 NDC: 82584-010-35 3 in 1 CARTON 02/26/2026 3 10 in 1 POUCH 3 0.3 g in 1 VIAL, SINGLE-DOSE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA217307 02/26/2026 Labeler - Thea Pharma Inc. (117787029)
Trademark Results [ZOLYMBUS]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ZOLYMBUS 98316660 not registered Live/Pending |
LABORATOIRES THEA 2023-12-15 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.