Umcka Coldcare Lemon by ProSolutions, Inc. / Schwabe North America, Inc.
Umcka Coldcare Lemon by
Drug Labeling and Warnings
Umcka Coldcare Lemon by is a Homeopathic medication manufactured, distributed, or labeled by ProSolutions, Inc., Schwabe North America, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
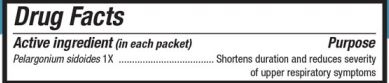
UMCKA COLDCARE LEMON- pelargonium sidoides powder
ProSolutions, Inc.
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
| UMCKA COLDCARE LEMON
pelargonium sidoides powder |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - ProSolutions, Inc. (070769782) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| ProSolutions, Inc. | 070769782 | label(83102-5146) , pack(83102-5146) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Schwabe North America, Inc. | 831153908 | manufacture(83102-5146) | |
Revised: 4/2025
Document Id: 326cbf07-4e79-f367-e063-6294a90a17f3
Set id: f5776f96-13fb-0570-e053-2a95a90aa12c
Version: 2
Effective Time: 20250410
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.