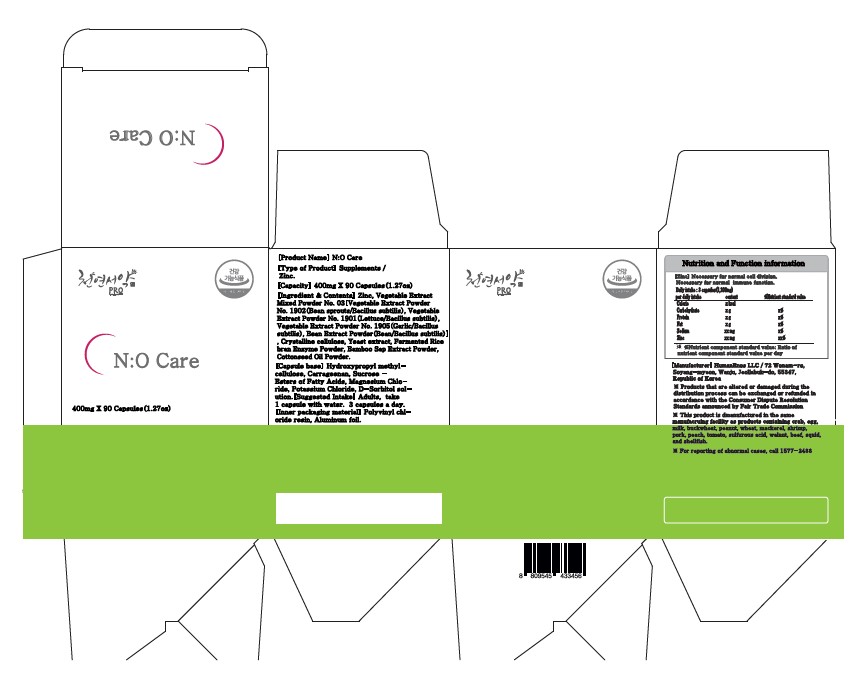
NO Care (Helps improve matabolic disorder, Lettuce fermented powder)
NO Care (Helps improve matabolic disorder, Lettuce fermented powder) by
Drug Labeling and Warnings
NO Care (Helps improve matabolic disorder, Lettuce fermented powder) by is a Otc medication manufactured, distributed, or labeled by HumanEnos LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
NO CARE (HELPS IMPROVE MATABOLIC DISORDER, LETTUCE FERMENTED POWDER)- nitric oxide, zinc, glutathione capsule
HumanEnos LLC
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
NO Care (Helps improve matabolic disorder, Lettuce fermented powder)
| NO CARE (HELPS IMPROVE MATABOLIC DISORDER, LETTUCE FERMENTED POWDER)
nitric oxide, zinc, glutathione capsule |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - HumanEnos LLC (695801540) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| HumanEnos LLC | 695801540 | manufacture(83202-2288) | |
Revised: 3/2025
Document Id: 3057bda9-17f4-6866-e063-6394a90a82ab
Set id: f515efa4-34b9-6414-e053-2995a90a79ce
Version: 3
Effective Time: 20250314
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.